This web page was produced as an assignment for an undergraduate course at Davidson College
Sleepy? Blame your hypocretins.
What is Narcolepsy?
Narcolepsy is a sleep disorder that is estimated to affect 5 in every 10,000 Americans each year. Symptoms of this condition include excessive daytime sleepiness, sudden onset of sleep, and cataplexy. Those who suffer from narcolepsy often experience symptoms beginning in adolescence (Figure 1) (Hungs & Mignot, 2001).
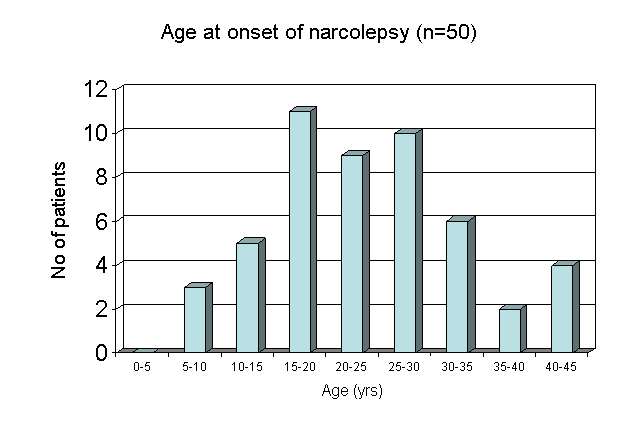
Figure 1. A bar graph showing age of onset of narcolepsy in humans. Most patients from this sample group first noticed symptoms between ages 15 and 20. Permission pending, http://www.narcolepsy.org.uk/narcolepsy/Onset/show_page
Although the cause of narcolepsy is unknown, the recent discovery of hypocretin (also called orexin), a neurotransmitter in the brain, has shed some light on this debilitating and puzzling syndrome. Two genes in dog and human genomes, Hcrtr1 and Hcrtr2, appear responsible for the production of preprohypocretin, the precursor to hypocretin-1 and -2. Three separate mutations have been found in the Hcrtr2 gene in dogs that cause symptoms of narcolepsy. Two mutations are exon skipping alterations that cause the truncation of Hcrtr2 receptors, and the third is a single base pair substitution that changes glutamic acid to lysine, disabling ligand binding. All three mutations result in a complete loss of function (Hungs & Mignot, 2001).
Strong behavioral links exist between human and canine narcolepsy, leading us to the question, is it possible that a mutation in the hypocretin receptor 2 gene, or Hcrtr2, could be responsible for narcolepsy in humans?
Sleep: What is it, and Why do we need it?
Although we view sleep as an escape from the activity of the day, sleep is a fairly active process itself. Throughout a healthy night's sleep, structures in the hypothalamus and brain stem cycle repeatedly through five stages characterized by different brain waves produced during the cycles. The five cycles can be broken up into two main categories: REM (rapid eye movement) and non-REM (slow wave) sleep cycles (Waxman, 2003). This web page will focus on REM and non-REM sleep, but for more information on the specifics of the four sleep cycles, visit http://www.medicinenet.com/sleep/article.htm.
REM sleep, the most crucial part of the sleep cycle (studies have shown that rats deprived of REM sleep have prolonged learning and memory difficulties (Kim et al., 2005), occurs 70 to 90 minutes after falling asleep. REM sleep is initiated by dorsal midbrain and pontine tegmentum neurons. During the REM cycle, sometimes referred to paradoxical sleep, the locus ceruleus releases norepinephrene, a neurotransmitter in the brain that affects food intake, levels of arousal, and mood (Kalat, 2004). The brain then produces delta waves, waves nearly identical to ones observed when a person is awake (Figure 2). Along with a shift in brain waves, often heart rate and blood pressure increase, intense visual imageries (dreams) occur, and muscles can often become temporarily paralyzed. Narcoleptics still experience REM sleep, but instead of entering sleep at stage one and progressing to REM sleep, they often instantly enter REM sleep upon falling asleep. This sudden loss of consciousness and muscle control endangers narcoleptics and sometimes those around them (MedicineNet, 2005).
