*This webpage was created as part of an assignment for an undergraduate course at Davidson College*
My Favorite Yeast Genes
Annotated Gene: TKL1
Location:
TKL1 is found on chromosome XVI of Saccharomyces cerevisiae. Its chromosomal location can be seen below:
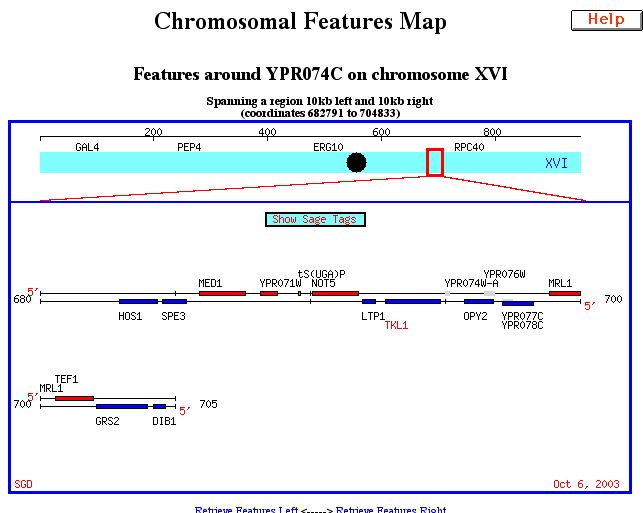
Fig.1: Chromosomal features map showing chromosomal location of TLK1 on Saccharomyces Cerevisiae chromosome XVI. (SGD, 2003; http://db.yeastgenome.org/cgi-bin/SGD/ORFMAP/ORFmap?sgdid=S0006278.
Gene Ontology:
TKl1 encodes transketolase 1. Its molecular function is transketolase activity, its biological process is pentose-phosphate shunt and its cellular location is the cytoplasm (SGD, 2003;http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?locus=TKL1).
Molecular Function: TLK1 functions in transketolase activity. In 1993 Sundstrom et al. cloned TLK1 from budding yeast Saccharomyces cerevesiae. They predicted that TLK1 was a 74kd protein. Through gene disruption and overexpression experiments they found that cells with disrupted and overexpressed TLK1 could not grow on fermentable carbon sources but grew normally on gluconeogenic carbon sources. They also found that cells without TLK1 could not produce aromatic amino acids due to the fact that transketolase is used to produce aromatic amino acid precursors.
Biological Process: Transketolase functions in the pentose-phosphate shunt. According to SGD 2003 the pentose-phosphate shunt is "The process by which glucose is oxidized, coupled to NADPH synthesis. Glucose 6-P is oxidized with the formation of CO2, ribulose 5-phosphate and reduced NADP; ribulose 5-P then enters a series of reactions interconverting sugar phosphates. The pentose phosphate pathway is a major source of reducing equivalents for biosynthesis reactions and is also important for the conversion of hexoses to pentoses" (SGD,2003; http://db.yeastgenome.org/cgi-bin/SGD/GO/go.pl?goid=6098).
Cellular Location is the cytoplasm. This excludes the plasma membrane and nucleus. The Kyte-Doolittle hyropathy plot below shows no peaks above 1.8. This indicates that TLK1 has no transmembrane domains.

Figure 2: Kyte-Doolittle Hydropathy plot based on the amino acid sequence of TLK1. The window setting is 19. The lack of peaks above 1.8 indicates the absence of transmembrane domains (http://occawlonline.pearsoned.com/bookbind/pubbooks/bc_mcampbell_genomics_1/medialib/activities/kd/kyte-doolittle.htm).
Protein Information:
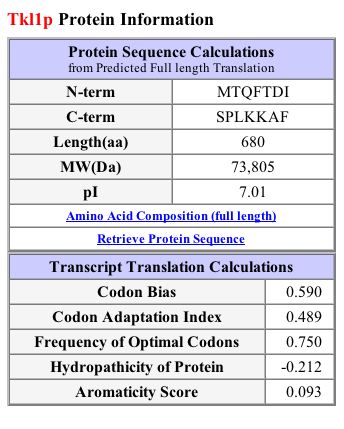
Figure 3: Protein information for TLK1 (SGD,2003; http://db.yeastgenome.org/cgi-bin/SGD/protein/protein?sgdid=S0006278).
