This web page was produced as an assignment for an undergraduate course at Davidson College
My Favorite Yeast Proteins: Fis1 & YIL067c
Fis1
I used the "find" function on my browser to search the Yeast-2-hybrid results from the Fields lab at the University of Washington, and found that Fis1 (also known as YIL065c) interacts with JSN1 and SFH1. The screen shot of what I saw is pictured below (Uetz et al., 2000).
![]()
Figure 1: Screen shot of Y2H-determined Fis1 interactions (Uetz et al., 2000 <http://depts.washington.edu/~yeastrc/th_11.htm>).
Searches in the MIPS database and at SGD revealed that Jsn1 suppresses certain alleles of Tub2 ( a constsituent of microtubules), and is involved in control of mitosis. Jsn1's molecular function is mRNA binding, suggesting that it binds to mRNA to suppress certain Tub2 alleles. Its cellular component is reported as unknown (SGD, 2003 <http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?locus=JSN1>; MIPS, 2003 <http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YJR091c>). It would make some sense for Fis1 to interact with Jsn1, as Jsn1 might be a suppressor that regulates translation of certain genes like Fis1 and Tub2.
Searches in the same databases showed that Sfh1 is part of the RSC complex, which alters the structure of chromatin. On a molecular level, it binds proteins, but in a larger view, its assistance in remodeling chromatin aids in the progression of the cell cycle (SGD, 2003 <http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?sgdid=S0004313>; MIPS, 2003 <http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YLR321c>). Because Fis1 and Sfh1 are both involved in remodeling of cell structures, it seems like it might be possible for them to interact.
A search at PathCalling produced the following figure for Fis1 (YIL065c).
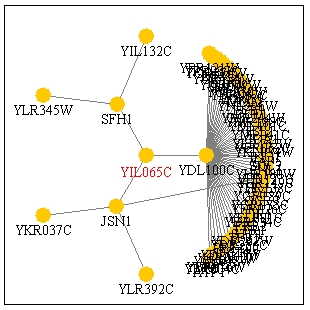
Figure 2: PathCalling Information for YIL065c, depicting proteins YIL065c interacts with. Figure data from Uetz et al., 2000. Figure from: PathCalling, 2003 <http://portal.curagen.com/extpc/com.curagen.portal.servlet.PortalYeastGene?geneIdIn=2837&keywordIn=YJR091C>.
It seemed odd to me that this figure shows Fis1 interacting with YDL100c, because
when I searched the Uetz et al. data above (both original and additional),
I did not find that Fis1 interacted with YDL100c. I'd like to know if YDL100c
was actually included in the original data, because that does not appear to
be the case. Regardless, if Fis1 does interact with YDL100c, it would be useful
to know its function. YDL100c is located in the endoplasmic reticulum and cytoplasm,
and is similar to an e.coli arsenical pump-driving ATPase (MIPS, 2003
<http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YDL100c>).
From this figure, it is clear that YDL100c interacts with many proteins, which
means it likely has a central role as an ATP-ase in a wide variety of pathways.
Interactions between Fis1 and YDL100c, Jsn1, and Sfh1 seem like they could be probabe, but researchers must always consider the possibility that results are incorrect or do not accurately describe what's actually happening in the cell. Some of these interactions could merely be artifacts.
To learn more about the protein interactions of Fis1, I searched the DIP database for Fis1. My search produced this graph:
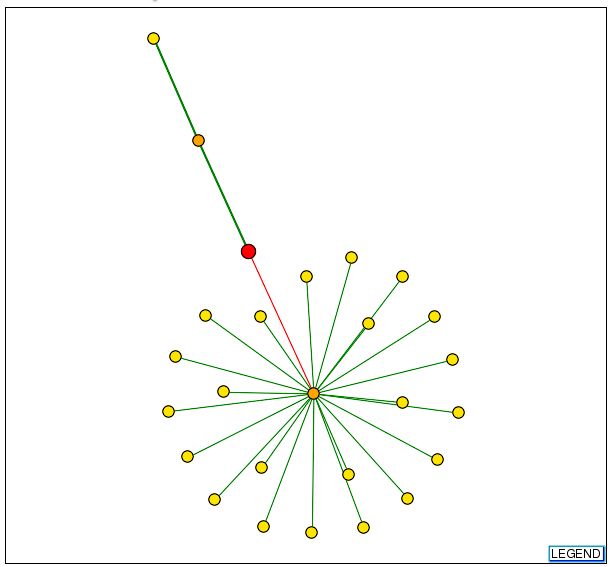
Figure 3: DIP graph for Fis1. Red node represents Fis1. Orange nodes are those one away from the root. All other nodes are yellow. Green lines represent intractions that have been verified; wider lines indicate that more experiments have verified the interaction. Red lines represent unverified interactions that have only thus far been seen in the high-throughput screen. The orange node above Fis1 represents Mdv1. The yellow node above Mdv1 represents Dnm1. The yellow node below Fis1 represents Jsn1 (DIP, 2003 <http://dip.doe-mbi.ucla.edu/dip/DIPview.cgi?PK=1897>).
