This web page was created as an assignment for an undergraduate course at Davidson College.
The Story of a Yeast Receptor Gene and an Unknown Neighbor
PART 1: YEAST GENE
Annotated Gene: STE2 (YFL026W)
STE2 encodes the alpha-factor pheromone receptor found on mating-type a cells in the yeast species Saccharomyces cerevisiae. Two haploid yeast are able to mate by fusing together in a mating process that involves a number of steps. STE2 is involved in one of these initial steps. There are two mating types in yeast: MATa and MATalpha. Each of these yeast types secretes a mating pheromone (a-factor or alpha-factor, respectively). The pheromone must be secreted by one mating type and bind to the pheromone receptor in the opposite mating type. The pheromone receptor is encoded by STE2 in MATa cells and STE3 in MATalpha cells. Both protein products of STE2 and STE3 are seven transmembrane domain receptors that transmit their signals through a heterotrimeric G protein; thus they are considered class D G-protein coupled receptors. After binding alpha factor, the receptor encoded by STE2 undergoes a conformational change, the associated G-alpha subunit exchanges GDP for GTP, and the beta-gamma complex is then released, activating downstream components of the pheromone response pathway. This pathway eventually affects morphogenesis, the cell cycle (arresting the cells in G1 until cell fusion), and transcription. When cells sense the pheromone, they initiate polarized growth towards the mating partner (shmooing). (Cherry et al., 1997 http://genome-www.stanford.edu/Saccharomyces/).
 a.
a.  b.
b.
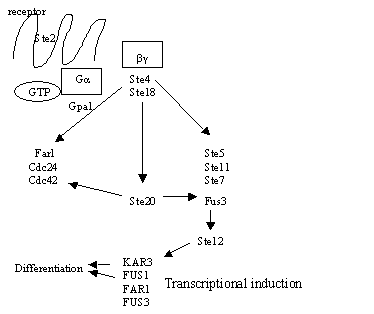
Figure 1: a. A very simple representation of the mating cascade in a cells once they sense alpha-factor (adapted from Dohlman, 2002; Sprague 1993). b. Example of shmooing (image pending permission from http://www.umassmed.edu/celldynamics/)
To recap, below is the gene ontology for STE2 (Cherry et al., 1997):
Molecular Function: mating-type alpha-factor pheromone receptor (Burkholder AC and Hartwell LH, 1985)
Biological Process: pheromone response
Cellular Component: integral plasma membrane protein
A recent paper looked at the range of "allowed amino acid substitutions" within the transmembrane regions of the recepter encoded by STE2. Investigators conducted random mutatgenesis and then screened for receptor function. They found that the receptor exhibits a surprising tolerance for amino acid substitutions. Every amino acid in the mutagenized regions of the transmembrane regions could be substituted by at least one other residue (Martin et al., 2002).
One group, however, did find a significant difference in receptor properties when they isolated a mutant that contained an amino acid substitution in the second half of the intracellular protruding C-terminal tail of the receptor. When this particular asparagine residue is replaced with serine, the receptor's interaction with the G-protein alpha and beta subunits is affected. These researchers concluded that the C-terminal tail of the receptor is involved in activation of the G protein (Duran-Alvela et al., 2001).
A null mutant is viable but sterile (it cannot mate). Yet yeast have the beauty of being able to reproduce by budding also. For this to occur, the STE2 receptor is not needed.
Location of STE2
STE2 is located on yeast chromosome VI (http://genome-www4.stanford.edu/cgi-bin/SGD/ORFMAP/ORFmap?sgdid=S0001868); its ORF name is YFL026W.
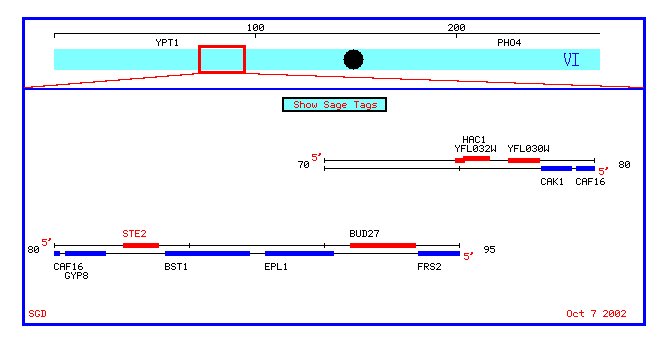
Figure 2: Screen shot of the chromosomal map surrounding the STE2 gene (from SGD).
