-
This web page was produced as an assignment for an undergraduate course at Davidson College -
My Favorite Yeast Protein
EST1 and YLR243W
Databases
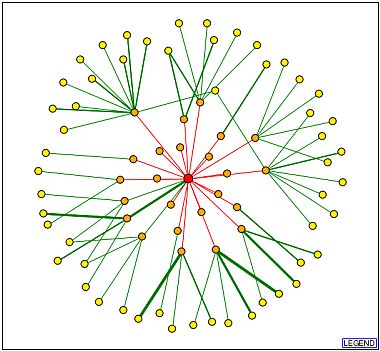
The Database of Interacting Proteins (DIP) generates graphs which characterize your favorite protein as a nod in an interacting system. As displayed below, the EST1 protein is placed in the center and the edges represent interactions. The red edges are unverified connections, but the thicker green lines represent more verifiable data. The strong green line links EST1 to CDC13, cell division control protein. This protein has well-characterized DNA binding function associated with telomere capping and maintenance.
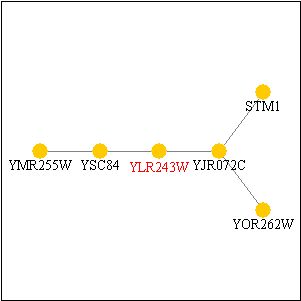
Before beginning with the proteomic characterization of YLR243W, I must first mention that I neglected to display the PathCalling characterization of the gene in the first web assignment. Although EST1 was previously displayed, YLR243W was not. I have displayed that figure below. All the genes in the figure are not characterized except STM1, which is listed by the Saccharomyces Genome Database as having "translation initiation factor activity" and being located in the ribosome.
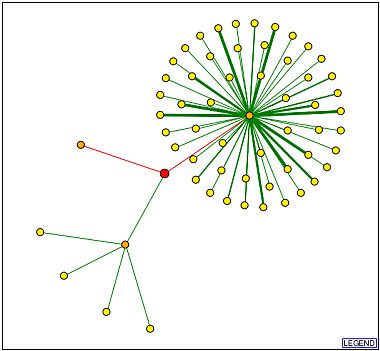
The Database of Interacting Proteins (DIP) also lists the YLR243W protein. As displayed below, the YLR243W protein is placed in the center and the edges represent interactions. The red edges are unverified connections, but the green lines represent more verifiable data (thicker lines indicate more experiments which verify the data). The only green line that touches YLR243W links it to YJR072C, a hypothetical unannotated protein. This is the same link displayed in the PathCalling picture above.
EST1 and YLR243W were not listed in the following databases: Protein Data Bank (PDB), Triples, Enzymes and Metabolic Pathways, EsPASy 2d Gels, or the updated Yeast 2-Hybrid database. The older Y2H database information from the Nature paper is the same data reflected in the PathCalling webpages (Uetz et al., 2000). The genes interactions in the Nature Biotechnology paper were all described in the diagrams above (Schwikowski et al., 2000)
Experiments
For my annotated gene, EST1, I have the good fortune of already working with a well-characterized protein. That being said, I also notice from the DIP picture above, that there are many proteins that hypothetically interact with EST1. Since these are predicted interactions but not experimentally determined, this warrants a specific and narrow type of investigation: verify or refute these hypothetical interactions. This is easy to do by creating a bait-prey system using the yeast-two hybrid system. It has been shown that EST1 does work in Y2H systems, only those red-line interactions were not confirmed in the Uetz experiment (Uetz et al., 2000). I predict some of these interactions will work (such as PET56, a methyltransferase) and others will not (such as URE2, a nitrogen catabolism protein).
In earlier web assignments, I speculated that YLR243W is associated with ribosome synthesis. If this hypothesis is true, then adjustments to YLR243W will affect the ribosomal output in the cell. This prediction may be tested using a previously described protocol to purify ribosomes (Inada et al., 2002). While knockout strains of YLR243W do not survive, yeast strains with YLR243W under pre-determined promoters can be designed. For example, two strains can be manufactured: one containing the gene under the control of a very strong up-regulating promoter, and another with the gene under a weaker down-regulating promoter. If YLR243W is involved in ribosome synthesis, I expect that ribosome content will directly correlate with the expression level of YLR243W. Since the YLR243W yeast knockout does not survive, I also predict that the experiment will not be confounded by redundant pathways.
Additionally, I would like to perform an immunoprecipitation to better determine the localization of this gene. Since it is an unannotated gene, we will have to first raise our own antibodies. Then, when precipitated, co-localized proteins can be separated and analyzed. Among those proteins, we can hope they will provide clues as to localization and function. For example, I previously predicted that the protein is cytosolic. That prediction was based on a hydropathy plot, which means that the hydrophilic nature of the protein may let it persist in the nucleus, the ER lumen, or the cell cytosol. For that matter, the plot may have been entirely misleading and the protein may be membrane-bound. Since the protein appears to be associated with ribosomes (and perhaps translation) I predict it will be found in the ER. I will wait on the edge of my seat until further experiments will more closely characterize this gene.
References
Inada T, Winstall E, Tarun SZ Jr, Yates JR 3rd, Schieltz D,
Sachs AB. (2002). One-step affinity purification of the yeast
ribosome and its associated proteins and mRNAs.
RNA.
8:948-58. (Abstract).
Schwikowski B, Uetz P, S Fields. (2000). A network of protein-protein interactions in yeast. Nature Biotechnology. 18: 1257-61. (Abstract).
Uetz P, Giot L, Cagney G, Mansfield TA, Judson RS, Knight JR, Lockshon D,
Narayan V, Srinivasan M, Pochart P, Qureshi-Emili A, Li Y, Godwin B, Conover D,
Kalbfleisch T, Vijayadamodar G, Yang M, Johnston M, Fields S, Rothberg JM. (2000). A comprehensive analysis of protein-protein
interactions in Saccharomyces cerevisiae.
403: 623-7. (Abstract).
This page created by: Gray Lyons ![]()
Return to My
Genomics Homepage
Return to the Genomics Course Homepage
Return to the Davidson
College Homepage