~ This web page was produced as an assignment for an undergraduate course at Davidson College ~
Gene Guns
Background:
The gene gun was created as a new way of proceeding with gene transformation of grains in response to the difficulty of getting bacteria across grain cell walls. It was designed by John Sanford at Cornell University in 1987 and made introducing new genetic material into plant cells much easier than previous methods, such as the use of viruses or agrobacterium. The gene gun has a wide range of uses today and can be used on many organisms such as bacteria, yeasts, and mammalian cell lines, particularly those which have previously been difficult or impossible to transfect such as non-dividing cells or primary cells. The transformation does not apply only to unicellular organisms but also whole objects such as leaves or entire animals: Drosophila and mice (Wetterauer, Brigit et al.). It has been particularly useful for chloroplasts as well because no bacteria or viruses were known to infect chloroplasts and this method has allowed for a way to introduce foreign DNA into the chloroplasts.
There are three common methods of genetic engineering: the plasmid method, the vector method, and the biolistic (gene gun) method. The most well known of the three is the plasmid method, which is generally used for altering microorganisms such as bacteria. The vector method is similar to the plasmid method, but its products are inserted directly into the genome via a viral vector. The third method is the biolistic method which will be studied in detail below. For more detailed explanations of the plasmid and vector methods of genetic engineering, visit ThinkQuest .
Refer to this Table comparing the efficiencies of transfection methods such as retroviruses, adenoviruses, liposomes, direct DNA injection, and the biolistic method. The efficiency of the gene gun system varies, with skin cells showing the greatest uptake of 10–20% (Yang, 1990). This system has many modern uses and has been used to deliver a nucleic acid-based hepatitis B vaccine to both mice and humans, and is presently in clinical trials (Mumper, 2001).
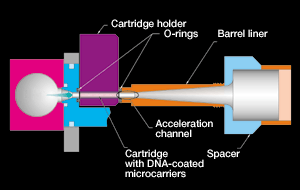
How the Gene Gun Works: The gene gun is part of a method called the biolistic (also known as bioballistic) method, and under certain conditions, DNA (or RNA) become “sticky,” adhering to biologically inert particles such as metal atoms (usually tungsten or gold). By accelerating this DNA-particle complex in a partial vacuum and placing the target tissue within the acceleration path, DNA is effectively introduced (Gan, 1989). Uncoated metal particles could also be shot through a solution containing DNA surrounding the cell thus picking up the genetic material and proceeding into the living cell. A perforated plate stops the shell cartridge but allows the slivers of metal to pass through and into the living cells on the other side. The cells that take up the desired DNA, identified through the use of a marker gene (in plants the use of GUS is most common), are then cultured to replicate the gene and possibly cloned. The biolistic method is most useful for inserting genes into plant cells such as pesticide or herbicide resistance. Different methods have been used to accelerate the particles: these include pneumatic devices; instruments utilizing a mechanical impulse or macroprojectile; centripetal, magnetic or electrostatic forces; spray or vaccination guns; and apparatus based on acceleration by shock wave, such as electric discharge (Christou and McCabe, 1992).


Above left: gold particles used in gene gun. Above right: tungsten particles used in the gene gun.
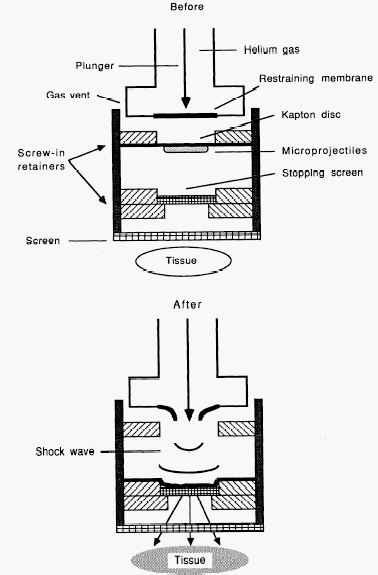
Above: Sketch of how a hand-held helium powered gene gun works by pushing DNA coated particles into tissue. (Images from Williams, 1991)
There are several variables in these experiments that must be controlled in order to attain maximal transformation efficiency. Optimal responses have been shown to be dependent on the delivery of a sufficient number of DNA-coated particles, as well as how much DNA coats the metal particles (Eisenbraun, 1993). Temperature, amount of cells, and their ability to regenerate also has an effect on the overall efficiency, as well as the type of gun used: helium powered vs. gun-powder, hand-held vs. stand-alone, etc. It is also important to adjust the length of the flight path of the particles: fragile tissues cannot be bombarded at the same high speed as those which have more resistance to foreign particles entering. How to adjust these variables depends mainly on which metal particles you are using to transfer the genetic material, and what type of cells you are attempting to transfect.
Significance: Another important use of the DNA gun involves the transformation of organelles as mentioned above: chloroplasts, as well as yeast mitochondria. The ability to transform organelles is significant because it enables researchers to engineer organelle-encoded herbicide or pesticide resistances in crop plants and to study photosynthetic processes. DNA delivery with the gene gun also offers new advantages for research in such areas as DNA vaccination/genetic immunization, gene therapy, tumor biology/wound healing, plant virology and many others.
The major limitations are the shallow penetration of particles, associated cell damage, the inability to deliver the DNA systemically, the tissue to incorporate the DNA must be able to regenerate, and the equipment itself is very expensive. An objection to this method is that the DNA could be inserted into a working gene in the plant and many of the public worry that this new DNA could then be transferred to wild plants as well and resistance could be conferred to weeds or insects. (ThinkQuest)