A Gene for Deafness?
This web page was produced as an assignment for an undergraduate course at Davidson College.
|
|||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
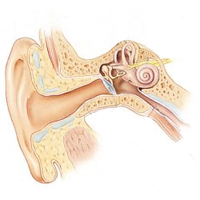
| Hearing is a method of acquiring information from our surroundings by interpreting air waves created by sound. Our normal hearing ability is astounding and something that most take for granted. We can hear frequencies ranging from 20 – 20,000 HZ (Chandler 2006). The ear is a complex design that has 3 different parts. First, the outer ear is responsible for “catching” sound waves and amplifying them into ear canal to the middle ear. The pinna (ear lobe) also is essential for determining where a sound originated (Harris 2006).
Next, the sound waves travel and bounce into the tympanic membrane causing it to vibrate. Two muscles control the function of the membrane. The tensor tympani muscle maintains the tautness of the membrane, and the stapedius muscle increases the tautness of the eardrum in the presence of loud noises. A gradual weakening of this muscle with age may be one cause of gradual ear damage that results in hearing loss of the elderly. This mechanism also allows us to focus our attention on different sounds such as a voice in a crowd (Harris 2006). |
Permission pending from NASA |
||||||||||||||||||||||||||||||||||||
Permission Pending from HowStuffWorks.com |
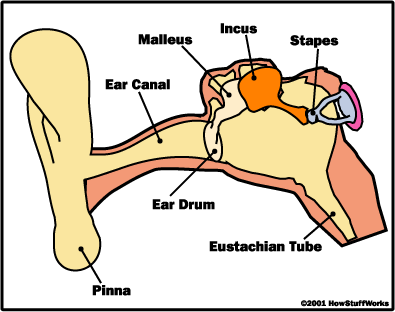
Vibrations of the tympanic membrane are transferred next to the bones of the inner ear called the ossicles. These consist of 3 different bones, the malleus (hammer), the incus (anvil), and the stapes (stirrup) bones. The malleus is connected to the ear drum and vibrates as the tympanic membrane vibrates. These vibrations are amplified by nearly 22 times as they travel along the malleus to the incus, and finally, to the stapes that is connected to the oval window (Harris 2006). The stapes causes vibrations in the fluid of the cochlea that surrounds the basilar membrane that is covered in long fibers know as the organ of corti. The hairs found on this “organ” are the basis behind mechanotransduction. Mechanotransduction is the conversion of a mechanical stimulus, sound waves, into an electric signal to the brain (Sotomayor 2006). |
||||||||||||||||||||||||||||||||||||
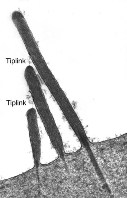
Hairs are organized by ascending height in a bundle called the stereocilia. Researchers have discovered that ion channels called tip-links exist at the ends of the stereocilia. The tip-link is a group of proteins that open and exhibit a slight voltage change when they are stimulated. Thus, we have finally discovered the mechanism that transfers this movement into an electrical stimulus to the cochlear nerve and the cerebral cortex (Sotomayor 2006). |
Tip linkage |
||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
Usher's Syndrome |
|||||||||||||||||||||||||||||||||||||
The disorder I will focus on is Usher’s Syndrome, which actually affects not only hearing but sight as well. Usher’s Syndrome accounts for 3-6% of all deaf children (NIDCD). Three types of Usher’s Syndrome exist that vary in severity from deafness at birth to normal hearing at birth with a slow decline in auditory abilities. I will focus on the most severe, Usher’s Syndrome Type 1 (USH1) (Boys Town National Research Hospital 2006). Usher’s Syndrome is an autosomal recessive disease discovered by Albrecht Von Graefe in 1858. Researchers currently estimate that 1 in 70 people are carriers of the mutant alleles (Goldberg 2005). Von Graefe noticed that frequently patients he treated for retinitis pigmentosa were also deaf, but not until 1914 did Charles Usher determine the disease pattern of this deaf-blindness association. Usher’s syndrome is characterized by deafness at birth, a lack of inner ear balance, and blindness progressing from infancy. Blindness is due to a condition known as retinitis pigmentosa that progresses from night blindness in early childhood to tunnel vision by age 16, and eventual blindness (Boys Town National Research Hospital 2006). |
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
Retinitis pigmentosa |
|||||||||||||||||||||||||||||||||||||
| Retinitis pigmentosa is characterized by the deterioration of rods and then the cones responsible for light detection in the retina. One hundred and fifty million rods and 7 million cones exist in the eyes that are responsible for night vision and color vision. As the disease progresses, night vision is lost first followed by blind spots, and finally tunnel vision during the day. The relationship between retinitis pigmentosa and deafness is the same necessary conversion of information obtained by these sensory organs into signals the brain can understand. Thus, researchers believe that the proteins involved may be similar (Boys Town National Research Hospital 2006). |
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
Table 1. Mutations responsible for Usher’s Syndrome Type 1 (NCBI 2006).
