Back to Dan Heeren's Immunology Homepage
The Basics
pTα is an acronym for pre- T cell receptor α chain. During T cell development, it forms a heterodimer with TCRβ and complexes with CD3 at the cell surface to form a functional pre-TCR. This pre-TCR guides T cell development through the double negative stage (so-called because the T cell lacks both CD4 and CD8 co-receptors) to the double positive stage (CD4 +CD8 +). From here, the final α chain is expressed in place of pTα (Koyasu et al., 1997). One could say that pTα acts as a surrogate for the final α chain while its gene undergoes rearrangement in the nucleus and ultimate expression on the surface.
What it looks like
pTα is a transmembrane protein that is approximately 33 kD long (Ramiro et al., 1996). It has a cytoplasmic tail that is 114 amino acids long specifically with a crucial COOH-terminal proline-rich sequence (Aifantis et al., 2002). pTα bonds covalently to TCRβ and noncovalently with CD3 signal-transducing molecules (Mancini et al., 1999; Borowski et al., 2004). There are two isoforms, pTαa and pTαb. pTαb doesn’t have a transmembrane section, and it may be important in retaining TCRβ in the endoplasmic reticulum to prevent its over-expression on thymocytes, which would lead to negative selection (Aifantis et al., 2002; Trop et al., 2000). For the rest of this report, pTα shall refer to the standard pTαa isoform.
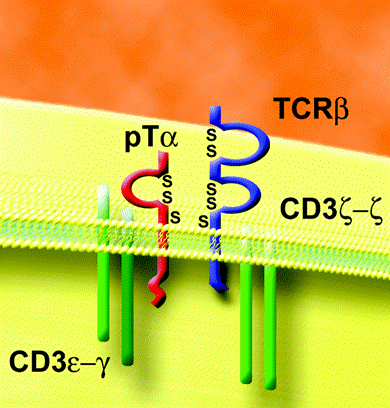
Figure 1:The pre-T cell receptor. The pTα is bound covalently to TCRβ, and the two are complexed with CD3 signal-transducers noncovalently. The pre-TCR is important in regulating T cell development before the final α chain can be produced. Permission given by Dr. Iannis Aifantis, image given in personal correspondence.
How it works
The pre-TCRs localize to membrane rafts where they are in close proximity to signaling molecules that are important for αβ T cell development such as Src kinases, ZAP-70 (Borowski et al., 2004) and the linker of activation of T cells (LAT) (Aifantis et al., 2002). It appears that the pre-TCR is ligand independent, meaning its mere surface expression provides the needed stimulus without needing to complex with any other extracellular molecules (Aifantis et al., 2002; Borowski et al., 2004). The proline-rich region on the cytoplasmic tail is similar to that on other stimulatory molecules that interact with Src kinases, so it is possible that the pTα behaves in a similar fashion by binding to their SH3 domains to begin a signal cascade. TCRα only has a cytoplasmic tail of 3 amino acids, greatly reducing the possibility that it directly binds or signals via cytoplasmic proteins; however, the greatly enlarged pTα tail strongly suggests that it does provide cytoplasmic signals. It is known for sure that the cytoplasmic tail is required for effective selection, proliferation, and survival of TCRβ-expressing thymocytes. An eventual downstream effect is the increase in intracellular [Ca 2+], needed for nuclear translocation of the transcription factors NFκB and NFAT (Aifantis et al., 2002). We know that pTα fills some role that TCRα can’t, as evidenced by experiments replacing pTα with TCRα. There is an extreme decrease in the amount of proliferation, survival, differentiation, and αβ T cell lineage commitment made by thymocytes (Borowski et al., 2004). Ultimately, there is only 2% of the normal level of double positive thymocytes when pTα is not expressed (Mancini et al., 2001).
TCR expression
During thymocyte development, the cell starts as a stem cell, becomes a double negative cell, then a double positive cell, then finally a single positive cell. Within the double negative stage, there are 4 substages where other surface molecules change. CD44 +CD25 - becomes CD44 +CD25 + then CD44 lowCD25 + and finally CD44 -CD25 -. It is only within the double positive stage that the TCRα replaces the pTα chain at the surface to make a complete TCR (Janeway et al., 2005; Koyasu et al., 1997).
The TCRB gene is first rearranged during the CD44 lowCD25 + stage and is expressed at the surface with pTα. This expression causes the phosphorylation of RAG-2, which prevents it from rearranging the TCRB locus any more. Also, it is thought that the pre-TCR activates the Src kinase which leads to cell proliferation and expression of CD4 and CD8. During proliferation, there is no TCRA gene rearrangement. After proliferation and the expression of both CD4 and CD8, the machinery for TCRA rearrangement is activated again, specifically RAG-1 and RAG-2. TCRα replaces pTα on the surface to make complete TCRs which are used during selection (Janeway et al., 2005).
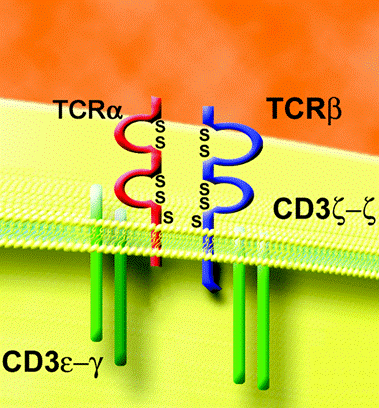
Figure 2: The final T cell receptor (TCR). Note that TCRα has replaced pTα. It has a drastically shorter cytoplasmic tail, implying that it does not directly transduce signals. Permission given by Dr. Iannis Aifantis, image given in personal correspondence.