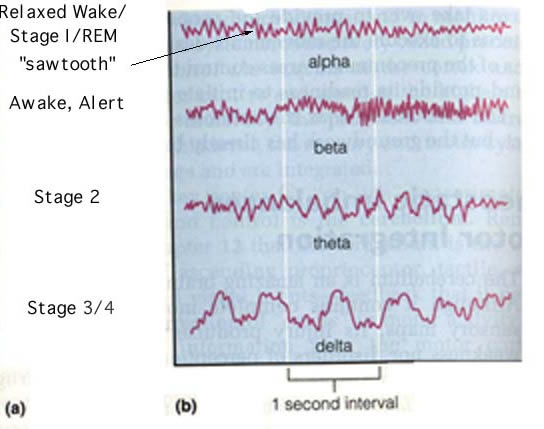 Figure 2. Brain waves collected by an electroencephalograph (EEG). Notice the similarity between the awake, alert waves and REM sleep waves. Permission pending, http://sun.science.wayne.edu/~bio340/StudentPages/Soliman/physweb.html
Figure 2. Brain waves collected by an electroencephalograph (EEG). Notice the similarity between the awake, alert waves and REM sleep waves. Permission pending, http://sun.science.wayne.edu/~bio340/StudentPages/Soliman/physweb.html
As stated previously, the hypothalamus is a structure in the brain that regulates the onset of sleep stages. Within the hypothalamus, hypocretins reside in cells and possibly aid in regulating the neurons involved in REM sleep. Some researchers believe that when Hcrtr2 is stimulated in humans with normal sleep patterns, hypocretins promote wakefulness and repress REM sleep. However, in narcoleptics, a mutation in Hcrtr2 disables this pathway, allowing the sudden onset of REM sleep (http://www.healthgene.com/canine/C110.asp ).
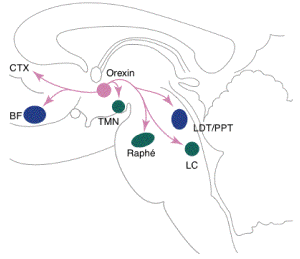
Figure 3. The pathway orexin follows in the brain. BF, basal forebrain cholinergic nuclei; LDT/ PPT, laterodorsal tegmental nuclei/pedunculopontine tegmental nuclei; CR, caudal raphe; PRF, pontine reticular formation; ACh, acetylcholine; PLM, periodic leg movements during sleep; NE, norepinephrine; GLY, glycine; GLU, glutamate. Permission pending, http://webperso.easyconnect.fr/baillement/recherche/flip-flop-switch.html
Treatments?
Some treatments for those suffering from narcolepsy include scheduled naps throughout the day, taking stimulants such as Methylphenidate (Ritalin) and Destroamphetamine (Dexedrine), or sometimes antidepressants are prescribed. Although these medications can improve the mood and mental sharpness of narcoleptics, they can cause dizziness, nausea, and headaches (http://www.umm.edu/patiented/articles/what_treatments_narcolepsy_000098_6.htm .).
Popular Press Articles
A short article appeared in the Health and Medical News section of Australian Broadcasting Corporation's web site in 2000 covering the discovery of Hcrtr2 (for the story, click here ). The headline of the story reads "Human narcolepsy gene found," and the first paragraph claims a cure for narcolepsy will soon arrive. After a one sentence description of narcolepsy, the author, Abbie Thomas, cites Mignot as stating both humans and dogs share a pathway in the brain that breaks down in narcoleptics in both species. Although failing to describe the pathway, or its breakdown, in any detail, Thomas does provide the name of the protein lacking in the cells of narcoleptic humans; it is called hypocretin. Hypocretin is either severely lacking, or not present at all, in narcoleptic patients. Mignot is quoted in the article stating, "We don't know how or why, but it's most likely an autoimmune disease" (Thomas, 2000).