STE2 can be seen as one of the genes in red. Just upstream from STE2 is an ORF named BUD27. This section of DNA encodes an ORF, but researchers are unsure of its exact use (if any) in the yeast cell. This webpage will explore its possibilities as a functional sequence.
STE2 Nucleotide Sequence
The nucleotide sequence of STE2 can be found in either SGD (the Saccharomyces cerevisiae Genome Database) or on NCBI. It has been copied and pasted below for convenience:
ATGTCTGATGCGGCTCCTTCATTGAGCAATCTATTTTATGATCCAACGTATAATCCTGGT CAAAGCACCATTAACTACACTTCCATATATGGGAATGGATCTACCATCACTTTCGATGAG TTGCAAGGTTTAGTTAACAGTACTGTTACTCAGGCCATTATGTTTGGTGTCAGATGTGGT GCAGCTGCTTTGACTTTGATTGTCATGTGGATGACATCGAGAAGCAGAAAAACGCCGATT TTCATTATCAACCAAGTTTCATTGTTTTTAATCATTTTGCATTCTGCACTCTATTTTAAA TATTTACTGTCTAATTACTCTTCAGTGACTTACGCTCTCACCGGATTTCCTCAGTTCATC AGTAGAGGTGACGTTCATGTTTATGGTGCTACAAATATAATTCAAGTCCTTCTTGTGGCT TCTATTGAGACTTCACTGGTGTTTCAGATAAAAGTTATTTTCACAGGCGACAACTTCAAA AGGATAGGTTTGATGCTGACGTCGATATCTTTCACTTTAGGGATTGCTACAGTTACCATG TATTTTGTAAGCGCTGTTAAAGGTATGATTGTGACTTATAATGATGTTAGTGCCACCCAA GATAAATACTTCAATGCATCCACAATTTTACTTGCATCCTCAATAAACTTTATGTCATTT GTCCTGGTAGTTAAATTGATTTTAGCTATTAGATCAAGAAGATTCCTTGGTCTCAAGCAG TTCGATAGTTTCCATATTTTACTCATAATGTCATGTCAATCTTTGTTGGTTCCATCGATA ATATTCATCCTCGCATACAGTTTGGAACCAAACCAGGGAACAGATGTCTTGACTACTGTT GCAACATTACTTGCTGTATTGTCTTTACCATTATCATCAATGTGGGCCACGGCTGCTAAT AATGCATCCAAAACAAACACAATTACTTCAGACTTTACAACATCCACAGATAGGTTTTAT CCAGGCACGCTGTCTAGCTTTCAAACTGATAGTATCAACAACGATGCTAAAAGCAGTCTC AGAAGTAGATTATATGACCTATATCCTAGAAGGAAGGAAACAACATCGGATAAACATTCG GAAAGAACTTTTGTTTCTGAGACTGCAGATGATATAGAGAAAAATCAGTTTTATCAGTTG CCCACACCTACGAGTTCAAAAAATACTAGGATAGGACCGTTTGCTGATGCAAGTTACAAA GAGGGAGAAGTTGAACCCGTCGACATGTACACTCCCGATACGGCAGCTGATGAGGAAGCC AGAAAGTTCTGGACTGAAGATAATAATAATTTATGA
The STE2 gene has a number of mammalian homologs because it falls into the class D G-protein coupled receptors category. You can see some of its homologs here.
STE2 Protein Information
The protein sequence was found in SGD.
