*This website was produced as an assignment for an undergraduate course at Davidson College*
Mantle cell lymphoma (MCL) is a subtype of non-Hodgkin lymphoma which arises from the uncontrolled proliferation of a subset of naive pregerminal center cells located in the mantle region of secondary follicles (Abbasi and Sparano, 2004). MCL accounts for between four and ten percent of the non-Hodgkin's lymphoma cases diagnosed each year (Bertoni et al., 2004). MCL usually pursues a relatively aggressive course, is resistant to long term remission, and is associated with a poor prognosis (Bertoni et al., 2004). Because of its quick metastasis and unresponsiveness to treatment, MCL poses a major challenge to clinicians and researchers. While there is still no definitive cure for MCL, chemothepary and stem cell/bone marrow transplants combined with long term drug therapy has shown promising results (Drach et. al, 2005 and Witzig, 2005).
Common Chromosomal Translocation and cylin D1 Overexpression
Approximately 70 percent of MCL cases are associated with a chromosome translocation at t(11;14)(q13;Q32) (see Figure 1)(Freedman and Harris, 2005). This translocation involves the cyclin D1 locus (which includes bcl-1) on chromosome 11 and the immunoglobulin heavy chain (IgH) locus on chromosome 14 (Freedman and Harris, 2005). Juxtaposition of the cyclin D1 gene to the potent B-cell IgH transcription enhancers leads to its overexpression (Freedman and Harris, 2005). Cyclin D1 is a protein synthesized in the late G1 phase of the cell cycle that helps induce progression into the S phase through the activation of cyclin-dependent kinases (Yatabe et al., 2000). Withdrawal from the cell cycle also seems to be dependent on suppression of cyclin-D1 (Yatabe et al., 2000). Overexpression of cyclin-D1 due to the previously mentioned chromosomal translocation is believed to be the major cause of the uncontrolled proliferation of MCL (Yatabe et al., 2000). In the 30 percent of cases of MCL that do not show a documentable rearrangement at t(11;14)(q13;q32), cyclin-D1 is still usually overexpressed; however, the mechanism for the cyclin-D1 overexpression in these case remains uncertain (Swerdlow et. al 1995). A small percentage of MCL cases are without overexpression of cyclin-D1, and this absence of cyclin d1 overexpression has been associated with favorable prognosis (Yatabe et al., 2000).
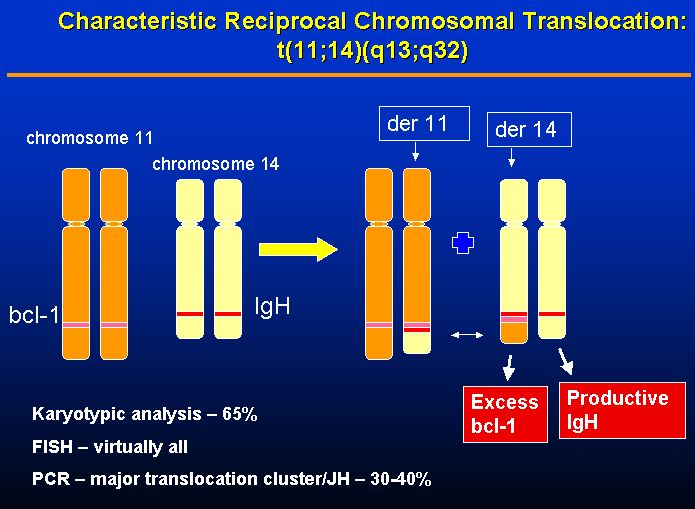
| Figure 1. The chromosomal translocation characteristic of 70 percent of MCL cases occurs between chromosomes 11 and 14 resulting in the overexpression of cyclinD1. Figure taken from Foley (2004), permission pending. |
Phenotype of MCL
MCL is characterized by expansion of the mantle zone of the lymph node with a homologous population of "atypical small to medium-sized lymphoid B cells with scant cytoplasm, slightly to markedly irregular indented nuclei, moderately dispersed chromatin, and inconspicuous nuclei" (see Figure 2) (Bertoni et al., 2004). Mantel cell lymphoma cells usually carry the following phenotype: CD19+, CD20+, CD22+, CD23-, CD24+, CD79a+, CD43+, CD5+, and CD10- (Bertoni et al., 2004). Because the cell surface molecule expression of MCL is variable and can overlap with other cancers like chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma, their use in diagnosis of MCL is limited; however, they are becoming of increasing value to development of new treatments for MCL (Witzig, 2005, Drach, 2005).
