| Jump to: | |
In this assignment I will analyze the protein products of the Saccharomyces cerevisiae genes MBP1 and YDL057W, which are annotated and non-annotated, respectively. Information about the genes MBP1 and YDL057W can be found here while information about the protein expression of MBP1 and YDL057W can be found here. Throughout this assignment, my own observations will be enclosed in purple-outlined boxes.
MBP1 is short for MluI-bow Binding Protein and it is a transcription factor composed of 833 amino acids. The transcription factor has a molecular weight of 93,907 Da and is involved in the regulation of the cell cycle as it progresses from the G1 phase to the S phase. It has an isoelectric point of 9.27 (MIPS).

Figure 1. Gene ontology information from NCBI Gene. Shows the molecular function, biological process, and cellular component of MBP1.
The structure of the protein can be visualized below (Figure 2).
A. 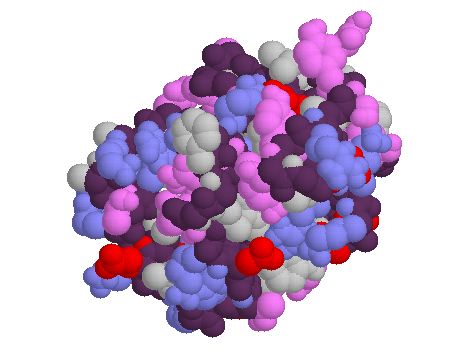
B. 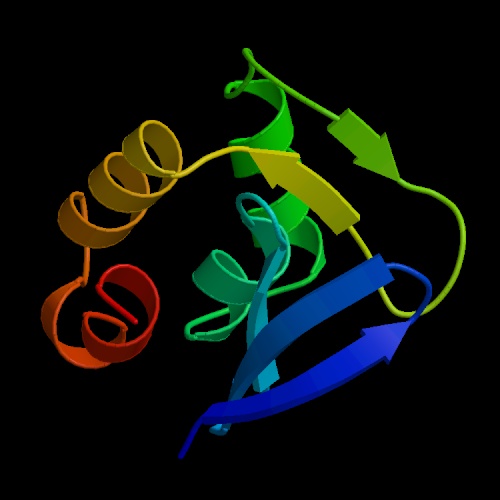
Figure 2. The structure of MBP1 in both "protein" (A.) and "cartoon" (B.) forms, from the Protein Data Bank.
Now let's look at how MBP1 interacts with other proteins. The Database of Interacting Proteins (DIP) provides us with a circuit diagram of MBP1's interactions:
A. 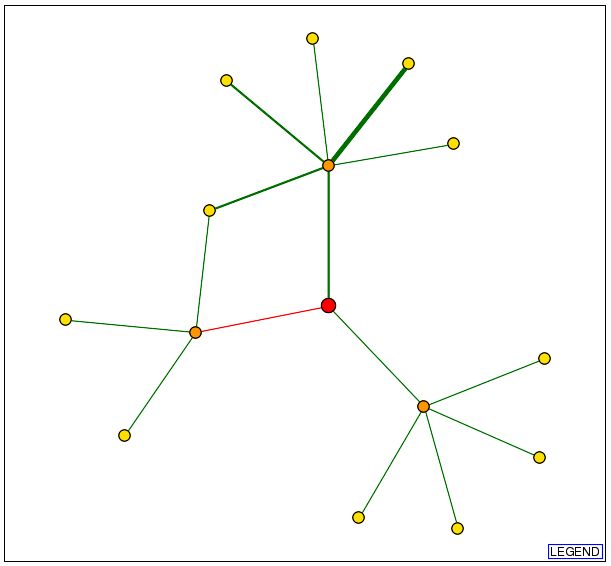
B. 
Figure 3. MBP1 protein interactions. A. A circuit diagram of MBP1's protein interactions. The two nodes connected to MBP1 are Swi6, with the thicker green line, and SKN7, with the thin green line. The red line shows the unverified results of high-throughput methods, linking MBP1 to FKH1. We will learn more about Swi6, SKN7, and FKH1 later. B. Legend for the circuit diagram. From DIP.
