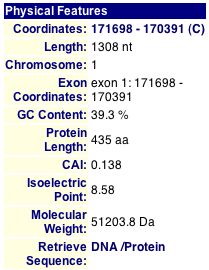
Fig. 1: Physical features of KIN3p. (MIPS, 2004; http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YAR018c&db=CYGD.)
My Favorite Yeast Expression
Annotated vs. Non-Annotated
This assignment, in a similar fashion as those before it, offers further
analysis of the genes KIN3 and FUN19. The previous assignments were aimed
at collecting data in hopes of learn more about these genes, namely their
individual characteristics and how they are expressed, but this assignment
focuses on the proteins that each of these genes produces. In this assignment
as in the previous two, the information collected in regards to KIN3 will
be analyzed in hopes of developing an understanding of how interpret the
information gathered about FUN19. From the information collected the goal
is to develop a hypothesis as to the function(s) of FUN19 protein. FUN19
is an non-annotated gene, which means that there is no published data on
this gene. Both KIN3 (YAR018c) and FUN19 (YAL034c) are genes belonging
to Saccharomyces cerevisiae located on chromosome I.
KIN3 / YAR018c
The molecular function of KIN3 lies in protein kinase activity, its biological process is chromosomal segregation, and its cellular component is unknown. KIN3 is responsible for the production of serine/threonine-protein kinase KIN3, which will be referred to as KIN3p throughout the rest of this assignment.
MIPS:
This database provides information on the molecular structure and functional network of the entirely sequenced, well-studied model eukaryote, the budding yeast Saccharomyces cerevisiae. In searching this database I was able gather basic information about KIN3p.

Fig. 1: Physical features of KIN3p. (MIPS, 2004; http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YAR018c&db=CYGD.)
The MIPS database also contained links to several PUBMED articles that
contained information in regards to KIN3 and KIN3p. The first paper I read
sought to identify the role of a proteins based on identified functional
gene interactions. To see this paper in its entirety click here http://www.genome.org/cgi/content/full/13/12/2568.
The conclusion that was derived in this article was that because KIN3 exhibited
a strong target-set similarity to GAS1, involved in cell wall organization
and biogenesis, and to BUD14, involved in bud site selection, then its
is likely that KIN3p is involved in cell growth, budding, or a closely
related processes.
Database for Interacting Proteins (DIP):
This database, as its name implies, provides information with regards to what genes produce proteins that interact with one another. This database was able to provide me with an interaction map, illustrating which proteins are involved in physical interactions with KIN3p.
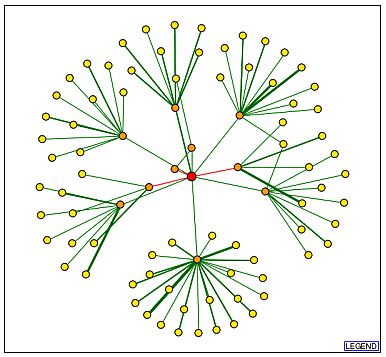
The genes producing proteins that directly interact with KIN3p, shown
above, are listed below in clockwise order, starting with the close orange
node that appears to be directly above KIN3, with their molecular function
listed next to them.
|
|
|
|
| SSZ1 | unfolded protein binding | cytoplasm |
| SNP1 | mRNA binding | commitment complex |
| RPS28 | structural component of a ribosome | cytosolic small ribosome subunit |
| ASC1 | unknown | cytoplasm |
| TEM1 | GTPase activity
protein binding |
spindle pole body |
| ADY3 | protein binding | prospore membrane spindle |
| NOG1 | GTPase activity | nucleolus |
| ZUO1 | unfolded protein binding | cytoplasm
cytosol mitochondrion ribosome |
| GTS1 | transcriptional activator activity | cytoplasm
nucleus |
| SRO77 | unknown | plasma membrane |
PathCalling:
This database is used to generate protein-protein interaction maps on a genome wide scale. This map does not show nearly as many interaction for KIN3 but it shows a much higher number of interactions for SNP1 and a comparable amount for TEM1. It seems odd that these two genes were shown and none of the others especially since, according to the thickness of the connecting lines in the previous map, there is not any stronger evidence for the interaction of KIN3 with TEM1 and SNP1 than there is with any of the other proteins.

I am guessing that the interaction that takes place between KIN3p
and the proteins of SNP1 and TEM1 are stronger than the other interactions
that were shown from the DIP database.