MSDAAPSLSNLFYDPTYNPGQSTINYTSIYGNGSTITFDELQGLVNSTVTQAIMFGVRCG AAALTLIVMWMTSRSRKTPIFIINQVSLFLIILHSALYFKYLLSNYSSVTYALTGFPQFI SRGDVHVYGATNIIQVLLVASIETSLVFQIKVIFTGDNFKRIGLMLTSISFTLGIATVTM YFVSAVKGMIVTYNDVSATQDKYFNASTILLASSINFMSFVLVVKLILAIRSRRFLGLKQ FDSFHILLIMSCQSLLVPSIIFILAYSLEPNQGTDVLTTVATLLAVLSLPLSSMWATAAN NASKTNTITSDFTTSTDRFYPGTLSSFQTDSINNDAKSSLRSRLYDLYPRRKETTSDKHS ERTFVSETADDIEKNQFYQLPTPTSSKNTRIGPFADASYKEGEVEPVDMYTPDTAADEEA RKFWTEDNNNL
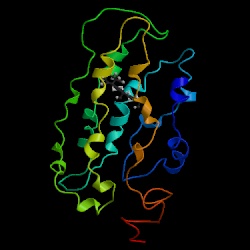
Because STE2 encodes an integral membrane protein, its structure is hard to determine within the membrane. I was unable to find picture of the protein. The ribbon structure below is another seven-transmembrane protein, bovine rhodopsin. Its structure is similar to seven-transmembrane receptors, and it is the only member of the receptor superfamily that has had its crystal structure solved (Pierce et al., 2002).
Figure 3: The crystal structure of bovine rhodopsin, a member of the seven-transmembrane superfamily of receptors (picture courtesy of PDB).
However, below is a screen shot of the Kyte-Doolittle (http://occawlonline.pearsoned.com/bookbind/pubbooks/bc_mcampbell_genomics_1/medialib/activities/kd/kyte-doolittle.htm) plot for the STE2 protein. As you can see, the protein is definitely a transmembrane protein, crossing the membrane at least 7 times.

Figure 4: Kyte-Doolitte plot of the STE2 protein product. Note that the protein crosses the red line of 1.8 seven times, confirming that it crosses the plasma membrane seven times.
Below is also the secondary structure prediction of the protein using PREDATOR (http://npsa-pbil.ibcp.fr/cgi-bin/npsa_automat.pl?page=/NPSA/npsa_preda.html) . As you can see, the protein seems to be only made up of alpha helices, extended strands, and random coils. By comparing the Kyte-Doolittle plot with the PREDATOR prediction, you can deduce that the outer pieces of the protein appear to be alpha helices. For example, look at amino acids 218 - 235. This corresponds fairly close to the outer membrane region of part of the protein as seen on the Kyte-Doolittle plot.
10 20 30 40 50 60 70
| | | | | | |
MSDAAPSLSNLFYDPTYNPGQSTINYTSIYGNGSTITFDELQGLVNSTVTQAIMFGVRCGAAALTLIVMW
cccccccccccccccccccccccceeeecccccccccccceeeeeehhhhhhhhhhhccchhhhhhhhhh
MTSRSRKTPIFIINQVSLFLIILHSALYFKYLLSNYSSVTYALTGFPQFISRGDVHVYGATNIIQVLLVA
hhcccccceeeeehhhhhhhhhhhhhhhhhhhhhccceeeeeecccccccccccceeeecchhhhhhhhh
SIETSLVFQIKVIFTGDNFKRIGLMLTSISFTLGIATVTMYFVSAVKGMIVTYNDVSATQDKYFNASTIL
hhhhhhhhheeeeeecccccceeeeeeeeeeecceeehhhhhhhhhcceeeecccccccccccccceeee
LASSINFMSFVLVVKLILAIRSRRFLGLKQFDSFHILLIMSCQSLLVPSIIFILAYSLEPNQGTDVLTTV
eeccccchhhhhhhhhhhhhhhhhhhcccccccceeeeecccccccccccceeeeccccccccccchhhh
ATLLAVLSLPLSSMWATAANNASKTNTITSDFTTSTDRFYPGTLSSFQTDSINNDAKSSLRSRLYDLYPR
hhhhhhhccccchhhhhhccccccceeeccccccccccccccccccccccccccccccchhhhhhhcccc
RKETTSDKHSERTFVSETADDIEKNQFYQLPTPTSSKNTRIGPFADASYKEGEVEPVDMYTPDTAADEEA
cccccccccccceeeccchhhhhhcccccccccccccccccccccccccccccccccccccccchhhhhh
RKFWTEDNNNL
hhhhhhccccc
Sequence length : 431
PREDATOR : Alpha helix (Hh) : 132 is 30.63% 310 helix (Gg) : 0 is 0.00% Pi helix (Ii) : 0 is 0.00% Beta bridge (Bb) : 0 is 0.00% Extended strand (Ee) : 70 is 16.24% Beta turn (Tt) : 0 is 0.00% Bend region (Ss) : 0 is 0.00% Random coil (Cc) : 229 is 53.13% Ambigous states (?) : 0 is 0.00% Other states : 0 is 0.00%
Figure 5: Secondary structure prediction of the protein product of STE2 (as predicted by PREDATOR).
