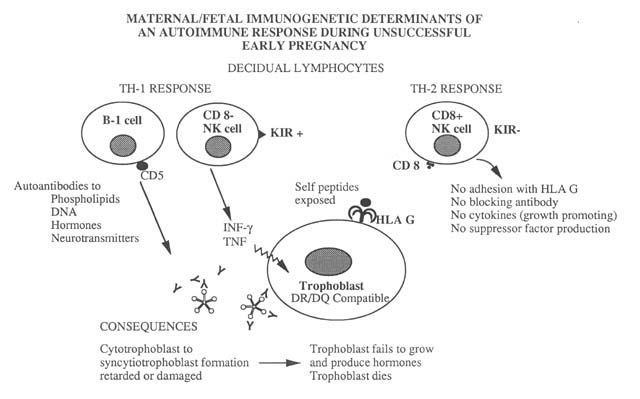
Possible autoimmune (ie-against fetal proteins expressed in self-MHC) pathways with pregnancy. Used with permission of authors. Source: http://www.repro-med.net/papers/normpreg.html
What is HLA-G?
What does HLA-G do?
Alloimmune
protection
Pathogen
protection
Autoimmune
protection
Where is it found?
Problems with HLA-G
What is HLA-G?
Those familiar with allogenic graft rejection may wonder
why the fetus, immunologically distinct from the mother, is not rejected
by the maternal immune system. HLA-G may be part of the fetal protection
system. HLA-G has a classic HLA-Class I structure, with a1, a2, a3
domains, covalently associated with a b2 microglobulin domain (Le Gal et
al, 1999). Alternate splicing provides both soluble and membrane
bound forms of HLA-G, with two soluble isoforms and four membrane bound
isoforms (O'Callaghan and Bell, 1998) (Le Bouteiller et al, 1999).
So what does HLA-G do?
Alloimmune protection
One function is to decrease or prevent the
maternal alloimmune attack on the fetus' paternally inherited MHC (Le Bouteiller
et al, 1999). HLA-G inhibits both the antigen-specific cytotoxic lymphocyte
(CTL) response and decreases NK cell function (Le Gal et al., 1999; O'Callaghan
and Bell, 1998). Thus, the low variability may be sufficient to
present antigen fragments (O'Callaghan and Bell, 1998). In comparison
to MHC-I and MHC-II molecules, there has been little evolutionary pressure for
HLA-G to evolve greater variability because of the limited number of pathogens,
and there has been pressure to not evolve variability, to avoid maternal autoimmune
reactions to the fetus (Le Bouteiller and Blaschitz, 1999).
Autoimmune Protection
Autoimmune reaction occurs when the appropriate proportion
of CD4:CD8 is disrupted (Beer and Kwak, 1999 http://www.repro-med.net/papers/normpreg.html)
Changes in the proportion of CD4:CD8 inhibit NK cell adhesion to HLA-G
producing an autoimmune response to the placental trophoblast and termination
of the developing conceptus (Beer and Kwak, 1999 http://www.repro-med.net/papers/normpreg.html).
Such losses are usually repetitive (Beer and Kwak, 1999 http://www.repro-med.net/papers/normpreg.html).

Possible autoimmune (ie-against fetal proteins expressed in self-MHC)
pathways with pregnancy. Used with permission of authors. Source:
http://www.repro-med.net/papers/normpreg.html
So how does HLA-G inhibit the maternal anti-fetal
immune response? Maternal NK cells have at least three receptors that
recognize HLA-G. One receptor, p49, is a member of the human killer inhibitory
receptor (KIR) group (Lopez-Botet, Navarro and Llano, 1999). These KIRs
have cytoplasmic immunoreceptor tyrosine-based inhibitory motifs (ITIM) that
when phosphorylated recruit protein tyrosine phosphatases that are involved
in the downregulation of NK activity (Lopez-Botet, Navarro and Llano, 1999).