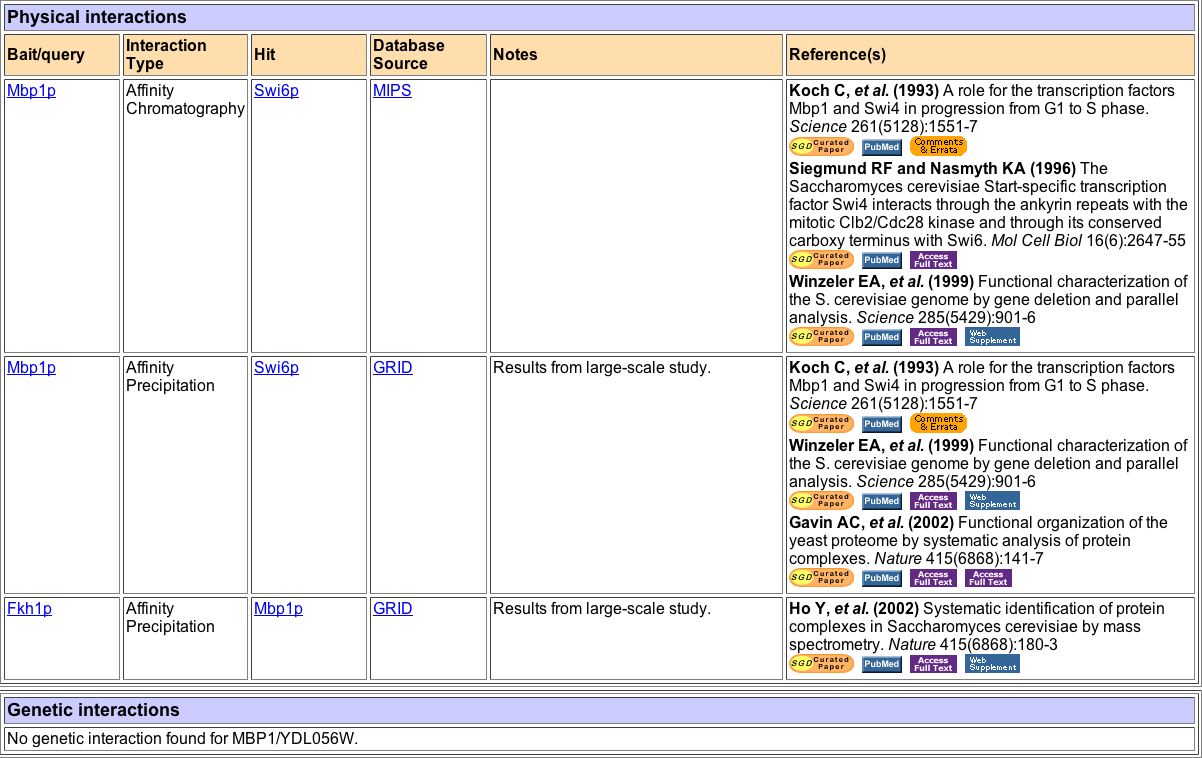
Figure 4. This chart, from the Saccharomyces Genome Database (SGD), shows all the known interactions of MBP1. MBP1 was used as the bait in Affinity Chromatography and Affinity Precipitation experiments 6 separate times (Koch et al. 1993, Siegmund and Nasmyth 1996, Winzeler et al. 1999, and Gavin et al. 2002). In all of these experiments, MBP1 attracted the protein Swi6p. In another study using Affinity Precipitation, a protein called FKH1P was used as the bait and attracted protein MBP1 (Ho et al. 2002). It is unclear why SKN7's interactions with MBP1, as found by DIP are not included in this table. There are no known genetic reactions.
Here is the basic information for the three proteins found to have physical interactions with MBP1. As mentioned in previous assignments, MBP1 and one of the genes it is found to interact with, Swi6, code for the proteins that make up what is called the "MCB-binding factor (MBF)." The MBF then "binds to [...] so-called MCB (MluI cell-cycle box) [...] and activates the transcription of [genes] at the G1--> S phase transition" (Xu et al. 1997).

Figure 5. Basic information for the protein Swi6, which forms a complex with MBP1 to regulate the G1 --> S progression in the cell cycle. From SGD.
FKH1 codes for the forkhead protein, which, like MBP1, is involved in the regulation of the cell cycle (Figure 6) (Hollenhorst et al. 2000, Zhu et al. 2000.

Figure 6. Basic information for FKH1, which encodes forkhead protein. FKH1 physically interacts with MBP1 and is shown here to be involved in regulation of the cell cycle, as is MBP1. From SGD.
SKN7 is a transcription factor, like MBP1. It is involved in osmoregulation.

