This web page was produced as an assignment for an undergraduate course at Davidson College.
Back to Dylan's Genomics Home Page
Genomics Assignment 2: Paper Review
Evaluating 'Lactobacillus plantarum strain maintains growth of infant mice
during chronic undernutrition' (Schwarzer et al.)
Summary
In
their
2016 publication, Schwarzer et al.
demonstrate
that a particular strain of Lactobacillus
plantarum
promotes growth of juvenile mice during days 21-56 post birth.
Throughout the study, Schwarzer et
al. compare
longitudinal growth and systemic growth of wild type (WT) and germ free
(GF)
mice when exposed to breeding diets and nutritionally depleted diets after
weaning (21 days post birth).
Schwarzer
et al. performed an array of
experiments, first to demonstrate that microbiota have an impact on infant
growth and finally to pinpoint a particular Lactobacillus
plantarum
strain as a major factor in growth while subject to nutritionally
depleted diets. First, they found that presence of microbiota was a factor
in
growth by comparing weight and body length of WT and GF juveniles fed a
standard breeding diet. They saw that GF mice weighed less and were
shorter than
their WT counterparts, despite eating similar quantities of food. Because
systemic growth is driven by the somatotropic axis, they investigated
circulating levels of growth hormone (GH),
insulin growth factor-1 (IGF-1), and IGF-1 binding protein-3
(IGFBP-3)
and found lower levels of IGF-1 and IGFBP-3 in GF mice when compared to WT
mice. They tested the role of IGF-1 in growth by injecting WT mice with an
inhibitor of IGF-1R called picropodophyllin (PPP), which slowed growth of
mice.
Their results indicate that microbiota is involved in production of IGF-1,
which is necessary for growth. To test chronic nutrition’s influence on
juvenile growth, they compared growth of WT and GF mice raised on
nutritionally
depleted diets and saw that WT mice lost less weight than GF mice and were
able
to recover weight when fed a normal diet, but GF mice were not. To test
effects
of microbiota on the somatotropic axis, they measured levels of GH, IGF-1,
and
IGFBP-3 in mice and found that reduced activity of the somatotropic axis
in GF
mice. Finally, they incorporated two different strains of L. plantarum into their tests to identify effects of Lactobacillus
strain on growth recovery,
and found that mice monocolonized with LpWJL experienced
recovery of
large amounts of lost weight while mice monocolonized with LpNIZO2877
experienced nominal
recapitulating effects.
Altogether,
Shwarzer
et al. show that GF mice,
when exposed to undernutriton, experience stunted growth, and that
microbiota
native to WT mice can recover growth by making mice more sensitive to GH.
Specifically, they identified a strain of Lactobacillus
that accounts for many of
the growth
enhancing effects of the WT microbiota. Clinically, the authors predict
that
microbial interventions can counteract undernutrition in humans and
promote
growth.
My
Opinion
I
found this paper to be well written, with clearly stated objectives,
hypotheses, and results. Their methods were logical and easy to follow, as
each
experiment brought up a question that was then addressed in the following
experiment, and their figures were typically presented in an
understandable
way. I was compelled to believe their claims because they looked at
differing
growth responses in GF and WT mice as well as circulating growth hormone
and
IGF-1 levels and gene expression levels.
Most
of
my complaints with this paper are related to small aspects of their figure
presentation. First, I found their figures showing femur length (Fig. 1E
and
3B) to be uninformative. While there was perhaps a noticeable difference
in
femur length, I would have preferred that they show a quantitative
difference (bar
graph) with standard deviation. By picking one bone from each treatment,
they
may have selected a pair that have the greatest size difference, rather
than offering
a measure of variance across the two treatment groups.
I
would have liked for them to include a figure showing variation in organ
size,
because they mention organ growth in the introduction but do not mention
it
anywhere else in the paper. Additionally, they introduced Fig. 4 before
they
wrote about their investigation with two strains of L. bacillus and without any discussion of DSMO, which was confusing
at first.
Finally,
the
authors state that this research gives evidence for bacterial intervention
to be able to counteract the negative growth effects of chronic
undernutrition
in children. I’m curious, because lack of growth when undernourished may
be
adaptive and forcing the body to grow without ample nutrients could have
detrimental effects. Also, I’m skeptical of spending money to treat
children
with bacteria when money could also go to providing nutrition more
conventionally, which would address many of the other problems that come
with
undernutrition, besides poor growth.
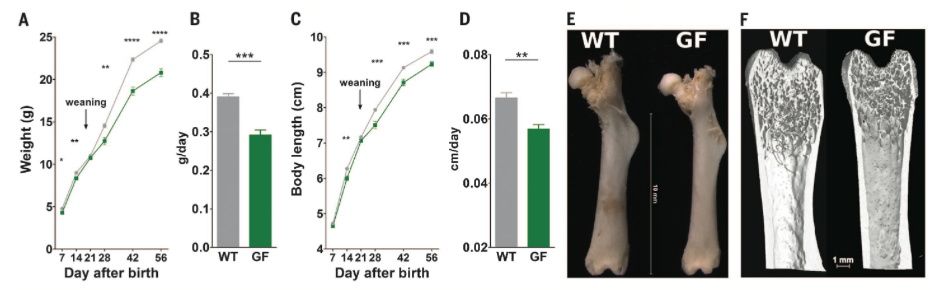
Figure
1*

Figure
1
compares growth of GF and WT mice in the first two months after birth.
Growth
is measured by weight, weight gain per day, body length, body
length gain per day, and femur
length. In each instance, GF
mice
exhibit less growth than WT mice. Thus, it appears that microbiota have an
influence on juvenile mouse growth.
