This web page was produced as an assignment for an undergraduate course at Davidson College.

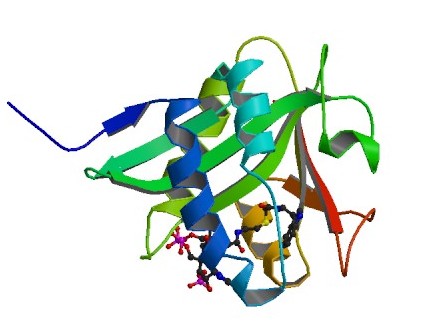
Figure 1. Serotonin N-Acetyltransferase coupled with a bisubstrate analog. Picture obtained from Protein Data Bank.
A. Why is Serotonin N-Acetyltransferase important?
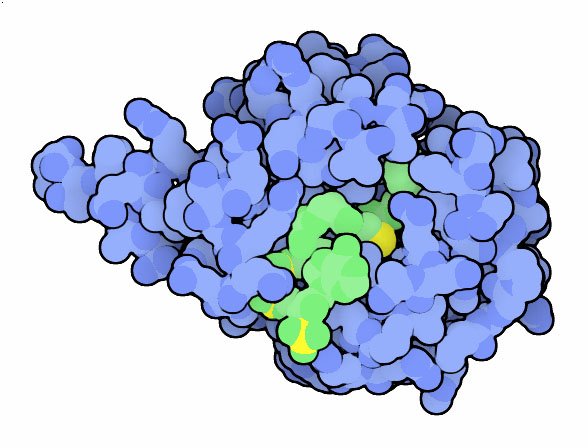 Before we can answer this question, we need to understand the importance of the hormone melatonin (Figure 2.) Melatonin is produced in the pineal gland and it plays a crucial role in regulating our bodies circadian rythms; high levels (~10 fold increase) of melatonin signify night time while low levels of it signify day time (Hickman et al., 1999).Such changes in the levels of melatonin have allowed vertebrate organisms to adapt to the changes that occur in their environments in the span of a 24-hour cycle (Zheng and Cole, 2002).
Before we can answer this question, we need to understand the importance of the hormone melatonin (Figure 2.) Melatonin is produced in the pineal gland and it plays a crucial role in regulating our bodies circadian rythms; high levels (~10 fold increase) of melatonin signify night time while low levels of it signify day time (Hickman et al., 1999).Such changes in the levels of melatonin have allowed vertebrate organisms to adapt to the changes that occur in their environments in the span of a 24-hour cycle (Zheng and Cole, 2002).
Serotonin N-Acetyltransferase, also known as arylalkylamine N-acetyltransferase (hereafter called AANAT), is one of the several enzymes that is involved in the metabolic pathway that is responsible for the creation of melatonin from tryptophan (Figure 3) (Zheng and Cole, 1187). It catalyzes the addition of an acetyl group, which it receives from the acetyl coenzyme A (AcCoA), to serotonin (Hickman et al., 1999). AcCoA is the first to bind to the AANAT and then serotonin completes the complex; serotonin undergoes acetylation and N-acetyltransferase and CoASH are released from enzyme (Hickman et al., 1999).More importantly, AANAT is the rate limiting enzyme in this pathway, which becomes apparent when levels of melatonin in the body are compared to AANAT levels. Like melatonin, the relative abundance of AANAT in the pineal gland fluctuates according to environmental light cues (Scheibner et al., 2002) with its levels reaching a maximum during the night (~10 - ~100 increase)(Klein, 2006). At night with the absence of light, AANAT can double its activity in 15 minutes and with a sudden exposure to light it can halve its activity in 3.5 minutes (Klein, 2006). Identifying the specific molecular mechanisms underlying the catalysis of the acetylation of serotonin by AANAT will be essential in the creation of drugs that will treat sleep and mood disorders (Klein, 2006)