Figure 7. Basic information for Skn7p, From SGD.
According to the GO Term Finder, Swi6 is mapped to some of the same molecular functions, biological processes, and the same cellular component as MBP1 (Figure 6), as you would expect from their interactions. In addition, FKH1 is mapped to some of the same molecular functions, biological processes, and the same cellular component as MBP1 (Figure 6), indicating that it probably closely interacts with MBP1 as well.
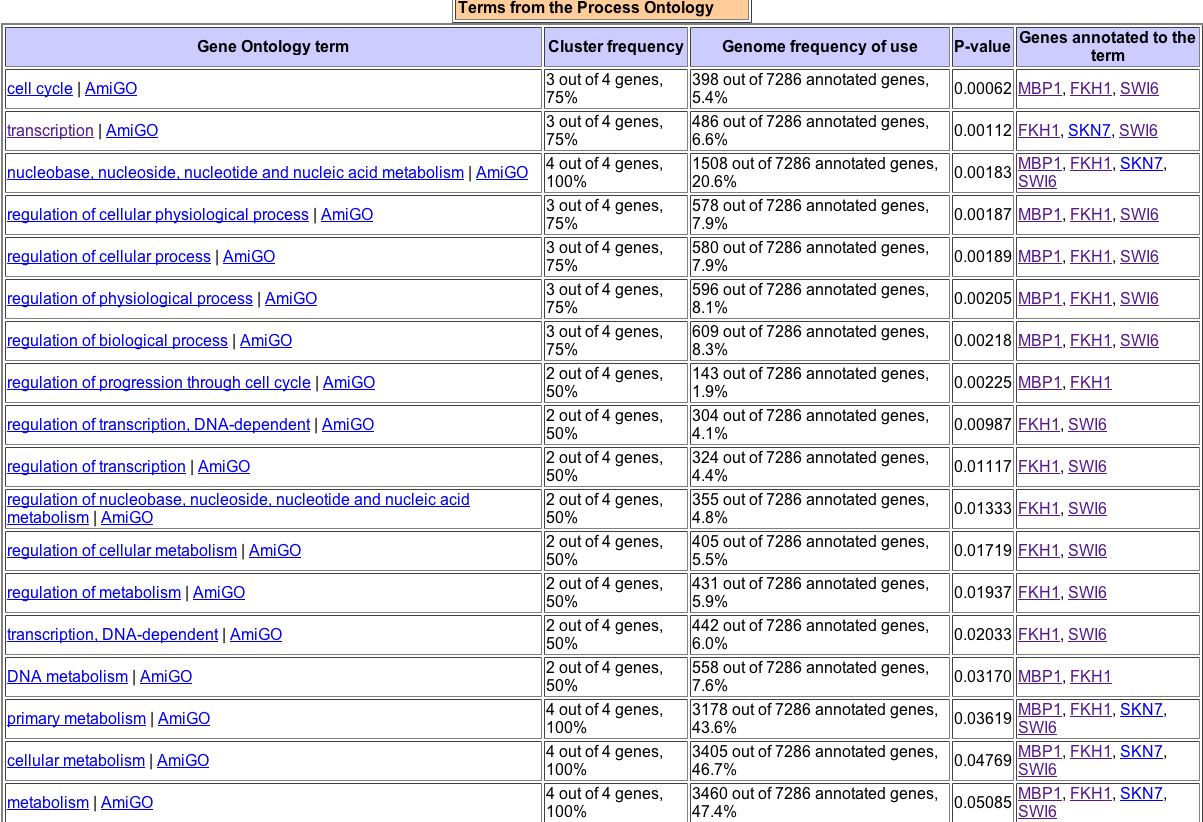
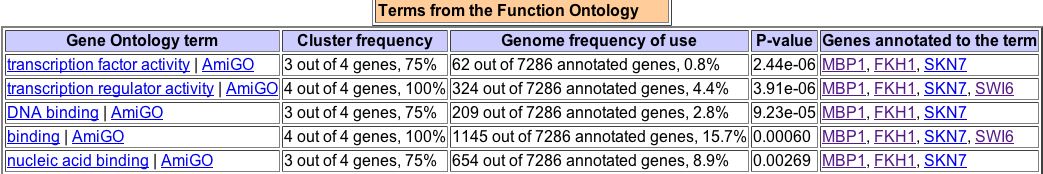
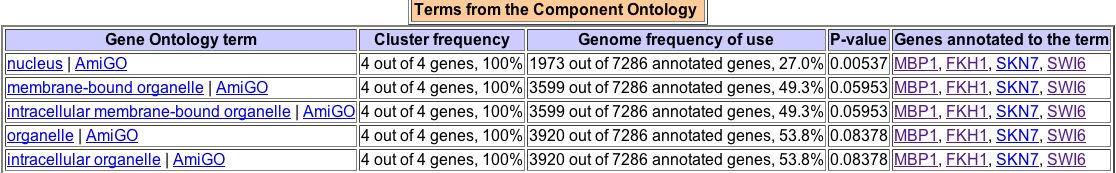
Figure 8. A search of GO Term Finder for the two proteins that physically interact with MBP1, along with MBP1 itself, yields this data.