BENNO FIGURE 1:
Due to the large number of genes included and somewhat sporadic nature of connecting lines it is difficult to accurately interpret much information from this figure. The light yellow box that KIN3 is written in signifies that the role of this protein is unknown. Also, if you look closely it appears that there are two grey lines which stem from KIN3, if followed they run to TEM1 and SNP1. A grey line means that the localizations or cellular roles are unknown for one or both of the proteins. To view this map in its entirety click here: Benno Figure 1.
Expert Protein Analysis System (ExPASy) 2D:
This database is dedicated to the analysis of protein sequences and structures and contains a large sample of 2-D gels, including one of Saccharomyces cerevisiae. 2-D gel electrophoresis separates proteins by their isoelectric points (along the X-axis) and their molecular weight (along the Y-axis). Below is the 2-D gel of yeast with the red box enclosing the area where KIN3p should be present. Notice how these coordinates correspond with the data from the MIPS database (pI = 8.58 and MW = 51203.8 Da).
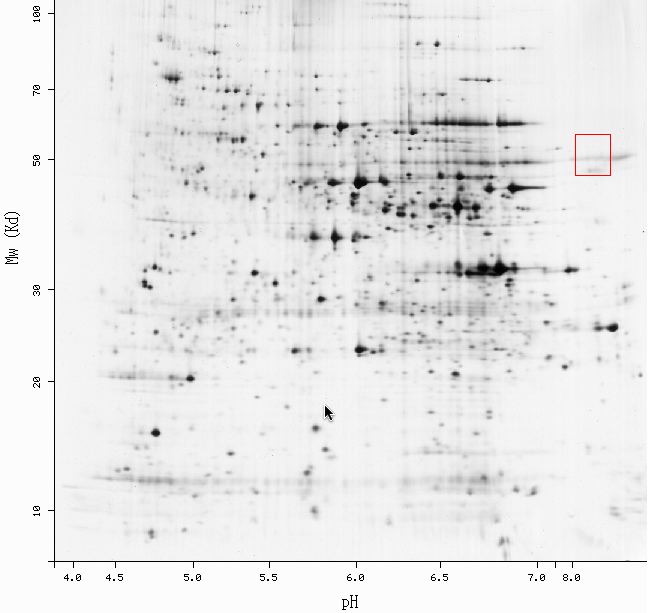
It seems as if KIN3p is not a protein that is produced in very large
quantities based on the fact that it appears as a faint streak on the 2-D
gel.
YEAST TWO-HYBRID ANALYSIS:
In searching this site the only two genes that were shown to interact
with KIN3 were SNP1 and TEM1. In both instances it was KIN3 that acted
as the prey with either SNP1 or TEM1 serving as the bait protein. In searching
the website with the additional Y2H results KIN3 also acted as the prey
for NOG1, SRO77, and GTS1 (twice). There was no data on either site
that had KIN3 being used as the bait protein. Links to the site of Y2H
Results and the Additional Y2H Results pages are http://depts.washington.edu/%7Eyeastrc/th_11.htm
and http://depts.washington.edu/%7Eyeastrc/th_12.htm
respectively.
Because the connection between KIN3 and SNP1 and TEM1 were placed
in the first website and the others were placed among the additional results
it leads me to believe that KIN3's interaction with TEM1 and SNP1 is stronger
than its interaction with the proteins on the additional page.
PROWL:
This database did not have very much information to offer. Below is some general information regarding KIN3p. This database has the ability to calculate the theoretical pI as well as the molecular weight but this information was already obtained.

PDF Files:
Under the Sequences column at the homepage of our text there are three pdf files. One that contains an interaction map of an extensive collection of genes involved in aging, another of an interaction map of the genes involved in degradation, and the final one is a map of the genes involved with membrane. KIN3 is present in all three of these maps but the design of the maps with many genes obstructing others makes it difficult to decipher very much if any information from them. The only information that can be accurately deciphered from these files is the role that each gene plays thanks to the schematic way in which the text boxes are colored. http://occawlonline.pearsoned.com/bookbind/pubbooks/bc_mcampbell_genomics_1/chapter6/deluxe.html
In the aging pdf file KIN3 was not assigned a function that the gene was involved in. This is most likely because KIN3's role in aging is still unknown but it is known to play some part or that it interacts with a gene of a known function.
In the degradation pdf KIN3 was also not assigned a function, which points to the same conclusion as was made before.
In the last pdf file, the one on membranes (no other information is
available as to whether the focus is membrane formation, maintenance, transport,
etc. is provided), KIN3 is shown to be involved in protein folding.