Figure 2.Space-fill version of Melatonin molecule. Picture obtained from PDB
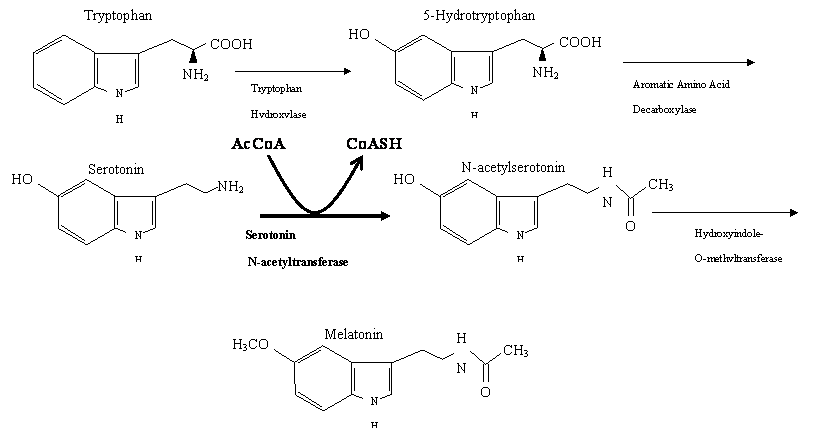
Figure 3. Metabolic pathway involved in the production of melatonin from tryptophan. Picture adapted from Zheng and Cole, 2002.
B. What is serotonin-N-acetyltransferase's basic structure?
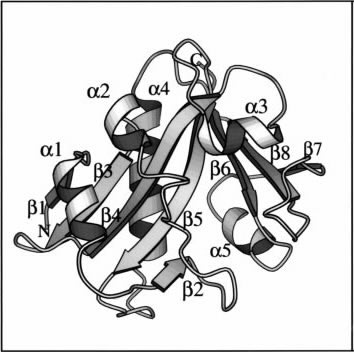
Figure 4. 3D structure of AANAT, emphasizing the secondary structure.a refers to a helices
and b refers to b sheets. Adapted from Hickman, Klein, and Dyda, 1999. Permission Pending.
AANAT is globular enzyme that is found in the cytoplasm; it is usually found as a monomer (Hickman, Klein, and Dyda, 1999) but, as Klein pointed out in his review, it can also form a "reversible regulatory complex with 14-3-3 proteins" (2006). As it can be seen in Figure 4, the protein has two groups of b sheets, (b1,b3,b4,b5,b2 and b7,b8,b6) which form V-shape cleft with a point of convergence at sheets b5 and b6. These strands of sheets are enclosed by five a helices (a1,a2,a3,a4,a5), when these helices adopt their final conformation they also create three loops which as I shall explain later play a very important role in the proteins catalytic activity (Hickman, Klein, and Dyda, 1999). AANAT's structural confirmation has four unique motifs (motifs A, B, C, D) that identifies it as a member of a larger superfamily of acetyltransferases, known as the GNAT family (Figure 5)(Hickman et al., 1999).
C. How does the structure of the serotonin N-acetyltransferase affect it's catalytic activity?
Like in any other enzyme, AANAT's catalytic depends upon the successful binding of its substrates to the active site, in this case the binding of AcCoA. and serotonin.The loops 1, 2 and 3, which I mentioned above form a "funnel-shaped pocket" where the active site is found(Klein, 2006) and the lining of this "funnel" is dominated by hydrophobic amino acids(Hickman, Klein, and Dyda, 1999). The way these three loops are arranged allows only for very small molecules to enter the hydrophobic funnel and get to the active site (Hickman, Klein, and Dyda, 1999). In addition, loop 1 in the absence of AcCoA is positioned in such a way that it blocks access to the enzyme's active sited(Hickman, Klein, and Dyda, 1999). Hickman et al. suggested that loop1 is flexible enough that once the coenzyme comes into contact with the AANAT, at the cleft created by sheets b5 and b6, the loop undergoes a major conformational alteration that reveals the rest of the active site and thus allows for the binding of both substrates(1999).