|
As seen in Figure 8, MBP1 is often similar in biological process to both FHK1 and Swi6. Only in five GO Terms is it similar to SKN7 in biological process -- transcription, nucleic acid metabolism, primary metabolism, cellular metabolism, and metabolism. This suggests that SKN7 has more to do with metabolism than with the cell cycle itself. This is also reflected in SKN7's description above, which says it is involved in osmoregulation. MBP1 is most often similar to FHK1 and SKN7 in terms of molecular function. All four molecules, MBP1, Swi6, FHK1, and SKN7 share cellular components. |
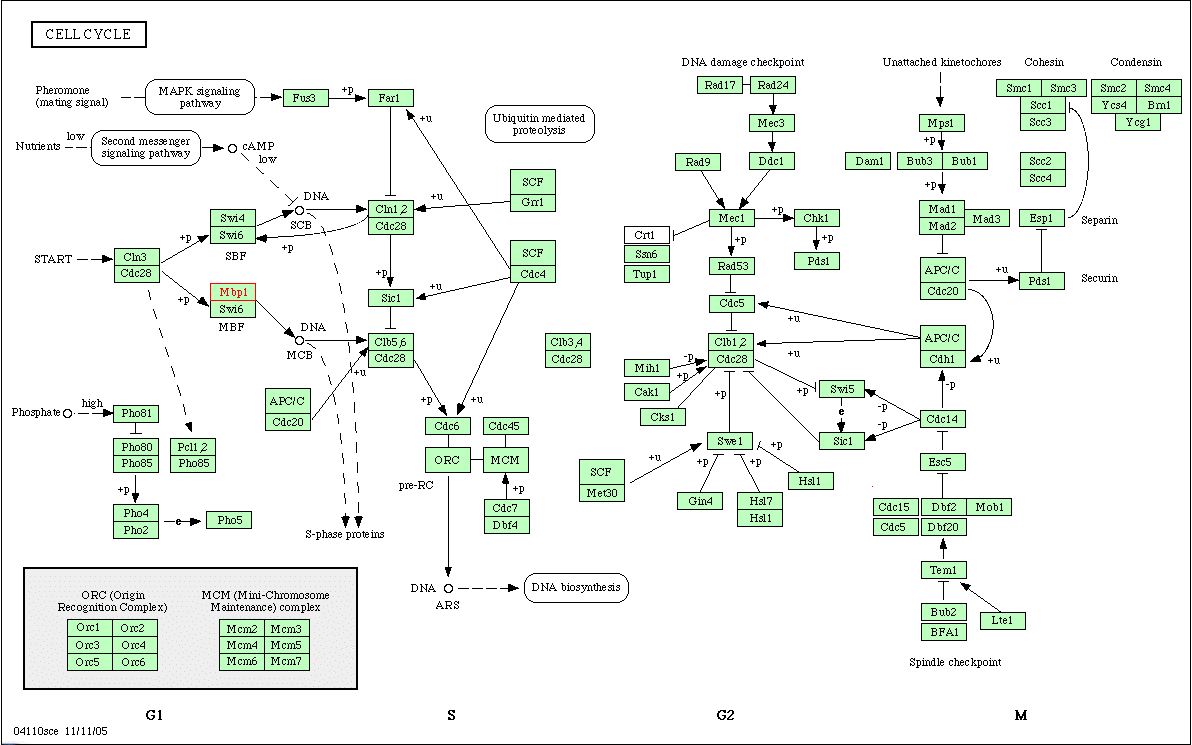
Figure 9. The Kyoto Encyclopedia of Genes and Genomes (KEGG) provides a circuit diagram of the cell cycle in S. cerevisiae. MBP1 is highlighted in red. In this diagram, its interaction with Swi6 to form the MBF is shown. The phase of the cell cycle is noted at the bottom of the diagram, and as aforementioned, MBP1 acts to transition the cell from the G1 phase to the S phase. While I searched for circuit diagrams that included FKH1 or Skn7p, the other proteins that physically interact with MBP1, I was unable to find any.