|
|||||||||||||||||||||||||||||||||||||
| Cadherin | |||||||||||||||||||||||||||||||||||||
Image courtesy of PDB. |
|||||||||||||||||||||||||||||||||||||
| I will focus on mutations of the cadherin-23 protein. Cadherins are a group of proteins located on the surface of cells that assist cell-adhesion. These proteins are reliant upon calcium ions for function, which also play a role in cell signaling similar to signals sent to the brain via the cochlear and optic nerves. | |||||||||||||||||||||||||||||||||||||
Gene ontology of Cadherin-23: |
|||||||||||||||||||||||||||||||||||||
Function:
|
Calcium ion binding Protein binding |
||||||||||||||||||||||||||||||||||||
Process: |
Calcium dependent cell-cell adhesion Cell adhesion Homophilic cell adhesion Response to stimulus Sensory perception of sound Visual perception |
||||||||||||||||||||||||||||||||||||
Component: |
Integral to membrane Membrane |
||||||||||||||||||||||||||||||||||||
| Cadherin-23 is a large, transmembrane glycoprotein that was identified within the cadherin superfamily because of 27 repeats showing homology within the cadherin ectodomain. Researchers first identified cadherin-23 as a component in tip-links in 2004 (Siemens et al. 2004). Since then, researchers discovered a relationship between mutations in cadherin-23 and a range of hearing loss and retinitis pigmentosa. Further research showed that normal stereocilia formation and function relied on a complex formed by myosin VIIa, harmonin b, and cadherin-23, and interactions between wild type cadherin-23 and protocadherin-15 assist with maintaining the structure of stereocilia throughout aging. Finally, researchers have determined that cadherin-23 mutations cause other deafness phenotypes (NCBI 2006). Missense mutations are associated with presbycusis, a correlation of hearing loss with aging, and nonsyndromic prelingual hearing loss (DFNB12), hearing loss without any other symptoms (McHugh and Friedman 2006). More than 30 different null mutations are known to cause Usher’s syndrome type 1 phenotypes (Genetics Home Reference).
|
|||||||||||||||||||||||||||||||||||||
| Most descriptions of cadherin-23’s role in cell signaling involve the mechanotransduction pathway because little is known about its function in the retina. Researchers know that it exists within photoreceptor cells in association with myosin VIIa and harmonin (Genetics Home Reference 2006) but have yet to determine its exact function. Scientists predict that it is essential for proper cell signaling to the optic nerve. |
 |
||||||||||||||||||||||||||||||||||||
The Scientific Article: Siemens, J., C. Lillo, R.A. Dumont, A. Reynolds, D.S. Williams, P.G. Gillespie, and U. Müller. 2004. Cadherin 23 is a component of the tip link in hair-cell stereocilia. Nature 428: 950 – 955. |
|||||||||||||||||||||||||||||||||||||
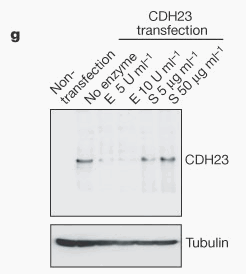
| Prior to this study, researchers knew that mutations within cadherin 23 caused Usher’s syndrome in some individuals, but no one knew how this mutation caused deafness. Researchers also understood the basic function of the extracellular filaments that interconnected the stereocilia, and only this linkage was in the correct location to control mechanotransduction. This small 150-200 nm stretch was previously unknown, but was similar with kinocilia, which was recognized by a tip link antigen. Other previous research that proved relevant to this study was the revelation that enzymes that prevented the prevalence of calcium ions also disrupted the tip link, which also prevents the function of the cadherin super-family. | 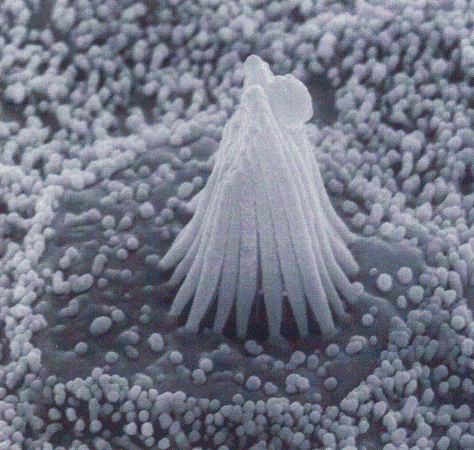 |
||||||||||||||||||||||||||||||||||||
Researchers began their search for the proteins responsible for the tip link by first determining that the features of the tip link were shared with the cadherin family. They next looked at size and determined that 27 repeats of the cadherin 23 domain, if interacting homophilically, could span the 200nm gap. Thus, their first step was to identify whether cadherin 23 was found in the inner ear of mice. |
|||||||||||||||||||||||||||||||||||||
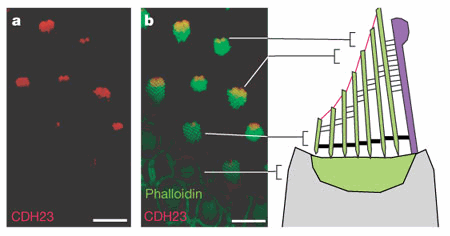
Cadherin 23 located at the tips of stereocilia. |