In contrast to the Kyte-Doolittle hydropathy plot, the SGD 2003 protein page for TLK1 predicts a transmembrane domain at amino acids 434-456. It also shows signal peptide cleavage sites at amino acids 580 and 625 (SGD, 2003; http://db.yeastgenome.org/cgi-bin/SGD/protein/protein?sgdid=S0006278).
PDB gives the following structure for TLK1:
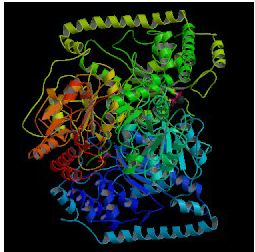
Figure 4: PDB image of assummed biological molecule of transketolase in Saccharomyces cerevisiae (PDB, 2003; http://www.rcsb.org/pdb/cgi/explore.cgi?job=graphics&pdbId=1TRK&page=0&pid=271071065499523).
In OMIM a human transketolase like-1 protein was found (OMIM, 2003; http://www.ncbi.nlm.nih.gov/htbin-post/Omim/dispmim?300044). The nucleotide sequence of this gene, called TKTL-1, was found in entrez and a blast2 comparison was made between the human TKTL-1 and yeast TKL-1. No significant alignment was found (NCBI Blast2, 2003;http://www.ncbi.nlm.nih.gov/gorf/wblast2.cgi?25057). OMIM describes the function of human tranketolase like- 1 as a "thiamine-dependent enzyme that linkes the pentose phosphate pathway with the glycolitic pathway" (OMIM,2003; http://www.ncbi.nlm.nih.gov/htbin-post/Omim/dispmim?300044).
Links for TLK1
Non-Annotated Gene: LEE1
Location:
The non-annotated gene was found on Sacchromyces cerevisiae chromosome XVI at the SGD website. The location of this ORF can be seen in figure 5.
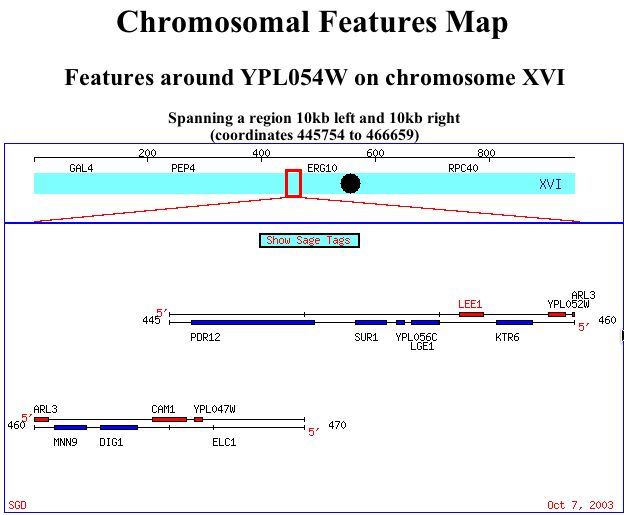
Figure 5: Map of ORF location on Saccharomyces cerevisiae chromosome XVI for LEE1 (SGD, 2003; http://db.yeastgenome.org/cgi-bin/SGD/ORFMAP/ORFmap?sgdid=S0005975).
Gene Ontology:
Based on information from SGD 2003's information page on LEE1 the molecular function, biological process, and cellular location of LEE1 are unkown (SGD, 2003;http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?locus=YPL054W).
Sequence Information:
The nucleotide sequence for LEE1 was obtained from SGD and used in a blast search. This search produced no close alignments. LEE1's next closest e values were .43. These corrresponded to the plasmodium falciparum chromosome 12, the Xestia c-nigrum granulovirus complete genome and a hypothetical protein in c-elegans. This means that a similar sequence is found in other organisms. This supports the belief that this ORF does infact code for a protien. However, these seqences are not very similar and they do not give any insight into the protein encoded by LEE1. The results from this search can be seen in figure 6.