These results reflect much of the research described in Web Assignment 2, where I noted that Fis1 is most likely necessary to localize Dnm1 and Mdv1 to the outer mitochondrial membrane. The mechanism (depicted below) proposed by the paper I cited suggested that that Fis1 initially only interacts with Dnm1, which in turn interacts with Mdv1. Througout the mitochondrial membrane remodeling process, Mdv1 may move to interact with Fis1, causing Fis1 to cease interaction with Dnm1 (Shaw & Nunnari, 2002). The results from the DIP figure are supported by this mechanism, although seem incomplete if Fis1 does in fact interact with Dmn1 on its own during initial stages.
The interaction between Fis1 and Jsn1 seems unlikely because it is not supported by any additional experimental results. Additionally, the DIP figure shows no interaction between Fis1 and Sfh1, as was seen in the Y2H results. This may indicate that this 'interaction' was merely an artifact.
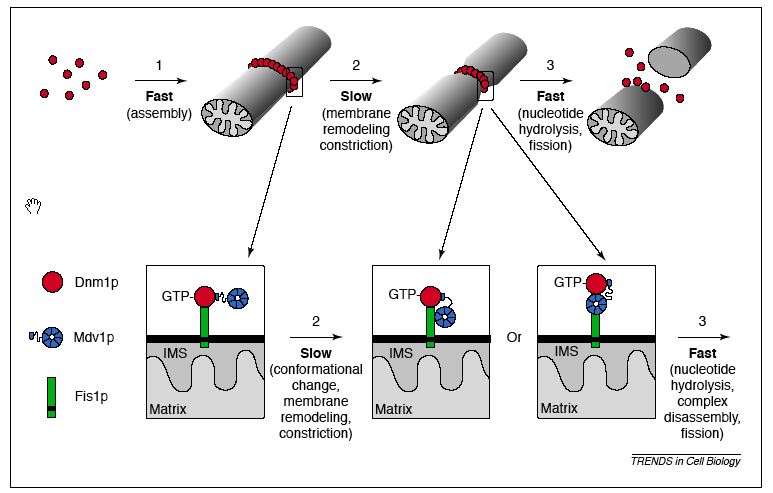
Figure 4: Proposed molecular model of mitochondrial fission: interactions between Dnm1p, Mdv1p, and Fis1p. Note: IMS stands for the intermembrane space of mitochondria. From: Shaw & Nunnari, 2002. Permission granted. Also included in Web Assignment 2.
I tried searching Benno Figure 1 for Fis1 (YIL065c), and found it in the section of the integraged circuit pictured below.