MBP1 appears in the document 'Degradation.pdf' that depicts a circuit diagram of proteins involved in degradation (Figure 9). It is colored yellow, indicating that it is involved in 'RNA-processing/modification."
A. 
B. 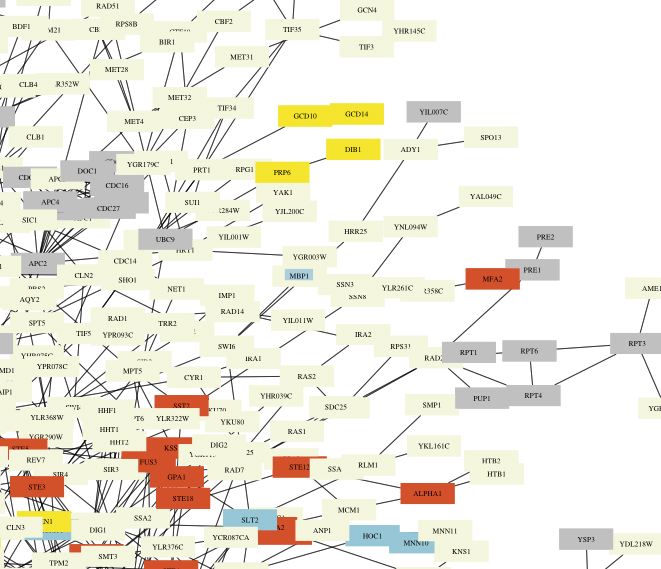
Figure 10. One section of the degradation circuit diagram. A. Legend explaining the colors of the boxes. B. MBP1 appears in a light yellow (almost white in this image) box with the text highlighted in blue in the center of the image. From Degradation.pdf.
I also searched the TRIPLES (TRansposon Insertion Phenotypes, Localization and Expression in Saccharomyces) database for information on MBP1.

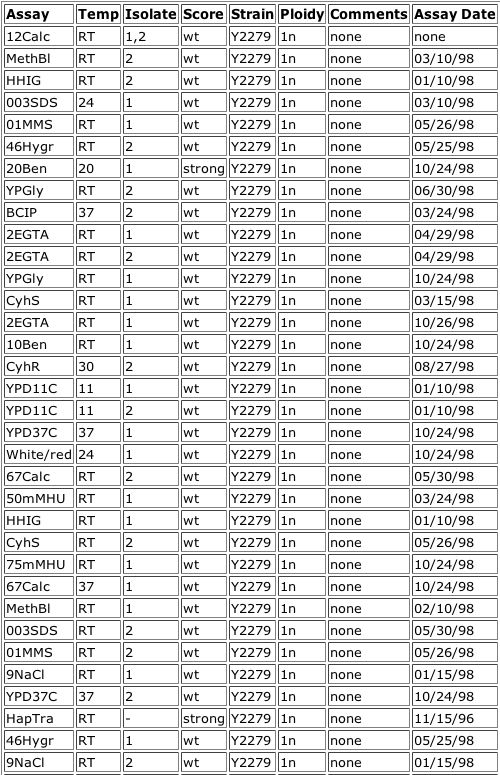
Figure 11. TRIPLES results for MBP1. As you can see, when a transposon interrupted the MBP1 gene, rendering it inactive, only two treatments showed growth different from that of the wild-type: 20Ben and HapTra. 20Ben is "Benomyl resistance as assayed by growth on YPD + 20mg/ml benomyl" and HapTra is "Cell inviability of haploid transformants." The list of assays has been abbreviated for space, but all assays that were omitted showed no significant change from the wild-type.
|
Since MBP1 is involved in the regulation of the cell cycle from the G1 phase and S phase, it is involved in DNA replication. This explains why effectively "knocking it out" with a transposon would cause a major difference in the viability of haploid transformants -- they would most likely be unable to effectively regulate DNA replication. |
I also searched the PROWL database for information on MBP1, but it did not provide any new insights.
