Add 2.5 µl of the 10X loading dye to each PCR reaction tube and electrophorese the DNA on a 1.5% agarose gel using 0.5X TBE and 200 ng/ml ethidium bromide. We usually run these gels at 90-100 volts for 1 - 1.5 hours. The exact time and voltage will depend on the gel box configuration and appropriate conditions can be refined accordingly.
0.5X TBE
45 mM Tris-borate
1 mM EDTA use 0.5 M stock that is pH 8.0
While the gel is running, we will learn how to calculate the molecular weights of bands on a gel. Turn to the next page to begin this portion of the lab.

The log of a molecule's molecular weight is proportional to the distance that molecule has migrated. Therefore, the first step is to generate a standard curve using molecules of known size (the molecular weight markers).
When using semilog paper (see the next page), the molecular weights (in units of base pairs (bp) for DNA; kiloDaltons (kDa) for proteins) is plotted on the Y-axis and the distance the molecule migrated (in mm) is plotted on the X-axis. When generating a standard curve, you will obtain a straight line (use a best-fit line).
Once your standard curve is ready, measure the distance traveled by your molecule of interest. Find that distance on the X-axis, and go up until you intersect with your standard curve. Move over to the Y-axis and that will indicate the molecular weight of the molecule you are studying.
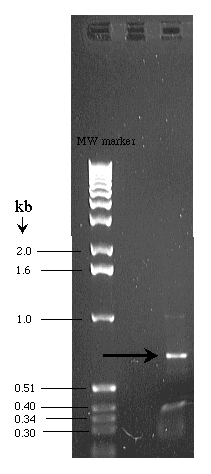
Use the first graph paper on the next page and the DNA gel shown to the right to determine the molecular weight of the unknown band indicated with an arrow.
The second graph is for you to use on your PCR DNA "fingerprint".