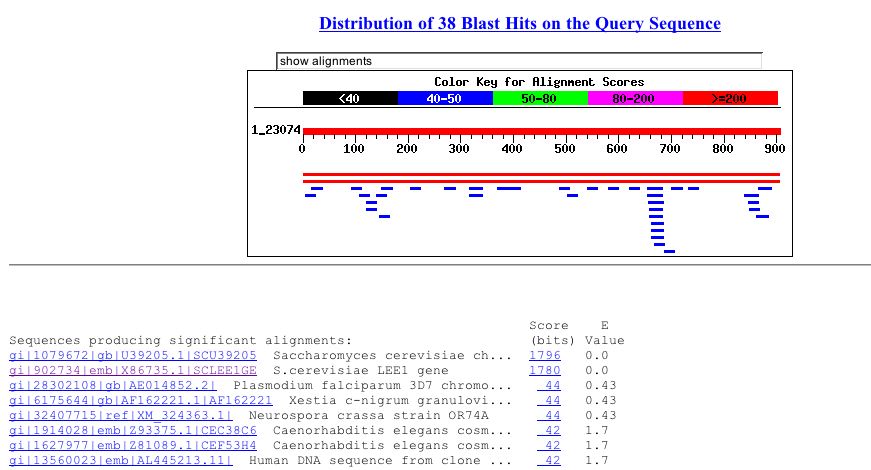
Figure 6: Blast performed using LEE1 nucleotide sequence. The most significant alignments other than LEE1 had e values of .43 (NCBI Blastn, 2003 http://www.ncbi.nlm.nih.gov/blast/Blast.cgi).
Conserved Domains:
A conserved domains search using the protein sequence of LEE1 found at the SGD 2003 protein information page indicated the possibility of a cleavege and polyadentylation factor with an e value of 2 e-37 and an alignement of 95.1%. This shows that the LEE1 protein may have a cleavage and polyadenylation specificity factor. They also list the possibilty of a LEE1 having a zinc finger based on an e value of 2e-04.

Figure 7: NCBI Conserved Domain results for the amino acid sequence of LEE1. This indicates the possibility of a cleavage and polyadenylation specificity factor(NCBI CD, 2003;.http://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi).
Blastp
Using the amino acid sequence of LEE1 from SGD a Blastp search indicated several alignments with proteins in saccharomyces pombe. Lee1 had an E value of 1e-11 with a spindle poison sensitivity related protein. Blastp also showed an E value of 4e-10 with a probable zinc finger protein in saccharomyces pombe. These results support the conserved domain search that indicated the presence of a zinc finger in the LEE1 protein. The blastp results can be seen in figure 8.
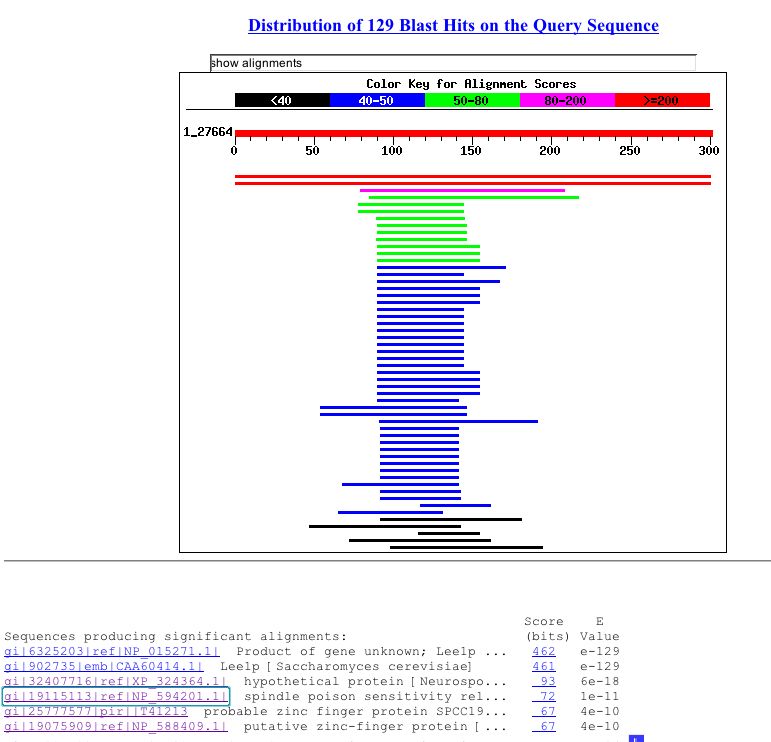
Figure 8: Blastp results for LEE1 amino acid sequence. Results show E value of 1e-11 with a spindle poison sensitivity related protien in saccharomyces pombe and 4e-10 with a probable zinc finger protein in saccharomyces pombe (NCBI Blastp, 2003; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi).