Non-annotated Gene: BUD27
BUD27 is an ORF located on yeast chromosome VI in close proximity to STE2. Its molecular function, biological process, and cellular component are all unknown, but its sequence has been determined and can be found at the SGD. Its function is required for yeast viability on toxin exposure, but I could not find any references pertaining to this information that was simply cited at SGD. The null mutant is viable but exhibits K1 killer toxin hypersensitivity and diploid null mutants exhibit random budding (Cherry et al., 1997). The paper that documents BUD27 establishes it as a protein needed for bud site selection (Ni L and Snyder M, 2001).
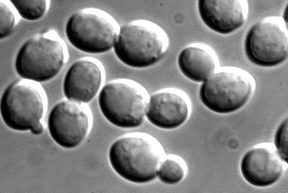
Figure 6: Yeast budding (image pending permission from http://www.acad.carleton.edu/curricular/ BIOL/faculty/szweifel/)
Protein Information for BUD27
The protein sequence can be found below (obtained from the SGD).
MDLLAASVESTLKNLQDKRNFLSEQREHYIDIRSRLVRFINDNDDGEEEGEGQGMVFGDI IISTSKIYLSLGYEYYVEKTKEEAITFVDDKLKLMEDAIEQFNLKIEEAKKTLDNLNHME DGNGIEEDEANNDEDFLPSMEIREELDDEGNVISSSVTPTTKQPSQSNSKKEQTPAVGPK EKGLAKEKKSKSFEENLKGKLLKRNDEVKKKVQPSKVDTENVYTFADLVQQMDQQDELED GYIETDEINYDYDAFENSNFKVNDNYEEDDEDEDEEEYLNHSIIPGFEAQSSFLQQIQRL RAQKQSQDHEREEGDVNKSLKPILKKSSFAENSDKKQKKKQVGFASSLEIHEVENLKEEN KRQMQSFAVPMYETQESTGIANKMTSDEFDGDLFAKMLGVQEADEVHEKYKEELINQERL EGEASRSNRRTRVSRFRKDRASKKENTLSTFKQETTRSVENEVVEKEPVVGDIIEKEPVV GDVIEKEPVVGDVIEKEPAVTDIVEREPAVNDIVERKPVVGDIIEKEPTINDIVEKEPEI NSKSEFETPFKKKKLKSLQKPRSSKSMKKKFDPKILENISDDDYDDDDDGNKKLLSNKSK NNTDEQDKFPSKIQEVSRSMAKTGATVGSEPVRITNVDYHALGGNLDDMVKAYSLGLYDD DLEEDPGTIVEKLEDFKEYNKQVELLRDEIRDFQLENKPVTMEEEENDGNVMNDIIEHEF PESYTNDEDEVALHPGRLQEEVAIEYRRLKEATASKWQSSSPAAHTEGELEPIDKFGNPV KTSRFRSQRLHMDSKP
By using PREDATOR to predict the secondary structure of the protein, you can see that it is mostly made up on alpha helices and random coils.