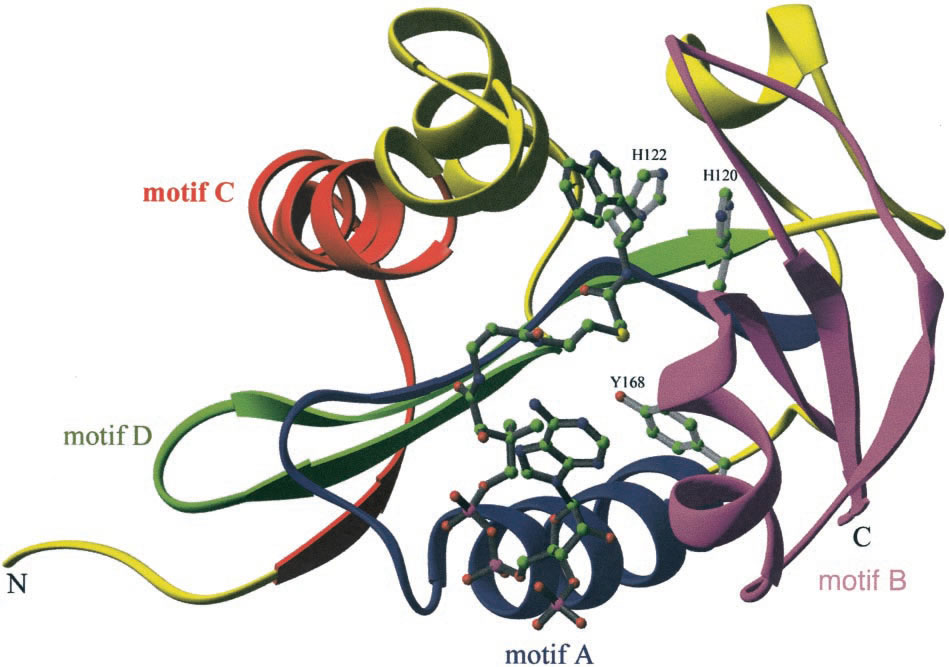
Figure 5. 3D structure of AANAT bound to bisubstrate analog, highlighting Histidine (H122 and H120)
and Tyrosine (Y168) residues. GNAT families motifs are also visible. Scheibner et al., 2002. Permission pending.
Lastly, researches have been trying to identify the specific residues in AANAT that are involved in the acetyltransferase activity of its active site. Three amino acids have received a great amount of attention and are considered major catalytic components of the enzyme; these are two histidine molecules at positions 120 and 122 and one tyrosine molecule at position 168 (Figure 5) (Schneibner et al., 2002). On the one hand, the histidine residues are thought to be indirectly involved in "the deprotonation of protonated amine" which is what initiates the transfer of the acetyl group to serotonin(Klein, 2006). On the other hand, the tyrosine residue is thought to be involved in the ejection of the CoASH molecule from the serotonin-AcCoA-AANAT complex after the transfer of the acetyl group (Klein, 2006).
Works Cited
Hickman AB, Klein DA, Dyda F. Melatonin Biosynthesis: The Structure of Serotonin N-Acetyltransferase at 2.5A° Resolution Suggests a Catalytic Mechanism. Molecular Cell 1999; 3: 23-32.
Hickman AB, Namboodiri MAA, Klein DA, Dyda F. The Structural Basis of Ordered Substrate Binding by Serotonin N-Acetyltransferase: Enzyme Complex at 1.8 A° Resolution with Bisubstrate Analog. Cell 1999; 97: 361-369.
Klein DC. Arylalkylamine N-Acetyltransferase: "the Timezyme". The Journal of Biological Chemistry 2006; 282: 4233-4237.
Scheibner KA, De Angelis J, Burley SK, Cole PA. Investigation of the Roles of Catalytic Residues in Serotonin N-Acetyltransferase. The Journal of Biological Chemistry 2002; 277: 18118-18126.
Zheng W, Cole PA. Serotonin N-Acetyltransferase: Mechanism and Inhibition. Current Medicinal Chemistry 2002; 9: 1187-1199.
Ilias Theodorou's Home Page Molecular Biology Home Page
For any questions or comments please email iltheodorou@davidson.edu