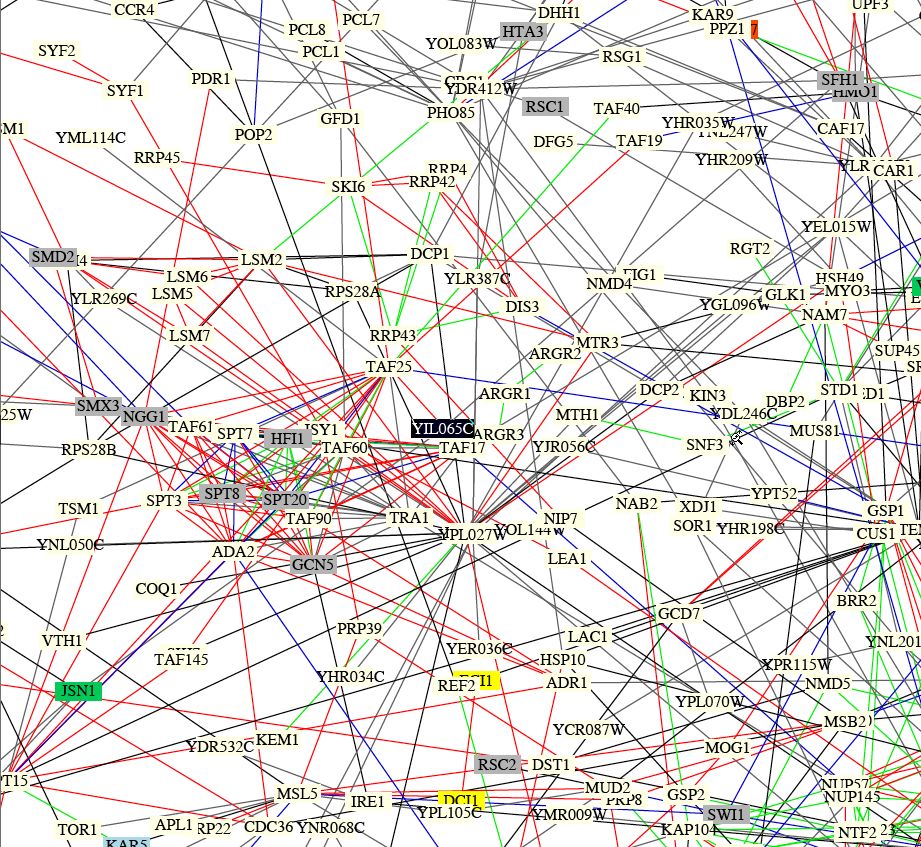
Figure 5: Screen shot of section of integrated protein circuit diagram. Red lines indicate that localization and cellular role for connected pairs are the same. Blue lines indicate similar localization, green lines indicate identical cellular roles, black lines indicate differences in both localization and cellular role, and gray lines indicate that localization and/or cellular role are unknown for one or both of the proteins. As fot the boxes, blue indicates a role in membrane fusion, gray indicates a function in chromatin structire, green proteins aid in cell structure, yellow proteins contribute to lipid metabolism, red indicates a role in cytokinesis, and light yellow indicates an unknown role. When YIL065c is not highlighted as it is in the figure, it appears light yellow (Schwikowski et al., 2000).
I searched the figure for Mdv1 and Dnm1 using gene names as well as SGD annotation, but found no results. This was frustrating, becuase I would have hoped to see Fis1 interacting with them to support results from other databases and papers. It is difficult to get any hard data from this figure, as many of the connections are unclear. However, Jsn1 (whose green box indicates that it plays a role in cell structure) is close to Fis1, indicating that an interaction between them is probably depicted in the figure. My eye tells me that the line connecting them is gray, which would indicate that little is known about the supposed interaction. It also appears that Fis1 is connected by a red line to Taf25, which is a transcriptional activator (MIPS, 2003 <http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YDR167w>). Taf25 interacts with many other proteins, indicating that it plays a more central role than Fis1. It also looks like Fis1 may be connected to Dcp1, which is involved in mRNA processing (MIPS, 2003 <http://mips.gsf.de/genre/proj/yeast/searchEntryAction.do?text=YOL149w>). Both of these connections seem likely, but I can't confirm much more because the figure is somewhat unclear. Fis1 also sits right on top of Taf17, which acts as a transcription factor for RNA polymerase II (SGD, 2003 <http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?locus=Taf17>). A connection here might be likely as well.
I also checked the PDFs for aging, membrane, and degradation. Fis1 was not assigned a particular function in any of these integrated circuits, but was was in the same location as in the above figure.
From PROWL, I learned that Fis1 has a molecular mass of 17.7 kDa and a pI of 9.3 (PROWL, 2003 <http://129.85.19.192/prowl/proteininfo.html>). These sepecifics are necessary when analyzing 2D gels for Fis1 results; they represent the coordinates where a spot for Fis1 should be found. However, I found no results for Fis1 in 2D gels: a search in the Swiss-2D PAGE database, as well as searches in other 2D PAGE databases yielded no results. When I looked at the S. cerevisiae 2D gel, I saw a few faint spots in the area where I would expect to see Fis1, but I can't be sure about any of them.