First, they used polymerase chain reaction (PCR) with reverse transcription of messenger RNA encoding cadherin 23 with and without exon 68, which are two alternately spliced isoforms. After they located these mRNA sequences in the inner ear, they used an antibody to recognize the cytoplasmic domain for each isoform. The antibody for the cadherin 23 including exon 68 was only found in the mature hair bundles at the stereociliary tips. The antibody for cadherin 23 without exon 68 located this protein in developing hair cells and Reissner’s membrane. They also tested reactivity of these antibodies to longer hair cells of bullfrogs and also found that cadherin 23 was located at the tips of the stereocilia. | ||||||||||||||||||||||||||||||||||||
Researcher’s next step was to test if cadherin 23 was a tip link protein by immunogold electron microscopy with cadherin 23 cytoplasmic domain antibody serum, which was located at the stereociliary tips of mice and bullfrog. They found more than half of the gold particles ended up at the tip link insertion point of a stereocilium, 20% were found at the end of the steriocilia, and another 20% were located either along the stereocilia at no particular point or were extracellular. The presence of multiple gold particles at each tip link led investigators to believe that multiple cadherin 23 proteins may be present in each tip link. |
|
Immunogold electron microscopy of mice stereocilia | |||||||||||||||||||||||||||||||||||
Next, researchers tested the effect of the elimination of calcium on tip link function. If tip links are reliant upon cadherin 23, they should cease to function if cadherin 23 ceases to function in the absence of calcium ions, which they showed by comparing these results to two controls. These controls were enzymes that temporarily removed calcium ions, tip links were not expressed, but reappeared when these enzymes were removed. In the absence of calcium ions with lanthanum, tip links never reappeared. |
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
Finally, researchers tested to see if myosin-1c, located at stereociliary tips, and cadherin 23 could interact directly or indirectly by using interleukin-2 for cadherin 23 to precipitate cadherin 23. Researchers found that myosin-1c precipitated with interleukin-2/cadherin23, showing that these two proteins can interact directly at the tip links. |
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||
| Popular Press Articles | |||||||||||||||||||||||||||||||||||||
| Popular press articles should address the relevant background information, the methods of the study, the results, and their importance to science. On this basis, I will examine 3 articles published about the discovery of the function of cadherin-23. | |||||||||||||||||||||||||||||||||||||
The San Diego Union-Tribune “Deafness gene’s function is found” Bruce Lieberman, April 19, 2006 This article discussed many relevant research such as the connection between this protein and Usher’s syndrome, even though the research article did not draw this conclusion. Additionally, the author simplified the mechanotransduction pathway to a simple, easy to understand explanation of hearing to place cadherin 23 into context. Lieberman expresses only the main result of the study, but none of the smaller sections that led to this conclusion. He also does not describe the methods of this study accurately. Describing the process by “scanning all known proteins in the human and mouse genomes to see which one fit the profile of proteins of that type that had already been associated with stereocilia” is misleading because this implies that they searched for similar sequences, not that they conducted tests on the actual proteins located on the stereocilia. The justification for the study’s conclusion was never accurately described nor accurately alluded to. This article also states that researchers had narrowed the field to two families of proteins, cadherins and integrins, which is not mentioned in the research article. While this article could be improved, Lieberman should be applauded for his effort to stress that while this study is important, we have much to learn about mechantransduction and the role of cadherin 23. |
|||||||||||||||||||||||||||||||||||||
