I. Introduction.
RAG1 and RAG2 are two proteins necessary for immunoglobulin and T-cell receptor gene recombination. RAG stands for recombination-activating gene. RAG1 and RAG2 work closely together in receptor gene recombination. Therefore, immunologists often refer to the two proteins together as RAG protein. RAG1 and RAG2 make up the lymphoid-specific parts of V(D)J recombinase, a complex of enzymes that work together to join gene segments of B-cell and T-cell receptor genes (Janeway et al., 2005). All species that carry out V(D)J recombination have RAG1 and RAG2, and the expression levels of RAG1 and RAG2 correlate directly with V(D)J recombination activity (Oettinger et al., 1990). RAG1 contains most, and possibly all, of RAG1/RAG2 V(D)J reconbinase’s active site (Kim et al., 1999). RAG2 is essential in joining DNA segments during V(D)J recombination (Qui et al., 2001). In vitro, the RAG protein can act as a transposase. Speculation on this evidence suggests that the RAG protein originated as a transposase. One theory is that vertebrates adapted the RAG protein to allow V gene segment recombination, and this adaptation gave rise to the vertebrate adaptive immune system. The fact that no genes homologous to RAG genes have been found in nonvertebrates supports this theory (Janeway et al., 2005).
II. Expression
Developing lymphocytes express RAG1 and RAG2 while the lymphocytes assemble their antigen receptors. RAG1 and RAG2 have different levels of expression during different stages of B-cell and T-cell antigen receptor assembly. Lymphocytes actively express RAG1 and RAG2 at very early lymphoid development stages of initial gene rearrangements, turn off RAG1 and RAG2 expression during stages of lymphoid cell proliferation following successful initial gene rearrangements, and activate RAG1 and RAG2 expression when lymphoid cells stop dividing and go on to make final gene rearrangements. There are some slight differences in expression levels of RAG1 and RAG2. In both immature B-cells and thymocytes, RAG2 expression increases and decreases much more drastically than RAG1 expression during different stages of B-cell and T-cell development. This variation in fluctuation is likely due to a posttranscriptional mechanism (Lin et al., 1994).
After successful receptor gene rearrangement in immature B-cells, production of RAG1 and RAG2 continues. If the B-cell is not self-reactive then production of RAG protein decreases, but does not completely stop until the B-cell is fully mature. This decrease in RAG protein production is crucial in maintaining allelic exclusion. On the other hand, if the B-cell is self-reactive, production of RAG protein continues at the same levels, and receptor gene rearrangement continues as long as there are available and unused V and J gene segments. In thymocytes, the presence of the pre-T-cell receptors triggers the repression of RAG expression during the proliferative phase. The repression of RAG expression during the proliferative phase prevents rearrangement of the α chain locus during this phase. After the proliferative phase ends, RAG transcription begins again which allows rearrangement of the α chain locus. This variation in RAG expression allows each thymocyte with a successfully rearranged β chain to give rise to many different double-positive thymocytes (Janeway et al., 2005).