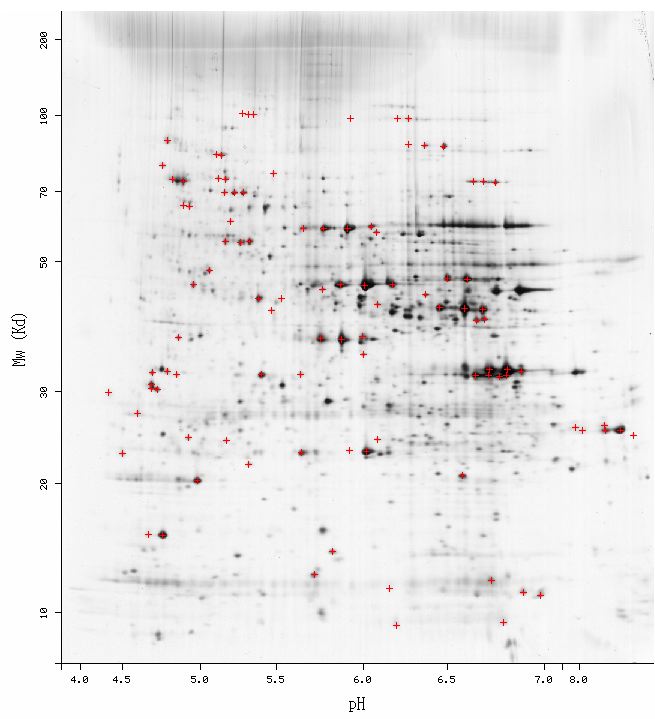
Figure 6: S.cerevisiae 2D gel. Red crosses indicate identified genes. We would expect to see Fis1 in the lower right hand corner at the coordinates (9.3, 17.7) (Swiss 2D PAGE, 2003 <http://ca.expasy.org/cgi-bin/map2/def?YEAST>).
I searched the TRIPLES database and found no clones with insertions in Fis1.
A search at What is There? yielded no results.
Conclusions
The searches described on this page have provided me with some new information about Fis1. From the Y2H experiments and PathCalling figures, I can predict the Jsn1 may regulate expression of Fis1 by binding to Fis1 mRNA. Fis1 may also have an interaction with Sfh1, but this seems unlikely because Sfh1 was not present in the DIP figure. However, Fis1 and Sfh1's similar roles in organization of cell structure is support for a possible interaction. Fis1 also likely interacts with the ATPase YDL100c, which interacts with a vast range of other proteins as well. Because YDL100c is present in the endoplasmic reticulum, it may aid in post-translational modification or transport of Fis1. My predictions from web assignment 2 were supported by the interactions I found using DIP, which showed interactions between Fis1 and Mdv1 and the subsequent interactin between Mdv1 and Dnm1. These proteins have all been implicated in mitochondrial fission. It was slightly perplexing that neither Mdv1 or Dmn1 showed up in any of my other searches, but more research must be done. From Benno Figure 1, I learned that Fis1 transcription may be initiated in part Taf17 and Taf25, and that it may undergo mRNA processing via interaction with Dcp1.
Research Suggestions
Although much is already known about Fis1, some further research could help confirm or negate some of the results I've found from searching the proteomics databases.
I would like to see some sort of confirmation that Fis1 interacts with Mdv1 and maybe Dnm1. It is possible that the Y2H experiments did not reveal these interactions because Mdv1 and Dnm1 have to be folded in certain specific ways to interact with Fis1. I would like to do co-immunoprecipitation experiments with Fis1 and Dnm1 and Mdv1, respectively. It would be necessary to have antibodies to all three proteins to perform this experiment. Still, this might not tell me if Fis1 interacts directly with the probed protein, as other proteins could be present in a binding clump.
Co-immunoprecipitation experiments could be performed with the other proteins that Fis1 has been suggested to interact with.
As an additional confirmation of interaction, if two proteins have the same subcellular localization, it is likely that they interact. I could perform immunofluorescence experiments with the proteins that Fis1 possibly interacts with (Dnm1, Mdv1, YDL100c, Sfh1, Jnm1, Taf17, Taf25, or Dcp1) and see if they localize to the same place as Fis1. This would require me to perform immunofluorescence with Fis1 as well. For example, if Fis1 truly does interact with Taf17 during transcription, I would expect to see fluorescence in the nucleus when Fis1 was probed and when Taf17 was probed. Probing would be done with a primary antibody to Fis1 or Taf17, followed by application of a secondary fluorescent antibody that would stick to the primary antibody. Similar experiments could be performed with the other suggested proteins.
Fis1's Biological process is still reported as unknown, but in order to elucidate this, the purposes of mitochondrial fission as a whole must be elucidated. It might be useful to create knockouts lacking all genes necessary for mitochondrial fission, and observe how yeast cells store and transport energy.
YIL067c
My previous two web pages included some predictions about YIL067c. By the guilt-by-association method, I predicted YIL067C may be integral to the nuclear membrane and may play a role in transcription. It might also be involved in signaling at either the cell membrane or the nuclear membrane. I also concluded that YIL067c is an intermembrane protein with an LCCL domain that may be a region of functional importance in the way YIL067c folds.
