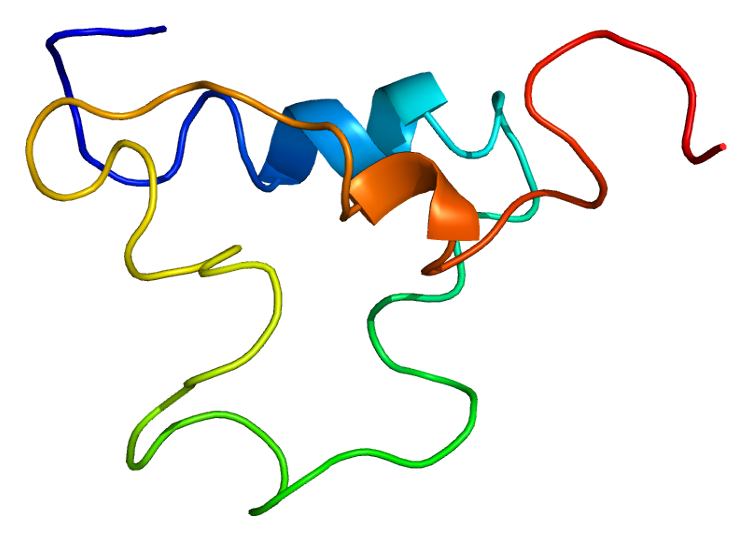
Figure 1: The 3-dimensional structure of IGF-1. Permission granted for image by Wikimedia Commons.
Goal: Elucidate the relationship between the structure and function of this critical protein
Human Insulin-Like Growth Factor 1,
or IGF-1, as it is typically called, is a small protein that plays an
extremely important role in the functioning of the human body. IGF-1 is
classified as a somatomedin - a hormone involved in cellular growth
(Rinderknecht and Humbel, 1978). IGF-1 is a single polypeptide
composed of 70 amino acids, with a molecular weight of 7649
(Rinderknecht and Humbel, 1978). The goal of this page is to connect
this protein's structure to its function; to do that, we must first
have a detailed understanding of IGF-1's structure. The linear sequence
of amino acids (primary structure) plays a large role in determining
the structure of the protein - the amino acids will interact in a way
unique to the sequence. The tertiary
(3rd level) of protein structure is determined by R-group interactions
between amino acids. Three disulfide bridges (connections involving the
amino acid cystine) help to determine the shape of this protein
(Rinderknecht
and Humbel, 1978).
A visual representation of disulfide bridges:
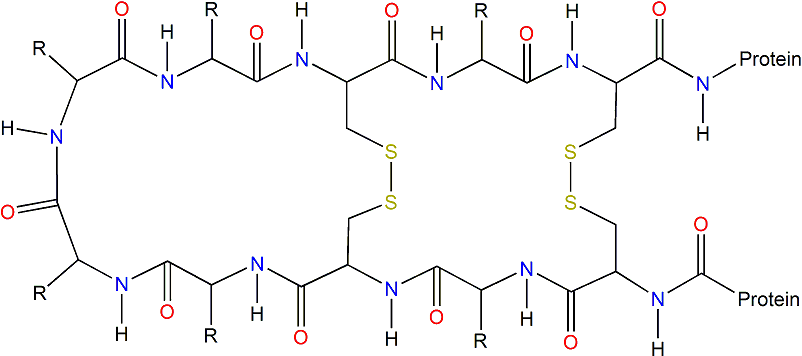
Figure 2: Disulfide bridges linking two cystine amino acids. Permission pending from European Bioinformatics Institute.
Rinderknecht and Humbel noticed a strong similarity between the amino acid sequences of IGF-1 and human insulin (1978). These researchers speculated that the conserved regions (specifically a 6 cystine stretch and three glycine amino acids) were especially important for IGF-1's 3-dimensional structure (rinderknecht and Humbel, 1978).
Narhi et al. state that the 3 disulfide bridges in Human IGF-1 are necessary for the protein to carry out its normal function (1993). The three disulfide bridges are formed between cystines at amino acid positions 6 and 48, 18 and 61, and 47 and 52 (Narhi et al., 1993). Without any of the 3 bridges, there is a significant decrease in binding with the IGF receptor (Narhi et al., 1993). These are logical and important conclusions - when the structure of IGF-1 changes, it cannot effectively bind and its activity is disrupted. Clearly, the disulfide bridges shape the protein into a conformation that allows it to bind with receptors.
We now have a firm grasp on the structure of IGF-1. But what exactly does it do, and how does the structure of this protein impact its ability to function?
IGF-1 is produced by liver cells when stimulated by human growth hormone (Delafontaine et al., 2004). IGF-1 is another hormone (a somatomedin), and it plays a large role in the process of cell growth (Delafontaine et al. 2004). Signal transduction is a hugely important theme in cellular biology, and IGF-1 does not deviate from that rule - the protein works by binding to a receptor (insulin-like growth factor 1 receptor, or IGF-1R) and causing a reaction (Delafontaine et al., 2004).