Figure 2*
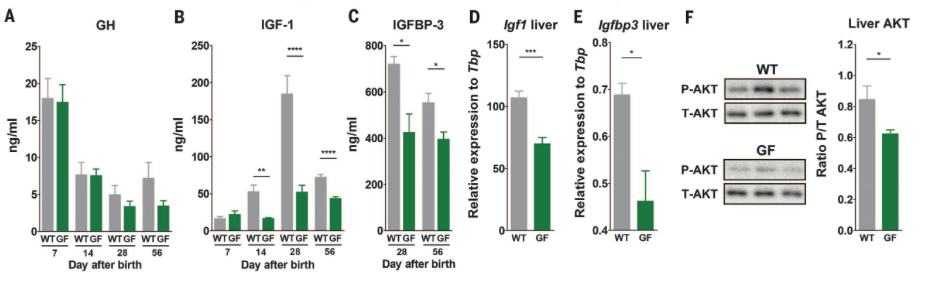
Figure
2
shows the effects of the somatotropic axis on juvenile mouse growth in GF
and
WT mice. Circulating levels of GH, IGF-1, and IGFBP-3 are lower in sera of
GF
mice than they are in WT mice across the board.. Additionally expression
levels
of Igf1 and Igfbp3 in the livers of GF mice are lower, and Akt phosphorylation
is lower. Overall, the data
in Figure 2
suggest that somatotropic axis activity is lower in GF mice and thus,
systemic
growth is limited.
Figure 3*
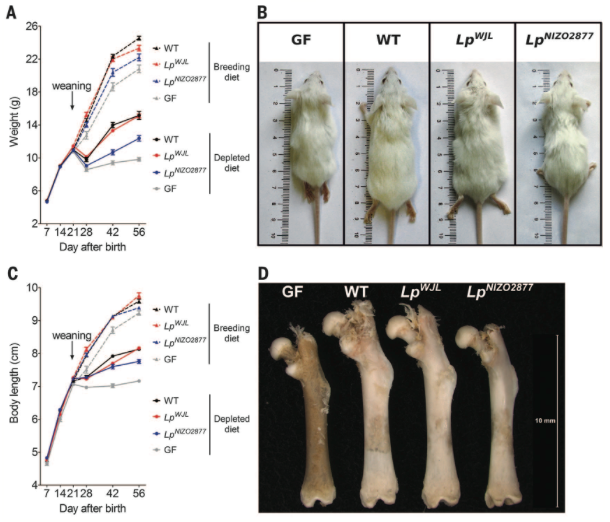
Figure
3
shows the growth, weight, and femur length of GF and WT mice, along with
mice
monocolonized with two different strains of L.
plantarum, when fed standard
breeding
diets or nutritionally depleted diets. Altogether, mice fed the standard
diet
weighed more and were longer than mice fed nutritionally depleted diets,
and WT
mice of a treatment exhibited more growth than their GF counterparts.
Additionally, LpWJL had
similar
growth to WT mice of the same diet, while LpNIZO2877
mice were more similar to GF mice of the same diet. This figure shows
that
mice monocolonized with particular bacterial strains can replicate the
growth
effects of WT micorbiota.
Figure 4*
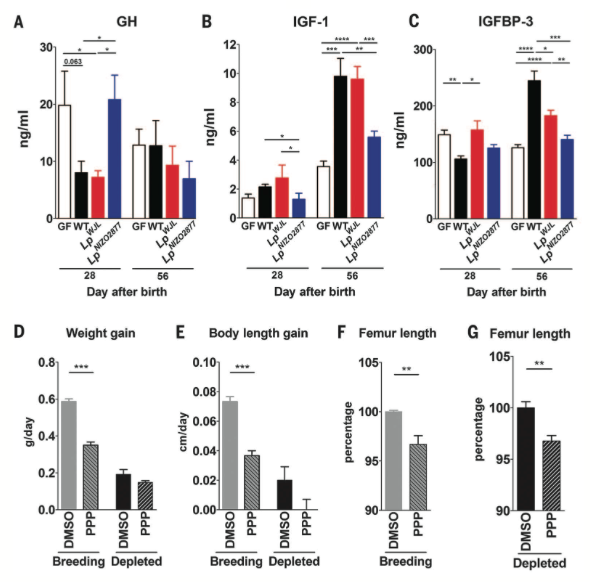
Figure 4 shows activity of the somatotropic axis in WT mice, GF mice, and mice monocolonized with either of the two L. plantarum strains when raised on nutrient depeleted diets. Like figure 2, figure 4 shows circulating levels of GH, IGF-1, and IGFBP-3 in mouse sera at different stages of growth (A-C). GH levels in WT and LpWJL was significantly decreased compared to GF and LpNIZO2877 at 28 days after birth. Levels of IGF-1 and IGFBP-3 at 56 days were increased in WJ and LpWJL mice and to a lesser extent, in LpNIZO2877 mice. This data is consistent with Figure 2 in suggesting that the somatotropic axis is less active in GF mice. Finally, WT mice injected with a PPP compound (IGF-1 inhibitor, and therefore inhibitor of the somatotropic axis) experienced less growth when compared to a DSMO control, and treated and untreated mice raised on depleted diets experienced less growth than mice raised on breeding diets. In summary, this figure shows that somatotropic activity is lower in GF mice and that microbiota are essential for growth.
*Permission pending from Shwarzer et al. for all figures
Schwarzer, M., Makki K., et al. 2016. Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition. Science 351(6275):854-857. Web.
Email Questions or Comments: dymaghini@davidson.edu
_________________________________________________________________________________________________________________
© Copyright 2016 Department of Biology, Davidson College, Davidson, NC 28035