I used the "find" function on my browser to search both sets of Y2H results from the Fields lab, but found no results.
There is a DIP citation for YIL067c, but the picture of the graph would not show up in any browser. I tried this on two separate occasions on different days.
When I searched for YIL067c in Figure 1 from Schwikowski et al., the search returned with no hits, indicating that it was not included in the figure, which contained 2,039 different yeast proteins. It was not included in the aging, membrane, and degradation PDF's either.
From PROWL, I learned that YIL067c has a molecular mass of 75.4 kDa and a pI of 6.0 (PROWL, 2003 <http://129.85.19.192/prowl/proteininfo.html>). As stated above in reference to Fis1, this information would be useful in interpreting a 2D gel.
A search in the Swiss-2D PAGE database, as well as searches in other 2D PAGE databases yielded no results.
A search at What is There? yielded no results.
When I searched the TRIPLES database, I found two clones with insertions in YIL067c.
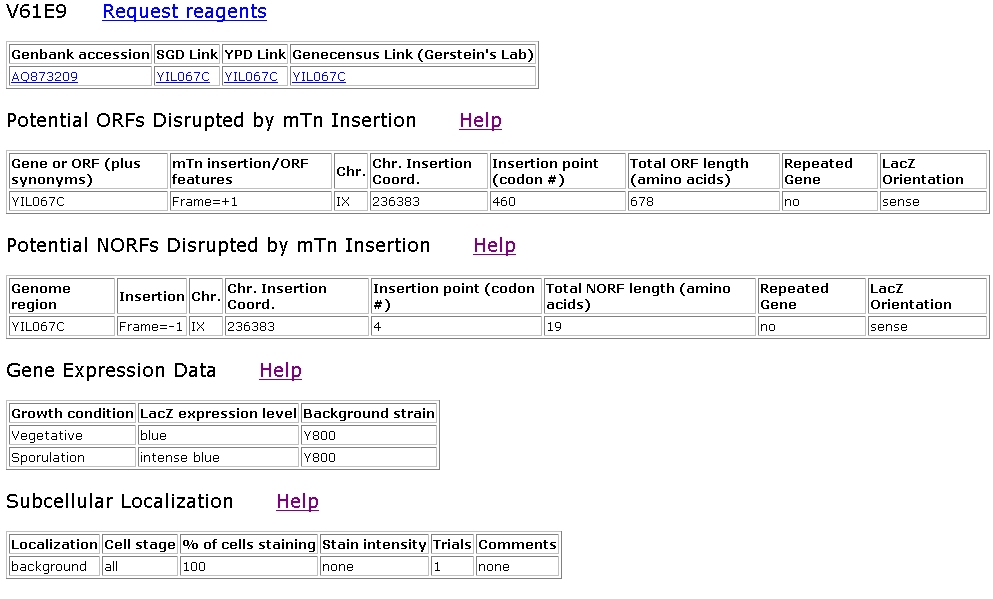
Figure 7: Screen shot of TRIPLES information for strain V61E9, which contained an mTn insertion in YIL067c (TRIPLES, 2003 <http://ygac.med.yale.edu/triples/get_clone_info.asp?cloneid=V61E9>).
LacZ expression levels were very high during sporulation and were high during a vegetative state. This indicates that the mTn certainly did insert downstream of the YIL067c promoter, and that YIL067c was turned on at the time as beta-galactosidase expression. Using monoclonal antibodies to a hemagglutinin tag on YIL067c of transformed strains, researchers were attempting to find a subcellular localization for YIL067c. However, no cell areas stained above a background level, so no conclusive results were found. The same was the case in the V54C10 strain detailed below.
![]()
Figure 8: Screen shot of only notable disruption phenotype from V61E9 clone (TRIPLES, 2003 <http://ygac.med.yale.edu/triples/get_clone_info.asp?cloneid=V61E9>).
The only non-wild-type disruption phenotype was a medium phenotype for HapTra, which indicates that haploid transformed cells were slightly inviable. Becuase this phenotype was not seen in the strain below, which also had an insertion in YIL067c, this indicates that the area around codon 460 may be important to cell viability. For example, this area may be part of an exon as opposed to an intron.
