This web page was produced as an assignment for an undergraduate course at Davidson College
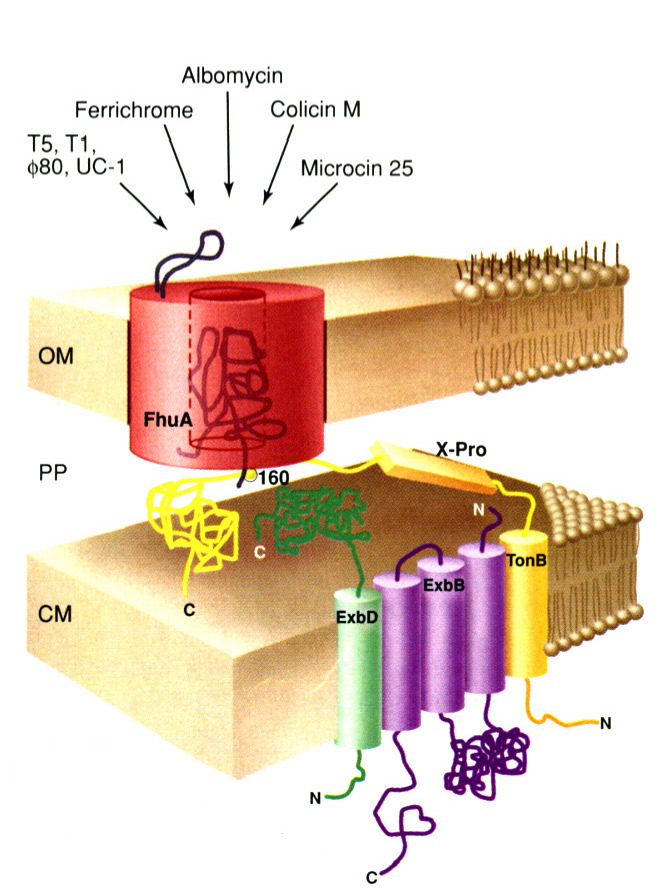
FhuA is a monomeric protein found in the outer membrane of Escherichia coli. It acts as an energy dependent protein channel whose primary function is to transport siderophore chelated ferrichrome-iron across the outer membrane of E. coli. It derives energy from a Ton based protein complex (consisting of TonB, ExbB, and ExbD) located in the cytoplasmic membrane of E. coli; this complex converts electrochemical potential energy form the cytoplasmic membrane into energy usable by the FhuA protein in the outer membrane (Ferguson et al 1998).
In addition to acting as a ferrichrome-iron receptor, FhuA also acts as a primary receptor for the antibiotic albomycin (which is structurally similar to ferrichrome-iron), four bacteriphages (T1, T5, UC-1, and f80), the peptide antibiotic microcin 25, and the bacterial toxin colicin M (Braun 1998).

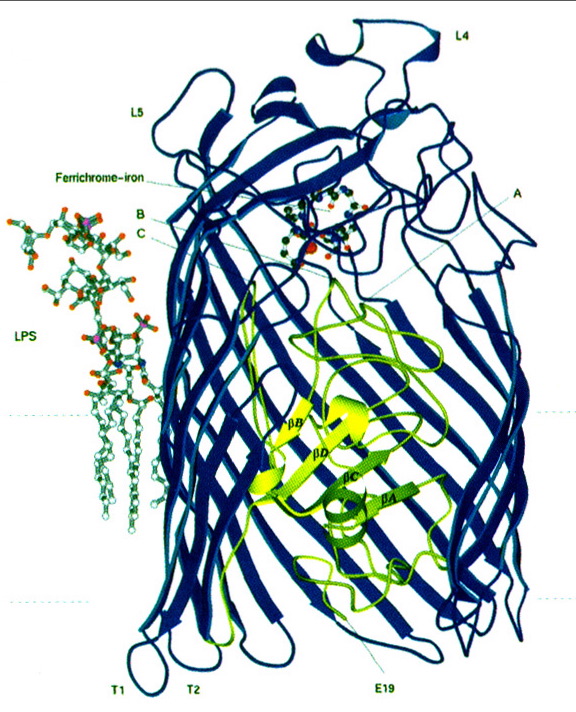
FhuA consists of two main regions: a "barrel" and a "plug" region. The hollow barrel region (consisting of residues 160-714) makes up the outer surface of the protein, while the plug region (consisting of residues 19-159) is located within the barrel region (Locher et al 1998). The barrel region of FhuA is similar to that of other porin proteins (Weiss et al); however, the presence of a plug region which obstructs the entire open, barrel region of the protein is completely unprecedented (Locher et al 1998). On the extracellular surface of the FhuA protein, there is a loop, known as loop 4, which protrudes quite far into the extracellular space. Bacteriophages bind to this loop prior to injecting their DNA into E. coli (see Figure 1); it is also suspected that loop 4 may serve some purpose in blocking the ferrichrome binding site when necessary (Braun 1998).
