This web page was produced as an assignment for an undergraduate course at Davidson College.
Introduction |
Analysis of Figures |
Conclusion and References |
|---|
Hartman et al. focused their studies on the juxtaposition of bacterial populations within the GI tract of small bowel transplant (SBT) patients with that of “normal” human microbial intestinal populations. Bacterial populations within intestines, often known as gut flora, provide necessary functions, such as aiding in the digestion of food and providing a pathogenic protection barrier (Hartman et al., 2009). The possibility for a shift in gut flora populations within individuals who recently received small bowl transplants was first explored by Hartman et al. in this paper: “Human gut microbiome adopts an alternative state following small bowel transplantation.”
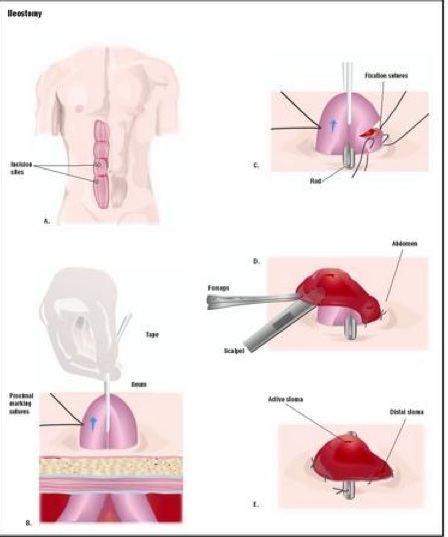
Hartman et al. capitalized on the surgical creation of an ileostomy (opening) as part of the SBT to gain access and gather samples of microbial communities within the gut from 17 individuals. Below is a diagram illustrating a surgical ileostomy, where the intestine is threaded through the epidermis for easy access and sampling.
Answers.com: Ileostomy, 2009
The researchers used quantitative PCR (qPCR), instead of 16s rDNA sequencing techniques, to determine the proportions of the four different bacterial species in the gut samples, because low bacterial diversity was found in initial 16s rDNA sequencing results indicating that qPCR was a feasible option for quantitative bacterial population assays. Quantitative PCR, also known as real time PCR, utilizes the same mechanisms behind PCR but relies on fluorescent tags (green in this paper) that attach to all dsDNA in the PCR amplified mixture, which allows for quantification of DNA along with amplification (Wikipedia: Real-time polymerase chain reaction, 2009). It is also less expensive and more efficient than 16s rDNA sequencing techniques, and it was therefore used by Hartman et al. to quantify proportions of bacterial diversity collected from the ileostomy of SBT patients.
Initial findings suggested that gut flora populations in SBT participants (with ileostomies) were mainly comprised of facultative anaerobes; whereas “normal” microbial populations consist of anaerobes, the inverse of the SBT patients. After more conditioned studies where SBT patients with closed ileostomies were studied (samples were collected with temporary illeostomies close to retrieval of intestinal samples), the researchers determined that the inversion of gut flora might be due to the ileostomy and not the SBT. In fact, data indicated that once the opening in the intestine was closed the gut flora tended to revert back to a dominance of anaerboic species. Hartman et al. hypothesizes that the diffusion of oxygen may account for the change in bacterial dominance among intestinal microbial populations. The paper further assesses the change in bacterial population samples in both pre and post ileostomy closure participants by exploring metabolic differences in the two sample groups. They determined by mass spectrometry that certain enzymes involved in the Krebs cycle (which requires oxygen) were “enriched” in the pre-closure samples. This data further supported the presence of oxygen in pre-closure samples as does the existence of increased facultative bacteria populations within the gut as compared to “normal” population proportions. All in all, the researchers determined that SBT participants, with an open ileostomy, had gut flora with more facultative bacteria than anaerobic bacteria and had enriched metabolites of the Krebs cycle. These basic distinctions led to the hypothesis that the opening (ileostomy) itself allowed possible diffusion of oxygen, which caused a shift in the microbial community proportions inside the gut from a “normal” dominant anaerobic to a facultative population. Although the shift did occur the researchers did not note any complications from the change and even indicated that reversal of such a shift was possible and often achievable.