10 20 30 40 50 60 70
| | | | | | |
MDLLAASVESTLKNLQDKRNFLSEQREHYIDIRSRLVRFINDNDDGEEEGEGQGMVFGDIIISTSKIYLS
ccchhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhhheeeeccccccccccccccceeeeeeecccceeec
LGYEYYVEKTKEEAITFVDDKLKLMEDAIEQFNLKIEEAKKTLDNLNHMEDGNGIEEDEANNDEDFLPSM
cchhhhhhhhhhhhhhchhhhhhhhhhhhhhhhhhhhhhhhhhhcccccccccccccccccccccchhhh
EIREELDDEGNVISSSVTPTTKQPSQSNSKKEQTPAVGPKEKGLAKEKKSKSFEENLKGKLLKRNDEVKK
hhhhhhcccccceeecccccccccccccccccccccccccchhhhhhhhcccccccccccccccchhhhh
KVQPSKVDTENVYTFADLVQQMDQQDELEDGYIETDEINYDYDAFENSNFKVNDNYEEDDEDEDEEEYLN
hcccccceeeccchhhhhhhhhhhcccccccccccceeeecccccccccccccccccccccccchhhhhh
HSIIPGFEAQSSFLQQIQRLRAQKQSQDHEREEGDVNKSLKPILKKSSFAENSDKKQKKKQVGFASSLEI
cccccccccchhhhhhhhhhhhhhhcccccccccccccccccccccccccccccchhhhhccccccccee
HEVENLKEENKRQMQSFAVPMYETQESTGIANKMTSDEFDGDLFAKMLGVQEADEVHEKYKEELINQERL
eechhhhhhhhhhhhhhccccccccccccccccccccccccchhhhhhhccchhhhhhhhhhhhhhhhhh
EGEASRSNRRTRVSRFRKDRASKKENTLSTFKQETTRSVENEVVEKEPVVGDIIEKEPVVGDVIEKEPVV
hccccccccccchhhhhhhccccccccccccccchhhhhhhhhhhcccceeecccccccccccccccccc
GDVIEKEPAVTDIVEREPAVNDIVERKPVVGDIIEKEPTINDIVEKEPEINSKSEFETPFKKKKLKSLQK
ccccccccccccccccccccccccccccceeeccccccccccccccccccccccccccchhhhhhhhhcc
PRSSKSMKKKFDPKILENISDDDYDDDDDGNKKLLSNKSKNNTDEQDKFPSKIQEVSRSMAKTGATVGSE
cccchhhhhhccccccccccccccccccccceeeecccccccccccccccccchhhhhhhhhcccccccc
PVRITNVDYHALGGNLDDMVKAYSLGLYDDDLEEDPGTIVEKLEDFKEYNKQVELLRDEIRDFQLENKPV
cceeeeeccccccccchhhhhhhhccccccccccccceeeeecchhhhhhhhhhhhhhhhhhhhhccccc
TMEEEENDGNVMNDIIEHEFPESYTNDEDEVALHPGRLQEEVAIEYRRLKEATASKWQSSSPAAHTEGEL
eeecccccccchhhhhhhccccccccccccccccccchhhhhhhhhhhhhhhhhhhhccccccccccccc
EPIDKFGNPVKTSRFRSQRLHMDSKP
cccccccccccccccccccccccccc
Sequence length : 796
PREDATOR : Alpha helix (Hh) : 273 is 34.30% 310 helix (Gg) : 0 is 0.00% Pi helix (Ii) : 0 is 0.00% Beta bridge (Bb) : 0 is 0.00% Extended strand (Ee) : 51 is 6.41% Beta turn (Tt) : 0 is 0.00% Bend region (Ss) : 0 is 0.00% Random coil (Cc) : 472 is 59.30% Ambigous states (?) : 0 is 0.00% Other states : 0 is 0.00%
Figure 7: Secondary structure prediction of the protein product of BUD27 (as predicted by PREDATOR)
It is also very evident by looking at the Kyte-Doolittle hydropathy plot below that the BUD27 protein product is not an integral membrane protein. It is easy to compare this plot with that of the STE2 protein product.

Figure 8: The Kyte-Doolittle hydropathy plot for BUD27, illustrating that it is not a membrane-spanning protein.
The protein product does have a conserved motif with the Saccharomyces cerevisiae protein aldose 1-epimerase, which is the enzyme responsible for the anomeric interconversion of D-glucose and other aldoses between their alpha- and beta-forms.