Kyte Doolittle Hydropathy Plot
The Kyte-Doolittle hydropathy plot for the LEE1 amino acid sequence showed no peaks above 1.8 when set to a window size of 19. This indicates that there are no transmembrane domains in the LEE1 protein. The Kyte-Doolittle Hydropathy can be seen in figure 9.
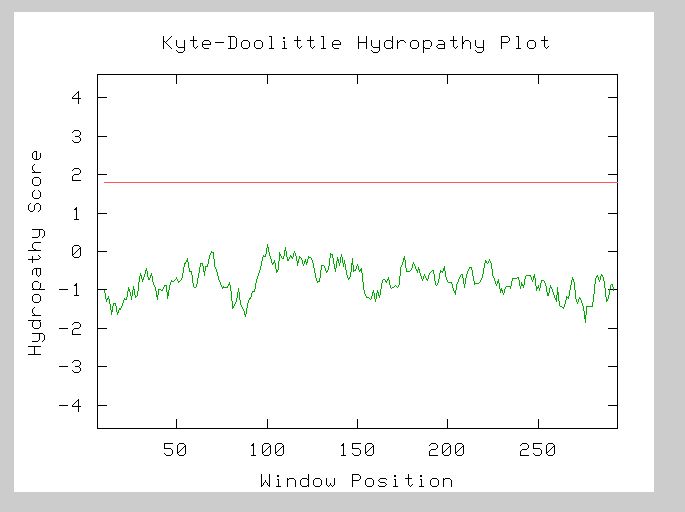
Figure 9: Kyte-Doolittle Hydropathy Plot for LEE1. The window is set at 19 and no peaks appear above 1.8 (http://occawlonline.pearsoned.com/bookbind/pubbooks/bc_mcampbell_genomics_1/medialib/activities/kd/slrkd.pl).
LEE1 Protein Information:
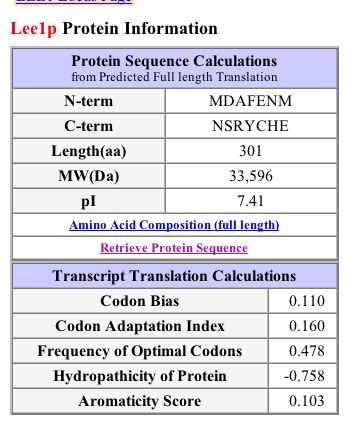
Figure 10: LEE1 protein information from SGD 2003's protein page for LEE1/YPL054W (SGD, 2003; http://db.yeastgenome.org/cgi-bin/SGD/protein/protein?sgdid=S0005975).
SGD's protein information predicts that the LEE1 protein has no transmembrane domains or signal peptide cleavage sites.
Cellular Role Predictions for LEE1
Based on its lack of transmembrane domains and signal peptide cleavage sites, LEE1 appears to function in the cytoplasm. It also shows significant alignments with a cleavage and polyadenylation factor. A blastp search gives further evidence of the presence of a zinc finger in LEE1. The LEE1 amino acid sequence has significant alignment with a possible zinc finger protiein in saccharomyces pombe. Therefore LEE1 may function in the cytoplasm as a zinc finger protien.
LEE1 Links:
SGD's Information Page for LEE1
References:
Dolinski, K., Balakrishnan, R., Christie, K. R., Costanzo, M. C., Dwight, S. S., Engel, S. R., Fisk, D. G., Hirschman, J. E., Hong, E. L., Issel-Tarver, L., Sethuraman, A., Theesfeld, C. L., Binkley, G., Lane, C., Schroeder, M., Dong, S., Weng, S., Andrada, R., Botstein, D., and Cherry, J. M. "Saccharomyces Genome Database" http://www.yeastgenome.org . Accessed: 7 Oct. 2003.
McKusick, Victor A. 1996 March, 28. "*300044 TRANSKETOLASE-LIKE 1; TKTL1" OMIM. http://www.ncbi.nlm.nih.gov/htbin-post/Omim/dispmim?300044 Accessed: Oct. 5 2003.
Sundstrom M, et al. (1993) Yeast TKL1 gene encodes a transketolase that is required for efficient glycolysis and biosynthesis of aromatic amino acids. J Biol Chem 268(32):24346-52. Abstract. Accessed: Oct 5 2003.