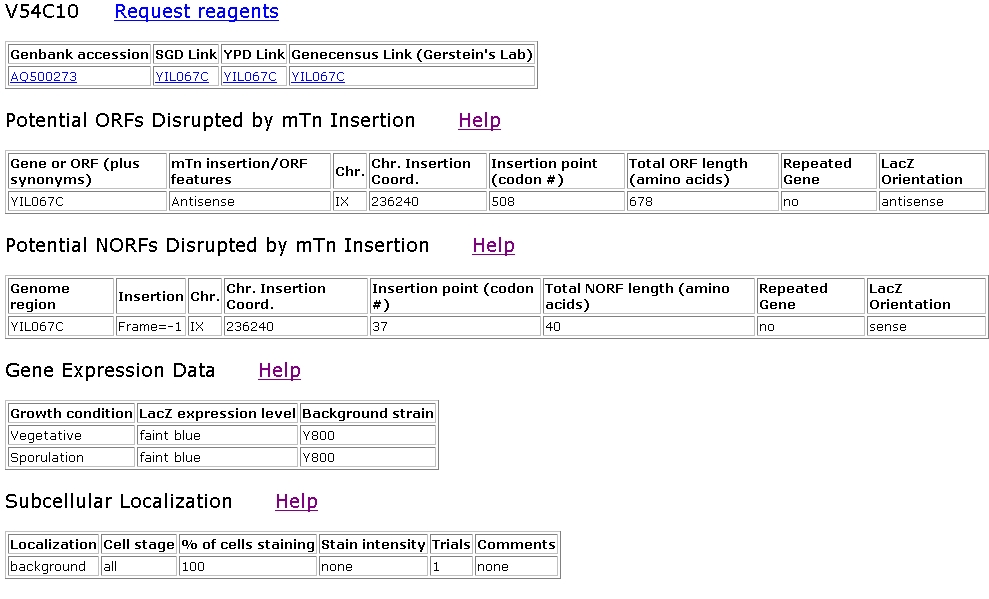
Figure 9: Screen shot of TRIPLES information for strain V54C10, which contained an mTn insertion in YIL067c (TRIPLES, 2003 <http://ygac.med.yale.edu/triples/get_clone_info.asp?cloneid=V54C10>).
The V54C10 strain reveals little about YIL067c. The fact that the insertion produced no phenotypes indicates that the insertion may have occurred in an intron, suggesting that codon 508 is within an intron.
Conclusions
Unfortunately, the proteomics databases revealed little new information about YIL067c. After my searches detailed in this web page, I do know that YIL067c's molecular mass is 75.4 kDa and its pI is 6.0. I can also predict that codon 460 is more important than codon 508, which suggests possible intron/exon regions or possible important /unimportant regions.
Research Suggestions
It would be useful to design yeast with YIL067c knocked out. Any altered phenotypes would suggest a functional role for YIL067c. Perhaps its function is associated with transcripton, as I predicted in web assignment 3. In this case, a knockout strain might show altered transcription of a number of genes. This would clue me in to which genes YIL067c serves to regulate.
I would also want to test my predicted localization of YIL067c to the nuclear membrane. I could perform immunofluorescense experiments by applying a YIL067c antibody to yeast cells and then adding a secondary fluorescent antibody. If my predictions were correct, I would hope to see glowing at the nuclear membrane.
Similarly, I could tag YIL067c with GFP and follow fluorescence throughout the cell cycle to see localization of YIL067c throughout the cell cycle.
Yeast 2 Hybrid experiments with YIL067c would be useful. In this method, YIL067c would be fused to a DNA binding domain and inserted in a plasmid. ORF's of interest would be fused to an activation domain and inserted in a different plasmid. Both plasmids would be transformed into a yeast cell, and only yeast transcribing the necessary reporter gene (which only happens if the two proteins interact) would survive. If researchers saw growing cells, they would know that the two proteins in the plasmids interact. if YIL067c is involved in transcription like I have predicted, I would expect to see it interacting with transcription factors and other molecules assocatied with transcription.
References
Schwikowski, B., Uetz, P., Fields, S. 2000. A network of protein-protein interactions in yeast. Nature Biotechnology.18: 1257-1261.
Shaw, J.M. & Nunnari, J. 2002. Mitochondrial dynamics and division in budding yeast. TRENDS in Cell Biol. 12(4): 178-184.
Uetz P., Giot L., Cagney G., et al. 2000. A comprehensive analysis of protein-protein interactions in Saccharomyces cerevisiae. Nature. 403:623-627. Results on web at <http://depts.washington.edu/%7Eyeastrc/th_11.htm>