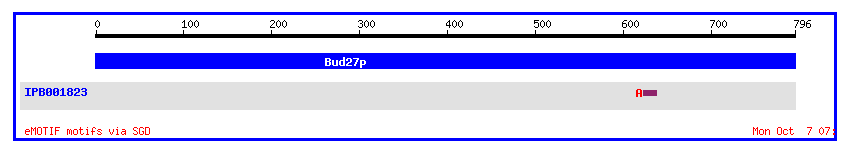
Figure 9: Conserved motifs of the protein product of BUD27 (http://genome-www4.stanford.edu/cgi-bin/SGD/protein/getDomain?sgdid=S0001871).
When the amino acid sequence was analyzed using NCBI's Conserved Domain database, only part of the protein's sequence matched that of another protein. In this case, amino acids 12-127 matched with 94.1% identity the transcriptional regulator DUF232. Maybe BUD27 is involved in transcription of other genes needed for budding? Upon BLASTing the nucleotide sequence, I found that BUD27 had very little similarity to any other genes. Very small parts of the sequences corresponded very well with small parts of other genes, such as the yeast phenylalanyl-tRNA synthetase beta subunit (FRS1) gene:
Yeast phenylalanyl-tRNA synthetase beta subunit (FRS1) gene, complete cds Length = 2005 Score = 79.8 bits (40), Expect = 2e-11 Identities = 40/40 (100%) Strand = Plus / Minus
Query: 2352 tttccgctctcaacggctccatatggacagtaaaccataa 2391
Sbjct: 2005 tttccgctctcaacggctccatatggacagtaaaccataa 1966
So far, there has been no structural information found concerning BUD27 or its protein product.
With this data, I cannot conclude anything concrete about the biological processes of this gene and protein product. It seems to have very little in common with other genes/proteins. It does have small similarities, but each of these is different as you can see from the sequence analyses. BUD27 could be involved in galactose metabolism, transcription regulation, or even in tRNA synthetase. A more comprehensive look into its particular functions needs to be assessed.
While the information obtained on this webpage is limited in the ways of sequence analyses, it is interesting to note that a gene involved in budding and a gene involved in the mating process are located on the same chromosome and rather close to one another. This could be an important aspect.
Go to STE2 Expression.
References
Burkholder AC and Hartwell LH. 1985. The yeast alpha-factor receptor: structural properties deduced from the sequence of the STE2 gene. Nucleic Acids Res 13(23):8463-75.
Cherry, J. M., Ball, C., Dolinski, K., Dwight, S., Harris, M., Matese, J. C., Sherlock, G., Binkley, G., Jin, H., Weng, S., and Botstein, D. 1997-2000. "Saccharomyces Genome Database" http://genome-www.stanford.edu/Saccharomyces/ Accessed: 04 October 2002.
Dohlman HG. 2002. G proteins and pheromone signaling. Annu Rev Physiol 64:129-52.
Duran-Alvela MJ, et al. 2001. The carboxy-terminal tail of the Ste2 receptor is involved in activation of the G protein in the Saccharomyces cerevisiae alpha-pheromone response pathway. FEMS Microbiol Lett. Apr 1; 197(1):65-71.
Martin NP, Celic A, Dumont ME. 2002. Mutagenic mapping of helical structures in the transmembrane segments of the yeast alpha-factor receptor. J Mol Biol. Apr 12;317(5):765-88 (abstract).
Ni L and Snyder M. 2001. A genomic study of the bipolar bud site selection pattern in Saccharomyces cerevisiae. Mol Biol Cell 12(7):2147-70 (paper)
Pierce KL, Premont RT, and Lefkowitz RJ. 2002. Seven-transmembrane receptors. Nature Reviews Molec Cell Biol Sept 3:639-650.
Sprague GF.
1991. Signal transduction
in yeast mating: receptors, transcription factors, and the kinase connection.
Trends Genetics 7(11-12):393-8.
Return Home
Return to the Genomics, Proteomics, and Bioinformatics Homepage
© Copyright 2002 Department of Biology, Davidson College, Davidson NC 28035
Send comments, questions, and suggestions to vistatler@davidson.edu