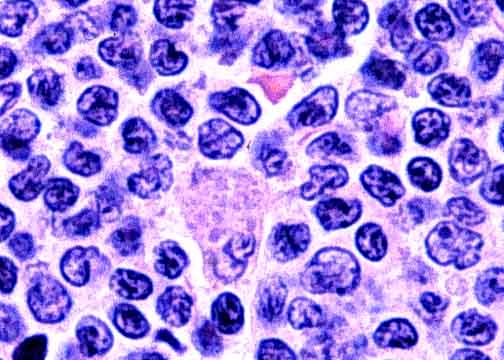
| Figure 2. Stain showing malignant mantle cell lymphoma cells. The figure illustrates the scant cytoplasm and irregular shaped nuclei characteristic of mantle cell lymphoma. Image taken from the University of Medicine and Dentisty of New Jersey, permission pending. |
Subtypes of MCL
Four different histologic patterns of MCL can be observed under the microscope (Friedman and Harris, 2005). MCL can present itself as mantle zone, nodular, diffuse, or blastic (see Figure 3)(The Leukemia and Lymphoma Society). Recent studies suggest transformation into blastic patterened MCL is associated with more aggressive disease and a worse prognosis (Bertoni, 2004).
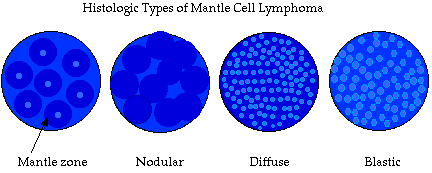
| Figure 3. Different histological patterns of mantle cell lymphoma. Transformation into the blastic variant indicates elevated disease activity and poor prognosis (Bertoni, 2004). Image taken from Warnock (1997), permission pending. |
Non-Hodgkin's lymphomas are five times more common than Hodgkin's lymphomas and represent four percent of all cancers diagnosed (Abbasi and Sprano, 2004). Because it has only been classified as its own subset of non-Hodgkin's lymphoma for twelve years, it is difficult to estimate the prevalence of MCL, but most sources cite MCL as accounting for between four and ten percent of non-Hodgkin's lymphomas (Abbasi and Sprano, 2004, The Leukemia and Lymphoma Society, Freedman and Friedberg 2005, Bertoni et al., 2004). The age range at presentation is usually between 35 and 85, with 60 as the median age at diagnosis (Abbasi and Sprano, 2004). Men are four times more likely to develop MCL (Abbasi and Sprano, 2004). No pathogenic agent has been directly linked to MCL; however, recent studies suggest the Hepatitis C virus can increase susceptibility to non-Hodgkin's lymphomas (Bertoni et al., 2004). While no particular genes have yet been linked to a predisposition for MCL, familial cases of MCL have occurred, suggesting MCL may have a genetic component (Bertoni et al., 2004).
MCL is usually diagnosed at advanced stages (III and IV) and frequently will involve peripheral blood, bone marrow, and other extranodal sites (Yatabe, 2000). Presence in the gastrointestinal tract occurs often, and occasionally MCL will present as multiple polyposis (Freedman and Harris, 2005). MCL usually takes a fairly aggressive course with a median survival of three years (Freedman and Harris, 2005). Ten year survival rate is approximately five to ten percent (Abbasi and Sprano, 2004).
MCL begins with a malignant B lymphocyte in the mantle zone or outer edge of the lymph node (Yashushi et al. 2000). This malignant B lymphocyte will give rise to a clonal population of malignant small to medium size lymphocytes that will colonize the mantle zone of the lymph node. Continual growth of the malignant population will lead to the breakdown of normal lymph node barriers, and the cancer spreading throughout the entire lymph node (The Leukemia and Lymphoma Society). Malignant cells may then spread through the blood stream to other lymph nodes and other organs, most commonly bone marrow (The Leukemia and Lymphoma Society).
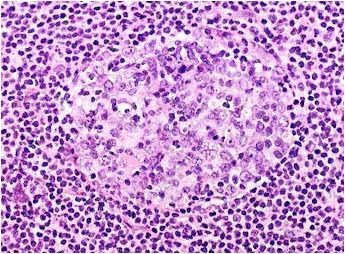
| Figure 4. Neoplastic mantle cell lymphoma cells infiltrating a germinal center. Image taken from Warnock (1997), permission pending. |
Diagnositc Procedures
MCL is often times difficult to diagnose because its morphology overlaps with other B-cell lymphomas. Symptoms can include but are not limited to fatigue, anemia, loss of apetite, tenderness around lymph nodes and/or spleen (Abbasi and Sparano, 2004). While there is considerable variance in symptoms between individuals, ninety percent of MCL patients present generalized swelling of lymph nodes (Abbasi and Sparano, 2004). An additional 60 percent have a swollen spleen (Abbasi and Sparano, 2004). Palpable masses in the skin, breast, and salivary glands are less common but can occur in patients with MCL (Abbasi and Sparano, 2004). Diagnosis of MCL usually involves a tissue biopsy of a lymph node (Witzig, 2005). A fine needle aspiration may be performed if a lymph node is not accessible; however, an aspiration is not sufficient for diagnosis (Freedman and Harris, 2005). There is no single test for MCL and the combined interpretation of a range of molecular and chemical tests along with microscope analysis of the lymph node tissue is used to diagnose MCL (Hankin and Hunter, 1999). Flow cytometry and immunohistochemistry can be used to differentiate small B-cell malignant lymphomas by the expression of surface immunoglobulins, CD19, CD20, CD5, CD10, and CD23 (Hankin and Hunter, 1999). Immunohistochemical stains for cyclin-D1 expression are then used to confirm the morphologic diagnosis of MCL (Hankin and Hunter, 1999). Less frequently Polymerase Chain Reaction and Southern Blot may be used to detect MCL's characteristic translocation at t(11;14)(q13;q32) (Hankin and Hunter, 1999). However, because some cases do not present this translocation, these tests can not be used to rule out MCL (Hankin and Hunter, 1999, Freedman and Harris, 2005 ).