The outer surface of the FhuA plug interacts quite extensively with the inner surface of the FhuA barrel (there are nine salt bridges and over 60 hydrogen bonds between the two surfaces), and the upper region of the plug is exposed to the extracellular space. It is to this upper region of the plug that the ferrichrome-iron molecule binds. The top of the plug contains a pocket, into which the head of the ferrichrome molecule fits perfectly (see Figure 2). Furthermore, strong hydrogen bonds are formed between the ferrichrome substrate and FhuA within this pocket. These interactions between FhuA and ferrichrome help to explain the strong affinity between the two molecules (Locher et al 1998).
As soon as the ferrichrome-iron molecule binds, structural changes begin to occur throughout the protein that eventually lead to the interaction between FhuA and the Ton protein complex located in the cytoplasmic membrane of the cell. The binding of the two molecules forces a vertical shift in the backbones of two helices within the plug region of FhuA. This structural alteration then causes the unfolding of another helix located in the periplasmic side of the plug region, which is thought to be substantial enough to cause the interactions between the NH2-terminus of the FhuA protein and the Ton protein complex. It is thought that after the Ton protein complex is activated, it interacts even further with the plug region of the FhuA protein to create an opening in the plug region through which the ferrichrome-iron molecule can travel (Locher et al 1998).
The exact sequence of events involved when other ligands or bacteriophages bind to FhuA have not yet been fully explored. It has been postulated, however, that since bacteriophages must inject double stranded DNA (which is much larger than the ferrichrome-iron molecule) into the cell, they more than likely displace the plug completely by means of physical force. The antibiotic albomycin (which, as mentioned previously, is structurally similar to ferrichrome-iron) is more than likely mistaken by FhuA for ferrichrome-iron; thus the process of albomycin uptake is likely very similar to that of ferrichrome-iron uptake. Similar to phage DNA, the bacterial toxin colicin M is also much larger than the opening created in the plug region of FhuA thorough which ferrichrome-iron can travel; thus Ton protein comlex mediated entry is not very likely. However, at the present, it is completely unknown how this toxin enters the cell (Locher et al 1998).
Braun V. 1998. Pumping Iron Through Cell Membranes. Science . 282: 2202-2203.
Ferguson AD, Hofmann E, Coulton JW, Diederichs K, Welte, Wolfram. 1998. Siderophore-Mediated Iron Transport: Crystal Structure of FhuA with Bound Lipopolysaccharide. Science . 282: 2215-2220.
Locher KP, Rees B, Koebnik R, Mitschler A, Moulinier L, Rosenbusch JP, Moras D. 1998. Transmembrane Signaling across the Ligand-Gated FhuA Receptor: Crystal Structures of Free and Ferrichrome-Bound States Reveal Allosteric Changes. Cell. 95: 771-778.
Weiss MS, Abele U, Weckesser J, Welte W, Schiltz E, Schultz GE. 1991. Molecular Architecture and Electrostatic Poperties of a Bacterial Porin. Science. 254: 1627-1630.