The Scripps Research Institute “Scripps research scientists find deafness gene’s function” Jason Bardi – April 5, 2004 This article was a shorter article based on the press release following the publication of the scientific paper that was written by the institution responsible for the study. Thus, the quality of the article is much higher. The study accurately portrays the context of the study through describing tip links and hearing mechanisms. Additionally, the result of the study was simplified into a single sentence that was simple and easy to understand, "discovered a key molecule that is part of the machinery that mediates the sense of hearing". In collaboration with these results, the paper is multifaceted in presenting results from a parallel study examining cadherin 23 mutations in zebrafish. Bardi, later in the paper, describes more background information as he describes the methods used by researchers to provide evidence that cadherin 23 is a protein found at the tip links. He described several more specific tests reseachers conducted to support their conclusion, but omitted those tests that may have been difficult for lay-people to understand. Furthermore, he did not try to extrapolate this study as leading to a cure for deafness as some other articles may suggest. Rather, he alludes to future directions of research about tip links or cadherin 23. Overall, as expected, this article was much more consistent with the actual scientific article. |
|||||||||||||||||||||||||||||||||||||
| Overall impressions | |||||||||||||||||||||||||||||||||||||
| The study by Siemens et al. (2004) is very convincing and combines several lines of evidence to prove their hypothesis. Unfortunately, none of the popular articles made the evidence for the function of cadherin 23 clear. Genomics is a complicated field to communicate with the general public; thus, many details that make the investigations interesting are skipped for the sake of imparting the most important facts about a study. Furthermore, popular press articles are prone to making generalizations and overreaching the conclusions of the papers. During research for this website, I found many articles that alluded to a discovery of a cure for deafness by defining the function of cadherin 23. Popular press articles must be careful during reporting to not excede their knowledge or the discoveries they describe. Overall, popular press articles bring new research to the public and try to impart the importance of this research, and for that, we should encourage more articles about genomic research. | |||||||||||||||||||||||||||||||||||||
| Links | |||||||||||||||||||||||||||||||||||||
National Institute on Deafness and Other Communication Disorders American Association of Deaf-Blind |
|||||||||||||||||||||||||||||||||||||
| Citations | |||||||||||||||||||||||||||||||||||||
Bardi, J.S. 5 April 2004. "Scientists Find Deafness Gene's Function". http://www.scripps.edu/newsandviews/e_20040405/muller.html Accessed 11 September 2006. BBC News. 26 August 2006. "Gene for age-related hearing loss". http://news.bbc.co.uk/2/hi/health/5284916.stm Accessed 3 September 2006. Boeda, B., A. El-Amraoui, A. Bahloul, R. Goodyear, L. Daviet, S. Blanchard, I. Perfettini, K.R. Fath, S. Shorte, J. Reiners, A. Houdusse, P. Legrain, U. Wolfrum, G. Richardson, and C. Petit. 2002. Myosin VIIa, harmonin and cadherin 23, three Usher I gene products that cooperate to shape the sensory hair cell bundles. EMBO J. 21: 6689-6699. Boys Town National Research Hospital. 2006. Information About Usher Syndrome. http://www.boystownhospital.org/usher/information.asp Accessed 10 September 2006. Chandler, E. 9 September 2006. The Ear. http://faculty. washington.edu/chandler/bigear.html Accessed 11 September 2006. Genetics Home Reference. 8 September 2006. CDH 23. http://ghr.nlm.nih.gov/gene=cdh23 Accessed 12 September 2006. Goldberg, J. 2005. On the Trail of Deafness Genes. http://www.hhmi.org/senses/c140.html Accessed 3 September 2006. Harris, T. 2006. How Hearing Works. http://www.howstuffworks.com/hearing.htm Accessed 10 September 2006. Lieberman, B. 19 August 2004. "Deafness gene's function is found". http://www.signonsandiego.com/uniontrib/20040419/news_1m19deaf.html Accessed 12 September 2006. McHugh, R.K. and R.A. Friedman. 2006. Genetics of Hearing Loss: Allelism and modifier genes produce a phenotypic continuum. Anat. Rec. A. Discov. Mol. Cell Evol. Biol. 288(4). http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Pubmed&cmd=retrieve&list_uids=16550584 Accessed 4 September 2006. NCBI. 27 August 2006. CDH23. http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=gene&cmd=Retrieve&dopt=full_report&list_uids=64072 Accessed 4 September 2006.
|
|||||||||||||||||||||||||||||||||||||
Questions? Please contact Krcecala@davidson.edu
© Copyright 2006 Department of Biology, Davidson College, Davidson, NC 28035