III. Function
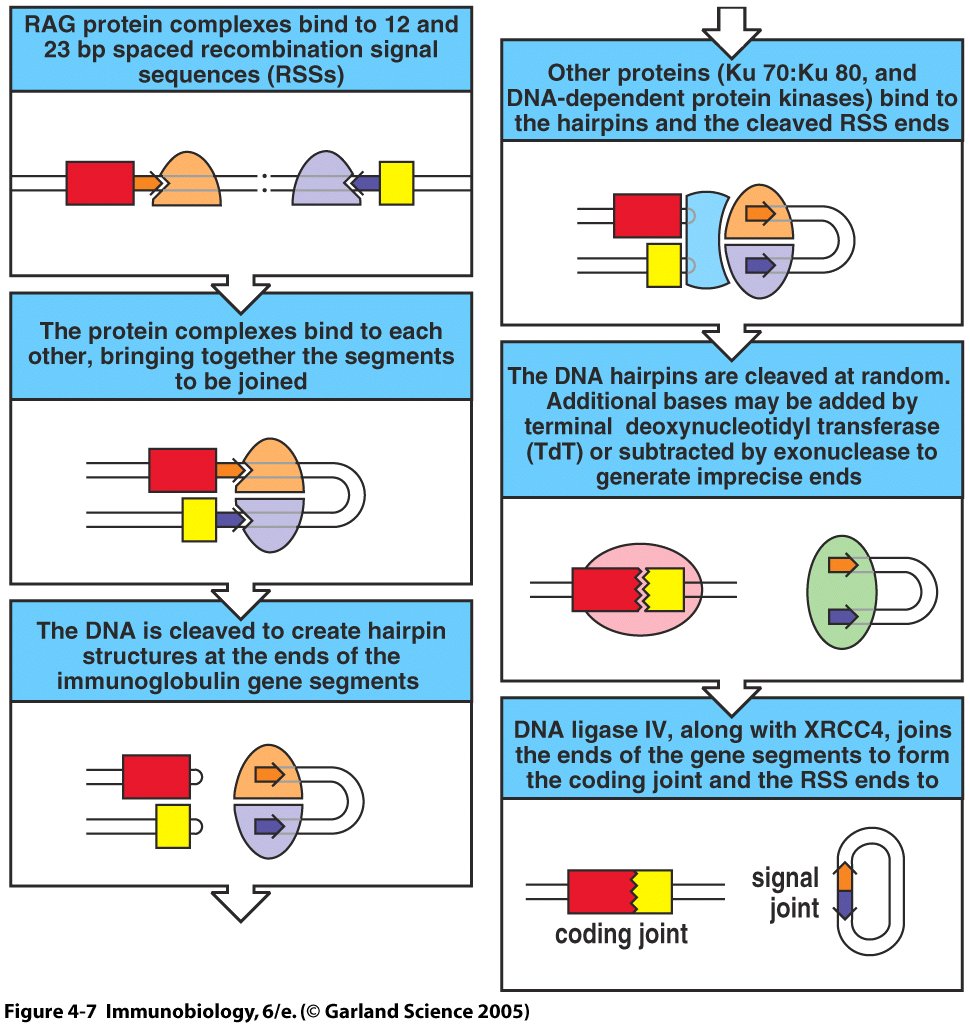
Both RAG1 and RAG2 are necessary for V(D)J recombination (Oettinger et al., 1990). RAG1 and RAG2 are not the only enzymes involved in V(D)J recombinase, but they have several crucial roles during V(D)J recombination. RAG1 and RAG2 initiate V(D)J recombination by cleaving DNA at recombination signal sequences. RAG1 and RAG2 also aid in V(D)J recombination by catalyzing the formation of the DNA hairpin (Melek et al., 200). In the first step, two RAG protein complexes (consisting of RAG1, RAG2, and high-mobility group proteins) recognize and align the two recombination signal sequences (Janeway et al., 2005). RAG1 directly recognizes the recombination signal sequences (Roman et al., 1996). Then, the RAG protein complex makes two single strand DNA breaks at the 5’ end of each bound recombination signal sequence. The free-floating 3’ –OH group creates a DNA “hair pin” by attacking the phosphodiester bond on the other strand. This creates a double strand break in the DNA. The RAG proteins help hold the DNA ends together. Finally, RAG1 and RAG2 nick open the DNA hairpin with a single-stranded break. The final result is recombination of V, D, and J gene segments (Janeway et al., 2005).

Figure 1: RAG protein involvment in the rearrangement of immunoglobulin gene segments. RAG protein complexes are illustrated as light purple and light orange colored domes even though all RAG protein complexes are identical (Janeway et al., 2005).
IV. Mutations
Mice and humans missing RAG1 or RAG2 or having a mutant form of RAG1 or RAG2 have problems with their immune systems. Mutations in either RAG1 or RAG2 result in an arrest of lymphocyte development because of failure to rearrange antigen receptor genes. Mice with genetically engineered mutations in the RAG gene and humans with no functional RAG protein completely lack both T-cells and B-cells (Janeway et al., 2005). Mice with a mutation in the RAG1 gene have small lymphoid organs, no mature B or T lymphocytes, and phenotypes of SCID, severe combined immune deficiency, mice (Mombaerts et al., 1992).