A second article on a sleep disorders web site began its slightly more in-depth coverage of Hcrtr2 by misspelling Emmanuel Mignot's name (Florence Cardinal, the author, forgot the g in Mignot). Cardinal also provides hope for a cure, but states it is a possibility that exists far into the future rather than the present. Cardinal dedicates an entire paragraph to general information about narcolepsy, including symptoms such as cataplexy. Also unlike Thomas, Cardinal attempts to describe the break down of the molecular pathway that is thought to cause narcolepsy, albeit only in a sentence. Cardinal briefly describes current research efforts and directs readers interested in the topic to an article written by Jerome Siegel in the January issue of Scientific American. Cardinal ends her article giving a realistic view of the number of questions still left unanswered about narcolepsy, but does claim the identification of Hcrtr1 and Hcrtr2 is progress in the right direction.
The Scientific Press Addresses the Issue
Two independent labs stumbled upon Hcrtr1 and 2 in 1998. de Lecea and colleagues located preprohypocretin, the precursor to hypocretin-1 and -2, after sequencing H35, a clone from a cDNA library created for locating hypothalamic-specific transcripts (to read the original publication, click here). At the time, de Lecea was searching for novel genes that regulated hypothalamic activity. After finding hypocretin-1 and -2 in the dorsolateral hypothalamus, de Lecea and colleagues performed immunocytochemistry on adult male Wistar rats and found that hypocretin mRNA was primarily expressed in rats three weeks after birth in hypothalamic neurons not previously thought to form their own network. de Lecea suggested hypocretins were novel neurotransmitters. The lab named these transmitters hypocretin-1 and -2 because of their location in the hypothalamus and structural similarity to secretin, a hormone in the gut (de Lecea et al., 1998). de Lecea and colleagues mapped the mouse Hcrt gene to a region within chromosome 11 that is syntenic to human chromosome 17 (17q21-q24). This fact, coupled with the perfect preservation of Hcrtr1 and 2 between rats and mice led de Lecea to believe his discovered neurotransmitter played an important functional role. (de Lecea et al., 1998).
That same year, Sakurai and colleagues characterized hypocretin-1 and -2 and receptors OX1R and OX2R while researching ligands for orphan G protein-coupled receptors (G-protein-coupled cell surface receptors (GPCRs) without known ligands). Using immunohistochemical analysis, Sakurai and colleagues located these peptides and their receptors in intrahypothalamic regions. Sakurai and colleagues observed precursors to these peptides being upregulated during fasting and administered during feeding, leading them to the conclusion Orexin-A and -B were involved in feeding regulation (orexin comes from the Greek word orexis, meaning appetite). Hypocretin-containing cell bodies are located in the lateral hypothalamus, an area involved in food intake, along with many other functions including maintaining homeostasis, cardiovascular and autonomic functions, and of course, regulating sleep. However, axons from these hypocretin-containing cell bodies projected to the limbic system, thalamus, and brainstem, suggesting to Sakurai and colleagues that Hcrtr-1 and -2 are involved in more than food regulation However, Sakurai and colleagues held onto their original theory that these hypocretins were involved in feeding for a good while. They administered hypocretin into the lateral ventricles of male rats. The rats consumed readily available food at a rate directly proportionate to the amount of hypocretin-1 they had received, an effect that continued for four hours after the initial injection of hypocretin-1. The same behavior was observed using hypocretin-2. These test results lead Sakurai and colleagues to conclude their initial publication by looking to hypocretins to aid in the creation of treatments for homeostatic irregularities such as obesity and diabetes (Sakurai et al., 1998).
Popular Press versus Scientific Press
In order to make reading as easy as possible for their audiences, the authors of the popular press articles left out some of the most interesting facts about Hcrtr-1 and -2. Neither popular press article described the research process that lead scientists to current hypotheses. The articles fail to mention anything about finding hypocretin-1 and -2 in the hypothalamus and the original hypothesis that hypocretins were involved in food regulation. Both articles mentioned molecular pathways, but even simple scientific terms such as receptor, mutation, or loss of function were absent from the popular press articles. Both of the popular press articles also ignored important differences between Hcrtr1 and Hcrtr2. Hcrtr2 knockout mice suffer from cataplexy and a disrupted sleep/wake cycles while Hcrtr1 knockout mice exhibit few behavioral changes. It is possible, then, that Hcrtr2 may be critical in the appearance of the narcoleptic phenotype and Hcrtr1 acts as a modifying gene (as cited in Hungs & Mignot, 2001). It is unfortunate that such an interesting detail was not shared with the general public.