Finally, I searched Stan Fields' lab's yeast two-hybrid (Y2H -- method described below) results, but found no information on MBP1.
|
More experiments can be done to further elucidate the role of MBP1 in the S. cerevisiae genome. Low-throughput methods have already been used to find some of the protein-protein interactions in which MBP1 is involved, but the high-throughput (albeit less reliable) yeast two-hybrid (Y2H) method can be used to find other interactions that may not yet be identified. To accomplish this, a transcription factor is divided into two parts: the DNA Binding Domain (DBD) and the Activation Domain (AD), neither of which can cause transcription by themselves. MBP1 would be the "bait" protein and would be bound to the DBD. Any known or unknown ORFs can be used as the prey by binding them to the AD. If the prey proteins physically interact with MBP1, the DBD and AD will be connected, causing transcription of the reporter gene His3. In this way, you can find protein-protein interactions in a high-throughput manner.
In addition, it would be nice to know how much MBP1 is present in the cell at various stages in the cell cycle. To do this, you could use isotope-coded affinity tags (ICAT). You would take proteins from two populations of yeast: one population in the G1 or S phase of the cell cycle and one population in the G2 phase of the cell cycle. Label one population with a heavy ICAT reagent and one with a light ICAT reagent. You would then isolate the proteins and mix them together, digest them with trypsin to simplify the experiment, and affinity purify them. Finally, the proteins would be analyzed using a mass spectrophotometer and you would get data that showed the relative abundance of MBP1 in the two populations. |
YDL057W is a non-annotated gene located right beside MBP1 on chromosome IV of the S. cerevisiae genome. It encodes a hypothetical protein, and hereafter "YDL057W" will refer to this hypothetical protein rather than the gene itself.
According to MIPS, the produced protein is 328 amino acids long with an isoelectric point of 7.14 and a molecular weight of 36,687 Da. The biological process, molecular function, and cellular component of YDL057W are currently unknown.
YDL057W yielded no results in the Protein Data Bank, which is to be expected, since it is not an annotated gene and therefore its protein product is unknown.
An SGD interactions search for YDL057W yielded no known physical or genetic interactions, indicating that research has not begun in earnest on this hypothetical protein. In addition, while DIP has YDL057W in its database, there is no graphic representation of any interactions.
I also searched the TRIPLES (TRansposon Insertion Phenotypes, Localization and Expression in Saccharomyces) database for information on YDL057W.

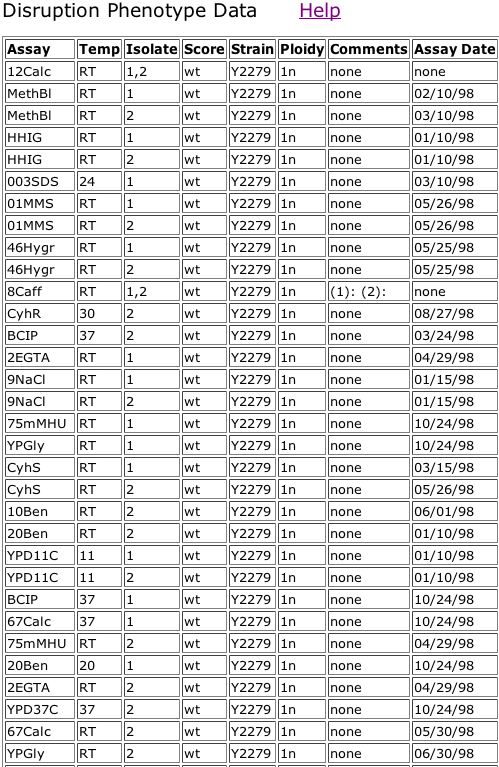
Figure 12. TRIPLES results for YDL057W. As you can see, when a transposon interrupted the YDL057W gene, rendering it inactive, none of the treatments showed growth different from that of the wild-type. The list of assays has been abbreviated for space, but all assays that were omitted showed no significant change from the wild-type.
I also searched the PROWL database for information on YDL057W, but it did not provide any new insights.
Finally, I searched Stan Fields' lab's Y2H results, but found no information on YDL057W.