To facilitate appropriate treatment strategies, MCL disease progression has been broken up into the following four stages according to the Ann Arbor system for classifying lymphomas (Freedman and Friedberg 2005):
Stage I- The cancer is found in a single lymph node region or a single extranodal site.
Stage II- The cancer is found in two or more lymph node regions on the same side of the diaphragm or there is localized involvement of an extra lymphatic organ or site and one or more lymph nodes on the same side as the diaphragm.
Stage III- The cancer is found in lymph node regions on both sides of the diaphragm and may also be accompanied by involvement of the spleen or by localized involvement of an extra lymphatic organ or site or both.
Stage IV- Diffuse or disseminated involvement is found in one or more extralymphatic organs or tissues, with or without associated lymph node involvement.
Molecules with Prognostic Significance
The presence of certain proteins in MCL tumor sites has been linked to overall disease prognosis. High levels of Ki-67 have been associated with high proliferation and poor prognosis (Bertoni et al., 2004). High expression levels of nuclear survivin, an inhibiting protein in both cell cycle and apoptosis, have also been strongly associated with the proliferative activity of MCL (Bertoni et al., 2004). High expression of the antiapoptotic molecule Mcl-1 in MCL patients has also been associated with proliferation, high expression of the tumor suppressor p-53, and blastoid transformation (Bertoni et al., 2004).
Evolution of Treatment Strategies
Treatment for MCL is still evolving. This is in part because MCL is a newly classified subtype of B-cell lymphoma, and research has not yet provided a universally accepted protocol. It is also in part because MCL carries a very poor prognosis when treated with conventional lymphoma treatment regimens. Patients presenting with limited disease (Stage I and II) seem to respond well to combined treatment with radiation and chemotherapy and have long term survival (Bertoni et. al, 2004). These regimens, though usually the initial treatment offered (The Leukemia and Lymphoma Society) are frequently inadequate for more advanced Stage III and IV disease (Bertoni et. al, 2004).
Chemotherapy
The typical chemotherapeutic regimen is CHOP (cyclophosphamide, doxorubicin hydrochloride, oncovin and prednisone) (Lenz et al, 2005). Another more aggressive regimen is also now being implemented called hyper-CVAD (cyclophosphamide, vincristine, doxorubicin, decadron, cytarabine, and methotrexate) (Witzig, 2005, Drach, 2005) These regimens may be used alone or in combination with bone marrow or stem cell transplantation (Witzig, 2005).
Bone Marrow Transplant
Both autologous and allogeneic bone marrow transplantations are also being used to treat MCL. In autologous bone marrow transplants, the patient's own stem cells are harvested while the patient is in remission following chemotherapy/radiation (Yvette et. al, 2004). Allogeneic bone marrow transplants, on the other hand, rely on harvested stem cells or bone marrow from an immunologically compatible donor, most often a relative (Yvette et. al, 2004). The patient then undergoes near lethal radiation and/or chemotherapy with the intention of killing all of the lymphoma cells. In the process all of their normal hemapoetic cells are also killed leaving them without the ability to survive since their bone marrow has been essentially "wiped out". They are then infused with the stored cells, and the new stem cells re-populate their bone marrow and allow the growth of new cancer-free cells. There is also evidence that donor allogeneic bone marrow lymphocytes can recognize and attacks cancer cells leading to a "graft vs. leukemia effect" (Janeway Jr., 2005).
Rituximab and Other Promising Drugs
More recently rituximab has been added to the chemotherapy regimen and/or transplantation regimen. Rituximab is an antibody that targets the CD20 receptor. Since MCL expresses CD20, its treatment with rituximab should and has been shown to have additive benefit when administered in combination with conventional treatments (Witzig, 2005, Drach, 2005). Unfortunately as a single agent, rituximab has not been proven to be successful in arresting MCL proliferation (Witzig, 2005). Radioimmunotherapy using anti-CD20 antibodies radiolabled with radioactive iodine or yttriumhas is now being tested (Drach et. al, 2005). Other novel treatments are currently being evaluated that target the cyclin-D1 pathway (Drach et. al, 2005). Bortexomib and flavopiridol are new drugs currently in clinical trials that target this pathway through proteasome inhibition (Drach et. al, 2005). Researchers are currently searching for more effective non-toxic regimens to treat mantle cell lymphoma.