This web page was created as an assignment for an undergraduate course at Davidson College.
Find out more about Epstein-Barr Virus Here!
Figure 1: Chime Image of C-Terminal ATPase Domain of the Trasporter Associated with Antigen Processing (TAP).
http://www.rcsb.org/pdb/cgi/explore.cgi?pid=67091048181782&pdbId=1JJ7

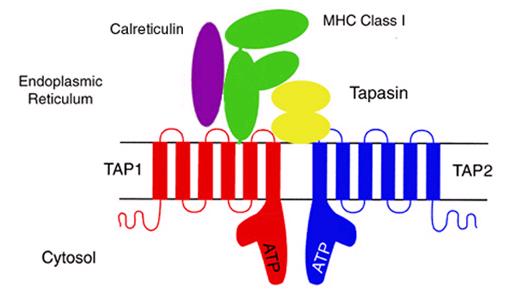
Figure 2: Peptide Loading Complex. The partially folded MHC I alpha chains and beta 2 microglobulin maintain their conformation by binding to Calreticulin. Tapasin interacts with both the partially folded MHC I and TAP1:TAP2 to facilitate peptide loading.
Figure courtesy of Dr Malini Raghavan
http://www.med.umich.edu/microbio/images/Figure1.jpg
Peptide Binding to TAP.
Peptides comprised of eight to sixteen amino acids bind to TAP with similar affinity. However, peptide translocation occurs most often for peptides eight to twelve amino acids in length. Experiments have shown peptides of up to 40 amino acids to translocate through the TAP complex. It appears that TAP binds peptides that are of optimal length, or slightly larger than those presented by MHC I. MHC I molecules generally load peptides of 8 to 10 amino acids long (Hewitt et al, 2001).
Barden MJ, Purcell AW, Gorman JJ, McCluskey J. 2000. Tapasin-Mediated Retention of Optimization of Peptide Ligands During Assembly of Class I Molecules. The Journal of Immunology. 165: 322-330.
de la Salle H, et al. 1999. HLA Class I Deficiencies Due to Mutations in Subunit 1 of the Peptide Transporter TAP1. The Journal of Clinical Investigation. 103: R9-R13.
Gaudet R, Wiley DC. 2001. Structure of the ABC ATPase Domainof human TAP1, the Transporter Associated with Antigen Processing.. The EMBO Journal. 20: 4964-4972.
Hewitt EW, Gupta SS, Lehner PJ. 2001. The Human Cytomegalovirus Gene Product US6 Inhibits APT Binding by TAP. The EMBO Journal. 20: 387-396.
Janeway CA, Travers P, Walport M, Shlomchik M. Immunobiology: The Immune System in Health and Disease. 5th Edition. New York: Garland Publishing.
Lacaille VG, Androwlewicz MJ. 1998. Herpes Simplex Virus Inhibitor ICP47 Destablilizes the Tranporter Associated with Antigen Processing (TAP) Heterodimer. Journal of Biological Chemistry. 273: 17386-17390.
Lankat-Buttgereit B, Tampe R. 2002. The Transporter Associated With Antigen Processing: Function and Implications in Human Diseases. Physiological Review. 82: 187-204.
Protein Data Bank. <http://www.rcsb.org/pdb/cgi/explore.cgi?pid=67091048181782&pdbId=1JJ7> Accessed 17 March 2003.
Raghuraman G, Lapinski PE, Raghavan M. 2002. Tapasin Interacts with the Membrane-spanning Domains of Both TAP Subunits and Enhances the Structural Stability of TAP1-TAP2 Complexes. Journal of Biological Chemistry. 277: 41786-41794.
For more information on the TAP complex, click here.
This page was created by Elizabeth Spitz
Davidson Biology Department Home Page