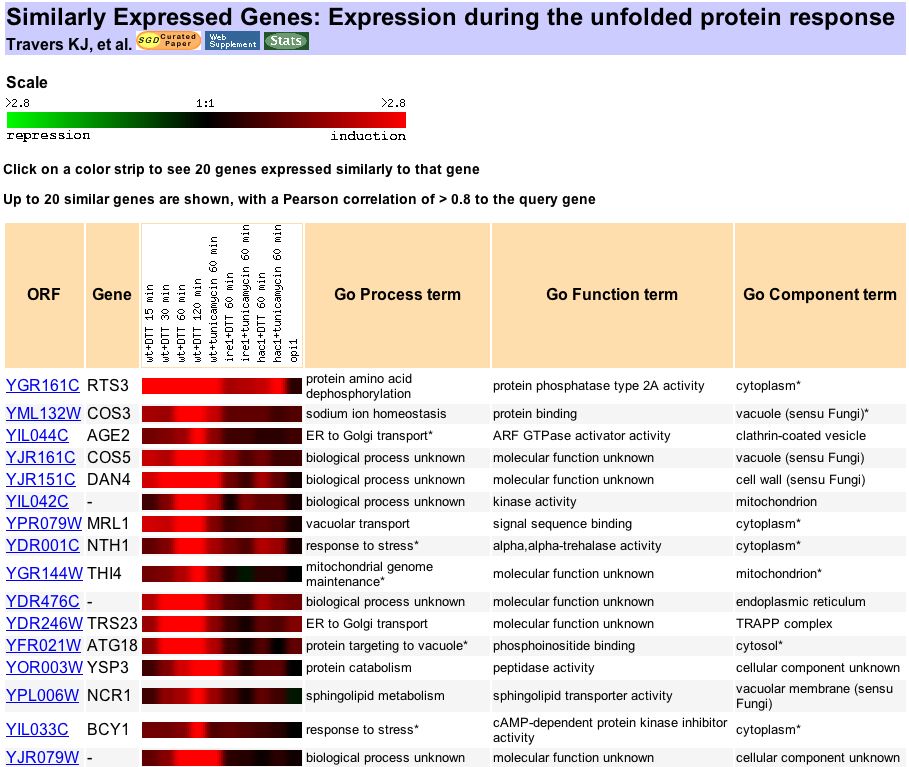
| YDL057W needs to be involved in many more experiments to elucidate its role in the S. cerevisiae genome. Here are some starting points: First, YDL057W needs to be localized. In order to localize a protein, you can use immunofluoresence. To do this, you obtain an antibody to the YDL057W protein. A secondary antibody (or an antibody to the first antibody) is also obtained and labeled with a fluorescent molecule. The yeast is then incubated with first the primary and then the secondary antibody, to produce fluorescence where the protein is located. Based on my prediction in the previous assignment that YDL057W is involved in the Unfolded Protein Response (UPR), I would expect it to be localized in the endoplasmic reticulum. According to Travers et al. (2000), "unfolded protein response (UPR) regulates gene expression in response to stress in the endoplasmic reticulum (ER)." Second, the high-throughput Y2H method should be performed as described above, this time using YDL057W as the bait protein. In this way, you could find any protein-protein interactions in which YDL057W is involved. I would expect YDL057W to interact with other proteins involved in the UPR response. According to Expression Connection, at least 16 proteins are expressed similarly to YDL057W during UPR.
Figure 13. Proteins that exhibit expression similar to YDL057W during UPR. From Expression Connection. Although YDL057W is the query gene, it is not shown at the top of the table for unknown reasons. Please check the URL for confirmation that YDL057W is, in fact, the query gene. In addition, YDL057W can be quantified under different conditions, which, by showing us the "timing" of the protein expression, may give us some clues as to its function. Since my hypothesis is that YDL057W is involved in UPR, two test populations should be grown: one undergoing stress in the ER and one in normal conditions. Label one population with the heavy ICAT reagent and the other with the light ICAT reagent and then complete the ICAT method as described above. The resulting data will show the relative abundance of YDL057W in the two populations. My hypothesis is that YDL057W will be many more times abundant in the first population, that is, the one undergoing stress in the ER. |
DIP. Database of Interacting Proteins. <http://dip.doe-mbi.ucla.edu/dip>.
Expression Connection. <http://db.yeastgenome.org/cgi-bin/expression/expressionConnection.pl>.
KEGG. Kyoto Encyclopedia of Genes and Genomes. <http://www.genome.jp>.
MIPS. Munich Information Center for Protein Sequences. <http://mips.gsf.de/genre/proj/yeast/search_index.jsp>.
NCBI Gene. <http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=gene>.