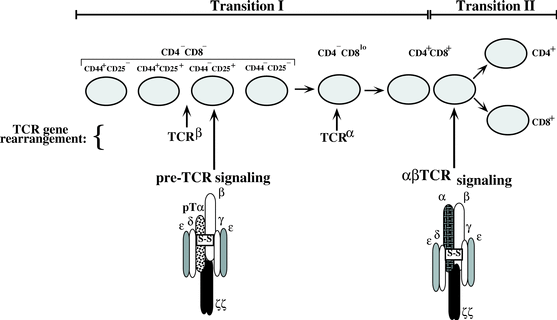
Figure 3: Summary of proteins involved in T cell development. Here one can see when TCRβ , pTα, and the final TCR are expressed and when the genes for the proteins are rearranged. Permission given by Dr. David Wiest, image from his personal site.
If the cell were to go through selection with both pre-TCR and TCR, there could conceivably be problems in allowing autoreactive cells through or preventing the passage of pathogen-specific cells. Normally, the pre-TCR complex is internalized and degraded at the same time that new TCR complexes are made in the endoplasmic reticulum. The expression of TCRα prevents the further expression of pre-TCR. This occurs at the post-translational level, as the presence of TCRα signals proteins in the endoplasmic reticulum to degrade any pTα. Also, there is preferential formation of TCR-αβ heterodimers over pTα/TCRβ heterodimers in the endoplasmic reticulum (Trop et al., 2000).
In modified cells where TCRα replaced pTα at the time when pTα would normally be expressed and in equal amounts, cells behaved just as they would if they had no pTα at all. This shows that pTα is not simply a place-holder until TCRα is produced. pTα might be important for localizing the pre-TCR to membrane rafts rich in transduction proteins or it might be needed for internalization and degradation of the pre-TCR (Borowski et al., 2004).
When something goes horribly wrong
When pTα isn’t made during T cell development, there is a complete breakdown of all the downstream events. In mice lacking pTα, there are still some T cells, but their numbers are greatly reduced. They are mostly the γδ T cells or double negative thymocytes. pTα must therefore play a critical role in expansion and αβ T cell pathway selection. Even so, the lack of pTα doesn’t appear to interfere with allelic exclusion at the TCRB gene locus (Xu et al., 1995).
Interestingly, even without pTα, cells can produce a diverse repertoire of TCRα chains. In mice deficient of pTα, TCRA rearrangement occurs in a few double negative cells, thus rescuing those few from cell death. This may occur in γδ T cells thus reverting them back to αβ T cells. This shows that while pTα is important for many aspects of development, it does not directly signal TCRA rearrangement (Mancini et al., 1999). This being the case, either the pre-TCR, TCR, or CD3 are needed to transcribe the TCRA gene (Mancini et al., 2001).
With an extreme lack of effective T cells in the immune system comes immunodeficiency. Without T cells, infections can run rampant because there are no T helper cells to activate B cells to release antibodies or macrophages to kill internal parasites or cytotoxic T cells to kill virally infected cells. There is no real adaptive immune response to speak of, and without early and intense protective care, a person would die, most likely from an opportunistic infection like pneumonia or sepsis.
References
Aifantis I, Borowski C, Gounari F, Lacoraza H, Nikolich-Zugich J, von Boehmer H. 2002 April 1. A critical role for the cytoplasmic tail of pT a in T lymphocyte development. Nature Immunology. < http://www.nature.com/ni/journal/v3/n5/pdf/ni779.pdf>. Accessed 2006 March 14.
Borowski C, Li X, Aifantis I, Gounari F, von Boehmer H. 2004. Pre-TCR a and TCR a Are Not Interchangeable Partners of TCR b during T Lymphocyte Development. The Journal of Experimental Medicine 199:607-615.
Janeway C, Travers P, Walport M, Shlomchik M. 2005. Immunobiology: the immune system in health and disease, 6 th edition. Garland Science Publishing: New York.
Koyasu S, Clayton L, Lerner A, Heiken H, Parkes A, Reinherz E. 1997. Pre-TCR signaling components trigger transcriptional activation of a rearranged TCR agene locus and silencing of the pre-TCR a locus: implications for intrathymic differentiation. International Immunology 9:1475-1480.
Mancini S, Candeias S, Di Santo J, Ferrier P, Marche P, Jouvin-Marche E. TCRA Gene Rearrangement in Immature Thymocytes in Absence of CD3, Pre-TCR, and TCR Signaling. The Journal of Immunology 167:4485-4493.
Mancini S, Candeias S, Fehling H, von Boehmer H, Jouvin-Marche E, Marche P. 1999. TCR a-Chain Repertoire in pT a-Deficient Mice Is Diverse and Developmentally Regulated: Implications for pre-TCR Functions and TCRA Gene Rearrangement. The Journal of Immunology 163:6053-6059.
Trop S, Rhodes M, Wiest D, Hugo P, Zuniga-Pflucker J. 2000. Competitive Displacement of pT a by TCR- a During TCR Assembly Prevents Coexpression of Pre-TCR and ab TCR. The Journal of Immunology 165:5566-5572.
Weist D. 1998. DEVELOPMENTAL REGULATION OF T CELL ANTIGEN RECEPTOR EXPRESSION AND FUNCTION. < http://www.fccc.edu/research/reports/current/wiest.html> Accessed 2006 March 16.
Xu Y, Davidson L, Alt F, Baltimore D. Function of the pre-T-cell receptor a chain in T-cell development and allelic exclusion at the T-cell receptor b locus. Proceedings of the National Academy of Sciences of the United States of America 93:2169-2173.
© Copyright 2006 Department of Biology, Davidson College, Davidson, NC 28035
Send comments, questions, and suggestions to: daheeren@davidson.edu