In humans, mutations in the RAG1 gene or RAG2 gene cause Omenn syndrome. Omenn syndrome is an autosomal recessive form of SCID. Clinical presentations of Omenn syndrome are similar to a graft versus host disease. These symptoms include rashes, eosinophilia, diarrhea, swollen lymph nodes, and failure to thrive (Janeway et al., 2005). Infants with Omenn’s syndrome present these symptoms anytime from within weeks of birth to age three months. The prevalence of Omenn’s syndrome in the United States is estimated to be approximately 1 in 50,000 (Shigeoka, 2006). Patients with Omenn’s syndrome have increased susceptibility to multiple opportunistic infections (Janeway et al., 2005). Patients develop life-threatening viral, fungal, and bacterial infections. Immediate treatment of patients with Omenn syndrome includes isolation to prevent contact with infectious agents, administration of antibiotics and sometimes antiviral agents to provide further protection against infection, and parenteral nutrition in response to patient’s diarrhea and failure to thrive. Ultimately, bone marrow transplant is the most successful therapy for patients with Omenn Syndrome. Scientists hope that identification of the recombinase mutations in RAG1 and RAG2 genes that cause Omenn Syndrome will make gene therapy available for patients with Omenn Syndrome.
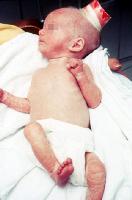
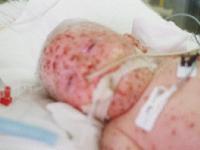 Figure 2: Infant with unique dermatitis that characterizes Omenn syndrome
Figure 2: Infant with unique dermatitis that characterizes Omenn syndrome
Permission for these images pending from www.emedicine.com/ped/topic1640.htm
V. Drug Interactions
In searching OMIM, no drugs are known to bind to RAG1 or RAG2.
Works Cited
Janeway, CA, Travers, P, Walport, M, Shlomchik, MJ. 2005. Immunobiology: The Immune System in Health and Disease. New York, NY: Garland Science Publishing.
Kim, DR, Dai, Y, Mundy, CL, Yang, W, Oettinger, MA. 1999. Mutations of acidic residues in RAG1 define the active site of the V(D)J recombinase. Genes Dev.13: 3070-3080.
Lin, WC, Desiderio, S. 1994. Cell cycle regulation of V(D)J recombination-activating Protein RAG2. Proc. Nat. Acad. Sci. 91: 2733-2737.
Melek, M, Gellert, M. 2000. RAG1/2-mediated resolution of transposition intermediates: two pathways and possible consequences. Cell 101: 625-633.
Mombaerts, P, Iacomini, J, Johnson, RS, Herrup, K, Tonegawa, S, Papaioannou,VE. 1992. RAG1-deficient mice have no mature B and T lymphocytes. Cell 68: 869-877.
Oettinger, MA. Schwatz, DG, Gorka, C, Baltimore, D. 1990. RAG1 and RAG2, adjacent genes that synergistically activate V(D)J recombination. Science 248: 1517-1523.
Qui, J, Kale, SB, Schlutz, HY, Roth, DB. 2001. Separation-of-function mutants revealcritical roles for RAG2 in both the cleavage and joining steps of V(D)J recombination. Molec. Cell 7: 77-87.
Roman, CA, Baltimore, D. 1996. Genetic evidence that the RAG1 protein directly participates in V(D)J recombination through substrate recognition. Proc. Nat. Acad. Sci. 93: 2333-2338.
Shigeoka, AO. 2 March 2006. Omenn Syndrome. <www.emedicine.com/ped/topic1640.htm> Accessed 9 March 2006.
Return to my immunology home page
Questions and comments to lirogers@davidson.edu
Return to Immunology home page
Return to Davidson College home page