PDB. Protein Data Bank. <http://www.rcsb.org/pdb>.
PROWL. <http://129.85.19.192/prowl/proteininfo.html>.
SGD. Saccharomyces Genome Database. <http://www.yeastgenome.org>.
SGD Gene Ontology Term Finder. <http://db.yeastgenome.org/cgi-bin/GO/goTermFinder>.
TRIPLES. TRansposon Insertion Phenotypes, Localization and Expression in Saccharomyces. <http://ygac.med.yale.edu/triples/triples.htm>.
Campbell AM, Heyer LJ. (2003) Discovering Genomics, Proteomics, and Bioinformatics. Benjamin Cummings: San Francisco. 161-204.
Gavin AC, Bosche M, Krause R, Grandi P, Marzioch M, Bauer A, Schultz J, Rick JM, Michon AM, Cruciat CM, Remor M, Hofert C, Schelder M, Brajenovic M, Ruffner H, Merino A, Klein K, Hudak M, Dickson D, Rudi T, Gnau V, Bauch A, Bastuck S, Huhse B, Leutwein C, Heurtier MA, Copley RR, Edelmann A, Querfurth E, Rybin V, Drewes G, Raida M, Bouwmeester T, Bork P, Seraphin B, Kuster B, Neubauer G, Superti-Furga G. (2002) Functional organization of the yeast proteome by systematic analysis of protein complexes. Nature 415(6868): 141-7. (LINK).
Ho Y, Gruhler A, Heilbut A, Bader GD, Moore L, Adams SL, Millar A, Taylor P, Bennett K, Boutilier K, Yang L, Wolting C, Donaldson I, Schandorff S, Shewnarane J, Vo M, Taggart J, Goudreault M, Muskat B, Alfarano C, Dewar D, Lin Z, Michalickova K, Willems AR, Sassi H, Nielsen PA, Rasmussen KJ, Andersen JR, Johansen LE, Hansen LH, Jespersen H, Podtelejnikov A, Nielsen E, Crawford J, Poulsen V, Sorensen BD, Matthiesen J, Hendrickson RC, Gleeson F, Pawson T, Moran MF, Durocher D, Mann M, Hogue CW, Figeys D, Tyers M. (2002) Systematic identification of protein complexes in Saccharomyces cerevisiae by mass spectrometry. Nature 415(6868): 180-3. (LINK).
Koch C, Moll T, Neuberg M, Ahorn H, Nasmyth K. (1993) A role for the transcription factors Mbp1 and Swi4 in progression from G1 to S phase. Science 261(5128): 1551-7. (LINK).
Siegmund RF, Nasmyth KA. (1996) The Saccharomyces cerevisiae Start-specific transcription factor Swi4 interacts through the ankyrin repeats with the mitotic Clb2/Cdc28 kinase and through its conserved carboxy terminus with Swi6. Mol. Cell Biol. 16(6): 2647-55. (LINK).
Travers KJ, Patil CK, Wodicka L, Lockhart DJ, Weissman JS, Walter P (2000) Functional and genomic analyses reveal an essential coordination between the unfolded protein response and ER-associated degradation. Cell 101(3): 249-58. (LINK).
Winzeler EA, Shoemaker DD, Astromoff A, Liang H, Anderson K, Andre B, Bangham R, Benito R, Boeke JD, Bussey H, Chu AM, Connelly C, Davis K, Dietrich F, Dow SW, El Bakkoury M, Foury F, Friend SH, Gentalen E, Giaever G, Hegemann JH, Jones T, Laub M, Liao H, Liebundguth N, Lockhart DJ, Lucau-Danila A, Lussier M, M'Rabet N, Menard P, Mittmann M, Pai C, Rebischung C, Revuelta JL, Riles L, Roberts CJ, Ross-MacDonald P, Scherens B, Snyder M, Sookhai-Mahadeo S, Storms RK, Veronneau S, Voet M, Volckaert G, Ward TR, Wysocki R, Yen GS, Yu K, Zimmermann K, Philippsen P, Johnston M, Davis RW. (1999) Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285(5429): 901-6. (LINK).
Xu RM, Koch C, Liu Y, Horton JR, Knapp D, Nasmyth K, Cheng X. (1997) Crystal structure of the DNA-binding domain of Mbp1, a transcription factor important in cell-cycle control of DNA synthesis. Structure 5(3): 349-58. (LINK).