The most misleading aspect of these two popular press articles is the de-emphasis on our knowledge about narcolepsy in model organisms and the emphasis on human narcolepsy, about which we know much less. Most research in narcolepsy has been done with dogs, rats, and mice. Although the amino acid sequence of Hcrtr-1 is identical to that in these four mammals, the amino acid sequence for Hctrt-2 in humans differs by an amino acid (Hungs et al, 2001). These genes are still very similar between dogs and humans, but this difference could certainly alter responses to treatments.
The scientific articles were not free from problems, either. Since two separate labs discovered the same novel neurotransmitter, two different names were given to the same biological entity. Although many years have passed since the initial discovery, a uniform set of terms has not yet been reached. The genes, neurotransmitters, and receptors involved with hypocretins have at least two, and sometimes it seems, three sets of names. This makes understanding the exact role and localization of these entities extremely difficult. It seems doubtful, however, that researchers in the field will soon take the time to agree upon names for these peptides.
The popular press articles presented above were not incorrect in the information they included, but the authors did a disservice by omitting information about the trail that led us to what we know today and some of the more interesting details, even if they do cloud the picture. If the media portrays scientific research as cut and dry, they create unrealistic expectations for the general public. Without understanding the hurtles and often unexpected data collected by scientists, the voting public will not understand the need for extensive funding for the sciences. The popular press should not refrain from including the details that muddle the water, for it is those facts that often provide the fun of science.
Works Cited
Cardinal, Florence. 2005. Narcolepsy Gene Breakthrough. <http://sleepdisorders.about.com/cs/narcolepsygenes/a/narco_gene.htm>. Accessed 2005 Sept 4.
HealthGene. Canine Narcolepsy. <http://www.healthgene.com/canine/C110.asp>. Accessed 2005 Sept 8.
Hungs, M, Fan, J, Lin, L, Lin, X, Maki, R, Mignot, E. 2001. Identification and functional analysis of mutations in the hypocretin (orexin) genes of narcoleptic canines. <http://www.genome.org/cgi/content/full/11/4/531>. Accessed 2005 Sept 4.
Hungs, M & Mignot, E. 2001. Hypocretin/orexin, sleep and narcolepsy. BioEssays 23: 397-408.
Kalat, J. Biological Psychology. Toronto, Canada: Wadsworth. p.600.
Kim, E, Mahmoud, G, Grover, L. 2005. REM sleep deprivation inhibits LTP in vivo in area CA1 of rat hippocampus. Neuroscience Letters 388: 163-167.
de Lecea, L, Kilduff, T, Peyron, C, Gao, X, Foye, P, Danielson, P, Fukuhara C, et al. 1998. The hypocretins: Hypothalamus-specifc peptides with neuroexcitatory activity. Proceedings of the National Academy of Sciences of the United States of America 95: 322-327.
MedicineNet. 2004. What are sleep disorders? <http://www.medicinenet.com/sleep/article.htm>. Accessed 2005 Sept 8.
Sakurai, T, Amemiya, A, Ishii, M, Chemelli, R, Tanaka, H, Williams, S, Richardson, J, et al. 1998. Orexins and orexin receptors: A family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92: 573-585.
Thomas, Abbie. 2000 Aug 30. Human narcolepsy gene found. <http://www.abc.net.au/science/news/stories/s168811.htm>. Accessed 2005 Sept 4.
[UKAN] Narcolepsy Association UK. 2005. Age at onset of narcolepsy. <http://www.narcolepsy.org.uk/narcolepsy/Onset/show_page>. Accessed 2005 Sept 12.
Waxman, S. 2003. Clinical Neuroanatomy. New York, NY: Lange Medical Books/McGraw-Hill. p.234-236.
Questions or comments? E-mail Caitlin Kiley at cakiley@davidson.edu
Return to Davidson College Biology Home Page
Return to Genomics Home Page
Return to Caitlin Kiley's Home Page
Return to Davidson College Home Page