Based on all the information that was collected from all the different databases it seems as if the KIN3 protein functions within the nucleus. Although the nucleus is very large and simply stating that a protein functions there is about as descriptive as saying someone lives in the northern hemisphere, based on the cellular components of the proteins that KIN3p interacts with the two largest trends are the nucleus and the cytoplasm. The reason I believe KIN3p functions in the nucleus is because of its interaction with proteins that work with spindle, specifically TEM1. It seems unlikely that a protein that functions in the cytoplasm would travel into the nucleus to interact with these proteins. It seems much more plausible that KIN3p interacts with proteins that are traveling to the cytoplasm to carry out their molecular function.
Knowing that KIN3 is involved in chromosome separation, KIP3p has
been shown to interact with proteins function around spindle, and its involvement
in aging it seems highly probable that the KIN3 protein is involved in
mitosis, more specifically anaphase. Perhaps this protein aids in the binding
of spindle fibers to the centromere of sister chromatids. This hypothesis
also works along with the hypothesis developed from the PUBMED paper that
the KIN3 protein is involved in cell growth.
FUN19 / YAL034c
Since FUN19 is a non-annotated gene nothing is known about its molecular
function, the biological process it is involved in, or its cellular component.
The FUN in its name stands for Function Unknown. FUN19 is responsible for
producing the protein hypothetical 53.1 kDa protein in PYK1-SNC1 intergenic
region, which will be referred to as FUN19p throughout the rest of this
assignment.
MIPS:
As was done with the protein produced by KIN3, this website was used to gather basic information about FUN19p.
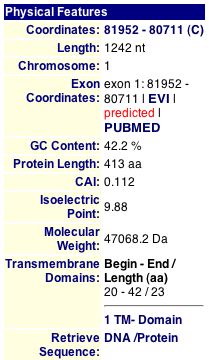
In searching through the MIPS database I searched through the PUBMED
articles pertaining to FUN19 but sadly they had very little information
to offer in regards to FUN19p.
DIP:
This database was used to find a protein-protein interaction map for FUN19p. The red lines signify that the results are unverified which means it is not certain that these interactions actually take place.
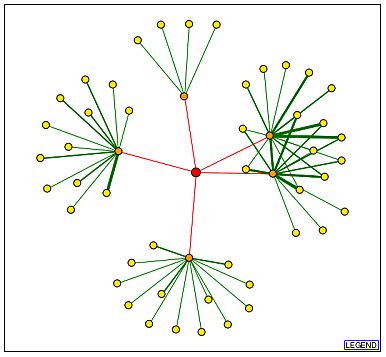
The genes producing proteins that directly interact with FUN19p are
listed below in clockwise order, starting with the orange node that appears
to be above FUN19p, with their molecular function and cellular components
listed next to them.
|
|
|
|
| ADH1 | alcohol dehydrogenase activity | cytosol |
| TAF1 | general RNA polymerase II transcription factor activity
histone acetyltransferase activity protein serine/threonine kinase activity |
transcription factor TFIID complex |
| TAF5 | general RNA polymerase II transcription factor activity | SAGA complex
SLIK complex transcription factor TFIID complex |
| KAP123 | protein carrier activity | cytoplasm
nuclear pore nucleus |
| SIN3 | histone deacetylase activity | SIN3 complex
histone deacetylase complex |
PathCalling:
This database was used to obtain a second protein-protein interaction map for FUN19. It appears as if this database requires a higher amount of evidence of an interaction taking place than the DIP database. According to PathCalling FUN19 does not interact with any other genes or protein.

This information causes me to strongly date the strength of the interaction
between FUN19 and the genes listed in Table 2.
BENNO FIGURE 1:
FUN19 was not a part of the interaction map created by Fields, Uetz,
and Schwikowski, but ADH1, KAP123, and SIN3 were all included. Each of
these genes was in a light yellow box signifying that role of the gene
was unknown.
ExPASy 2-D:
This is the same 2-D gel that was seen in the FUN19 section only on
this gel the red square is around where FUN19p is predicted to be. The
coordinates seem to match up with the information from the MIPS database
(pI = 9.88 and MW = 47068.2 Da). It is too bad that this was the only 2-D
gel that the database had of the S. cerevisiae proteome.

This gel does not do very much as far as providing data. Because only
part of the square is shown and for the part that is shown there is nothing
it gives the impression that the protein produced by FUN19 is not there.
Although this protein is not essential I would still expect some evidence
that it is present in yeast cells.
YEAST TWO-HYBRID ANALYSIS:
FUN19 does not appear as a bait or prey protein on either the Y2H Results
page or the Additional Y2H Results page.