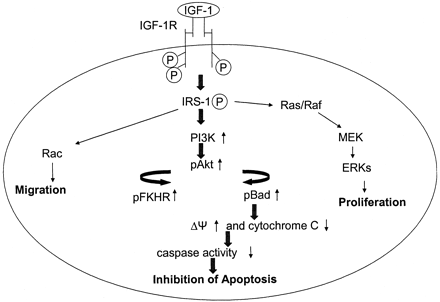
IGF-1 binds to IGF-1R, a transmembrane receptor protein. IGF-1R is made up of 2 a chains and 2 b chains, which are extracellular and cytoplasmic, respectively (Delafontaine et al., 2004). The cytoplasmic portion of the receptor is a tyrosine kinase (an enzyme that phosphorylates a substrate), activated when IGF-1 binds to its extracellular domains (Delafontaine et al., 2004). When IGF-1R is bound by IGF-1, a conformational change in the shape of the protein most likely occurs. It is this change that activates IGF-1R, phosphorylating different proteins responsible for cell movement, growth and reproduction, and for stopping potential apoptosis (Delafontaine et al., 2004).
These interactions are only possible because of the 3-dimensional structure of Human Insulin-Like Growth Factor 1. Any deviations from the specific shape and conformation would prevent IGF-1 from binding to IGF-1R. The exact primary, secondary, and tertiary structure (most importantly, perhaps, the 3 disulfide bridges) are essential for these two proteins to interact.
References:
Delafontaine P, YH Song, and Li Y. 2004. Expression, regulation, and function of IGF-1, IGF-1R, and IGF-1 binding proteins in blood vessels. Arteriosclerosis, Thrombosis, and Vascular Biology 24: 435-444.
Rinderknecht E, R Humbel. 1978. The amino acid sequence of human insulin-like growth factor 1 and its structural homology with proinsulin. The Journal of Biological Chemistry 253-8: 2769-2776.
Narhi LO, QX Hua, T Arakawa, GM Fox, L Tsai, R Rosenfeld, P Holst, JA Miller, Weiss MA. 1993. Role of native disulfide bonds in the structure and activity of insulin-like growth factor 1: genetic models of protein-folding intermediates. Biochemistry 32: 5214-5221.
Michael Rydberg's Molecular Homepage
Molecular Biology Homepage
A visual representation of disulfide bridges:

Rinderknecht and Humbel noticed a strong similarity between the amino acid sequences of IGF-1 and human insulin (1978). These researchers speculated that the conserved regions (specifically a 6 cystine stretch and three glycine amino acids) were especially important for IGF-1's 3-dimensional structure (rinderknecht and Humbel, 1978).
Narhi et al. state that the 3 disulfide bridges in Human IGF-1 are necessary for the protein to carry out its normal function (1993). The three disulfide bridges are formed between cystines at amino acid positions 6 and 48, 18 and 61, and 47 and 52 (Narhi et al., 1993). Without any of the 3 bridges, there is a significant decrease in binding with the IGF receptor (Narhi et al., 1993). These are logical and important conclusions - when the structure of IGF-1 changes, it cannot effectively bind and its activity is disrupted. Clearly, the disulfide bridges shape the protein into a conformation that allows it to bind with receptors.
We now have a firm grasp on the structure of IGF-1. But what exactly does it do, and how does the structure of this protein impact its ability to function?
IGF-1 is produced by liver cells when stimulated by human growth hormone (Delafontaine et al., 2004). IGF-1 is another hormone (a somatomedin), and it plays a large role in the process of cell growth (Delafontaine et al. 2004). Signal transduction is a hugely important theme in cellular biology, and IGF-1 does not deviate from that rule - the protein works by binding to a receptor (insulin-like growth factor 1 receptor, or IGF-1R) and causing a reaction (Delafontaine et al., 2004).

Figure 3: IGF-1
binding to IGF-1R (a transmembrane protein) and the signal transduction
that occurs. Permission pending from Dr. Patrice Delafontaine.
IGF-1 binds to IGF-1R, a transmembrane receptor protein. IGF-1R is made up of 2 a chains and 2 b chains, which are extracellular and cytoplasmic, respectively (Delafontaine et al., 2004). The cytoplasmic portion of the receptor is a tyrosine kinase (an enzyme that phosphorylates a substrate), activated when IGF-1 binds to its extracellular domains (Delafontaine et al., 2004). When IGF-1R is bound by IGF-1, a conformational change in the shape of the protein most likely occurs. It is this change that activates IGF-1R, phosphorylating different proteins responsible for cell movement, growth and reproduction, and for stopping potential apoptosis (Delafontaine et al., 2004).
These interactions are only possible because of the 3-dimensional structure of Human Insulin-Like Growth Factor 1. Any deviations from the specific shape and conformation would prevent IGF-1 from binding to IGF-1R. The exact primary, secondary, and tertiary structure (most importantly, perhaps, the 3 disulfide bridges) are essential for these two proteins to interact.
References:
Delafontaine P, YH Song, and Li Y. 2004. Expression, regulation, and function of IGF-1, IGF-1R, and IGF-1 binding proteins in blood vessels. Arteriosclerosis, Thrombosis, and Vascular Biology 24: 435-444.
Rinderknecht E, R Humbel. 1978. The amino acid sequence of human insulin-like growth factor 1 and its structural homology with proinsulin. The Journal of Biological Chemistry 253-8: 2769-2776.
Narhi LO, QX Hua, T Arakawa, GM Fox, L Tsai, R Rosenfeld, P Holst, JA Miller, Weiss MA. 1993. Role of native disulfide bonds in the structure and activity of insulin-like growth factor 1: genetic models of protein-folding intermediates. Biochemistry 32: 5214-5221.
Michael Rydberg's Molecular Homepage
Molecular Biology Homepage
Please contact me with questions or comments at mirydberg@davidson.edu