The other two identified receptors, LIR-1/ILT-2 and ILT-4, are members of the
Leukocyte Ig-like receptors (LIR) or Ig-like transcript (ILT) families (Le Bouteiller
and Blaschitz, 1999). Both receptors are NK-cell response inhibitory
(Le Bouteiller and Blaschitz, 1999). Receptor activation or deactivation
may initiate one of two cytokine pathways: the Th1
response, which is "bad for pregnancy", or the Th2 response,
which is "good for pregnancy"(Somigliana, Vigano and Vinali, 1999).
Where is HLA-G found?
One of HLA-G's most interesting qualities is its localization.
HLA-G is expressed on the fetal extravillous trophoblasts, placental macrophages
and the endothelial cells in the core of the mesenchymal chorionic villi
(Le Bouteiller et al, 1999 (Le Bouteiller and Blaschitz, 1999) (Loke,
Hiby and King, 1999). Soluble HLA-G is found in the amniotic fluid
(Le Bouteiller and Blaschitz, 1999). The trophoblast cells are immunologically
neutral: they express neither MHC-I nor MHC-II molecules (Loke, Hiby
and King, 1999). Cells lacking both MHC's are usually destroyed;
however, HLA-G inhibits this response.
In the image below, cell types that express HLA-G on
their surface are outlined in magenta.
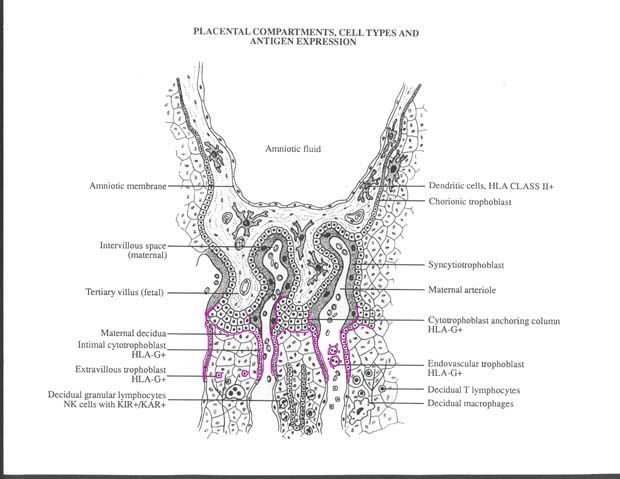
Physiology of maternal/fetal interface: placental villi invasion of endometrium. Original does not have magenta highlights. Used with permission of authors. Source: http://www.repro-med.net/papers/normpreg.htmlblems with HLA-G
Spontaneous Abortions
Women with high NK cell activity levels pre-conception
have a 3.5 greater risk of spontaneous abortion than women with normal
NK cell activity levels (Somigliana, Vigano and Vinali, 1999). However,
there was no significant difference when the raw numbers of NK cells were
compared between women who had recurrent spontaneous abortion and women
who had normal pregnancies (Emmer et al, 1999). It would seem the
difference was related to quality, not quantity. Women who are immunized
against their husband's leukocytes may decrease their NK cell activity
against paternally inherited MHC molecules (Somigliana, Vigano and Vinali,
1999). Women who underwent this immunotherapy, and whose NK cell
activity was successfully lowered had successful pregnancies (Somigliana,
Vigano and Vinali, 1999).
Problems with CD4:CD8 ratios also result in repetitve
losses of pregnancy (Beer and Kwak, 1999 http://www.repro-med.net/papers/normpreg.html).
Tumors
As HLA-G helps the fetus escape immunological regulation, HLA-G
could confer a similar immunity to tumor cells. LM Real et al examined
samples of solid tumor tissues (melanoma, breast, colon, larynx) and tumor cell
lines (melanoma, breast, colon, cervix, pancreas, lung, leukemia and lymphoma)
for HLA-G mRNA and surface expression (1999). Most samples had HLA-G mRNA,
but none had surface-expressed HLA-G molecules. However, they only used monoclonal
antibodies to two isoforms of HLA-G -- it is possible that the tumors expressed
one of the two other isoforms (Real et al, 1999).
Pre-Eclampsia
Placental HLA-G levels are significantly lower or absent in pre-eclamptic
placentas than in normal placentas (Le Bouteiller et al, 1999).
Now you can return to the Molecular Biology Home Page, or Immunology Home Page.
Return to Davidson College Biology Department Home Page
Return To Biology Course Materials