PROWL:
This database only provided basic information about FUN19, which appears below.
PDF Files:
FUN19 did not appear in any of the three pdf files.
Right now I feel like a magician who is attempting to pull an white
rabbit out of a top hat only I do not know have any optical illusions arranged
to discuss a slip of my hand. Since there is very little information available
its hard to develop a hypothesis that I would put a lot of faith in. Based
on the molecular functions and cellular components of the proteins with
which FUN19p interacts I would venture to guess that this gene serves some
sort of back-up function in transcription. Based on the mixture of cellular
components within the nucleus and in the cytoplasm as well as the many
different complexes listed it would make sense that this gene interact
with some proteins in the nucleus and others as they left with mRNA bound
for translation in the cytoplasm. The reason I believe that FUN19p serves
a back-up function is because it is not essential although I believe it
is involved in a process that is very essential therefore some sort of
safety mechanism must be present to pick up the slack if FUN19p is not
functioning as it should.
Future (and hopefully more productive) Experiments:
The first proposed experiment is in fact a combination of two experiments. Because this approach involves the successful accomplishment of two experiments it will be more difficult to perform than if only one experiment was performed. My idea for this experiment stems from the fact that the FUN19p is nonessential which leads you to believe that the function its serves is most likely served by another protein as well therefore if it were to be deleted the effects would not be lethal. Based on my hypothesis that FUN19p is involved in transcription if different genes involved in transcription could be knocked out in different cultures at the same time that the level of FUN19p is monitored hopefully one culture will show an increased amount of FUN19p. The transcription genes would ideally be knocked out throughout the life of the cell culture. The presence of FUN19p can be measured in a particular knockout culture in relation to a normal population by utilizing the stable isotope technique developed by Brian Chiat at Rockefeller University. Although this technique seems incredibly tedious if the technique proves successful then the function of FUN19p could be determined, a very big advancement on a non-annotated gene.
The second experiment that I am proposing is not as wishful as the previous one. This experiment focuses around the approach introduced by Mike Synder in his lab at Yale University. Synder developed a technique whereby he could insert a mTn (multipurpose minitransposon that contained a number of genetic alterations to improve its utility in genome wide studies) into the coding region of FUN19p. A segment of the mTn could then be excised and an epitope tag inserted. This tag could then be sought out using hemagglutinin monoclonal antibodies, which would provide new evidence as to the cellular component of the protein. The inserted mTn would also be able to show the researchers when FUN19p is being expressed due to the fact that the mTn segment turns blue when it is located downstream of a promoter. Therefore whenever the marker is illuminated then the yeast cell is attempting to produce FUN19.
A third possibility, though it is likely that it has already been attempted, is to perform the yeast two-hybrid experiment (Y2H) with FUN19p as either the bait or prey and each of the proteins from the DIP interaction map as the corresponding protein (i.e. if FUN19p is the bait then have KAP123p as the prey or vice versa).
All the information regarding where these techniques were developed,
how they were developed by, and the techniques employed in each technique
was obtained from Discovering Genomics, Proteomics, & Bioinformatics.
References
Additional Y2H Results. 2004. <http://depts.washington.edu/~yeastrc/th_12.htm>. Accessed 2004 Nov. 18.
Campbell, A. Malcolm and L. J. Heyer. 2004. Discovering Genomics, Proteomics, and Bioinformatics. Benjamin Cummings: San Francisco.
[DIP] Database of Interacting Proteins. 2004. <http://dip.doe-mbi.ucla.edu/dip/Search.cgi?SM=3>. Accessed 2004 Nov. 18.
MIPS Comprehensive Yeast Genome Database. 2004. <http://mips.gsf.de/genre/proj/yeast/index.jsp>. Accessed 2004 Nov. 18.
PathCalling Yeast Interaction Database. 2004. <http://portal.curagen.com/extpc/com.curagen.portal.servlet.PortalYeastList?modeIn=List>. Accessed 2004 Nov. 18.
PROWL. 2004. <http://129.85.19.192/prowl/proteininfo.html>. Accessed 2004 Nov. 18.
[SGD] Saccharomyces Genome Database. 2004. <http://www.yeastgenome.org/>. Accessed 2004 Nov. 18.
Schwikowski, Benno, Uetz P., Fields S., A Network of Protein-Protein Interactions in Yeast. Nature Biotechnology. 18: 1257-1261.
Y2H Results. 2004. <http://depts.washington.edu/%7Eyeastrc/th_11.htm>.
Accessed 2004 Nov. 18.