Introduction
The growing field of synthetic biology attempts to construct biological systems that mimic some of the characteristics of computers and other electronics devices. These machines typically feature some sort of counting system, and in a recent article published in Science, Friedland et al. (2009) describe two kinds of genetics “devices” that are able to count cellular events.
The first counter developed by the team was a riboregulated transcriptional cascade (RTC) counter. The ultimate purpose of this module was to count pulses of the molecule arabinose (often abbreviated “Ara” within the paper) as it was introduced to E. coli cells containing the genetic counter system on a transformed plasmid. The first of these RTC counters to be developed was designed to report after two arabinose pulses had been registered by the cell, and this system was engineered around two linked transcriptional cascades driven by different promoters (the entire system as described below is shown in Fig. 1A). The first transcriptional unit consisted of a constitutively activated (always activated) promoter (PLtet0-1) which drove the transcription of T7 RNA polymerase (RNAP). However, this particular T7 RNAP sequence was linked to a cis-repressor sequence (cr) that preceded the ribosome-binding site (RBS). This design resulted in constant transcription of a version of the T7 RNAP mRNA which was unable to undergo translation because the cr sequence prevented binding of ribosomes to the RBS, a necessary step for the initiation of translation. This led to the accumulation of T7RNAP mRNA in the cells (depicted at the “0” mark in the lower portion of Fig. 1A). However, RBS repression by the cr sequence can be relieved through the activity of a transactivating, noncoding RNA (taRNA) which was designed to be driven by an arabinose promoter (PBAD). Therefore, introduction of arabinose to the cell would result in taRNA expression which in turn would relieve RBS repression of the T7RNAP mRNA, leading to translation of the T7RNAP protein (depicted at the “1” mark in the lower portion of Fig. 1A). The resulting protein then binds to the T7 promoter which drives transcription of green fluorescent protein (GFP) mRNA. However, as with the T7 RNAP mRNA, the GFP mRNA is preceded by a cr sequence whose repression activity is only relieved through taRNA expression driven by a second pulse of arabinose. Thus, the researchers effectively developed a counter that reports via GFP protein expression (which can be easily detected through fluorescence techniques) after being exposed to two pulses of arabinose (shown at the “2” mark in the lower portion of Fig. 1A).
Figure 1
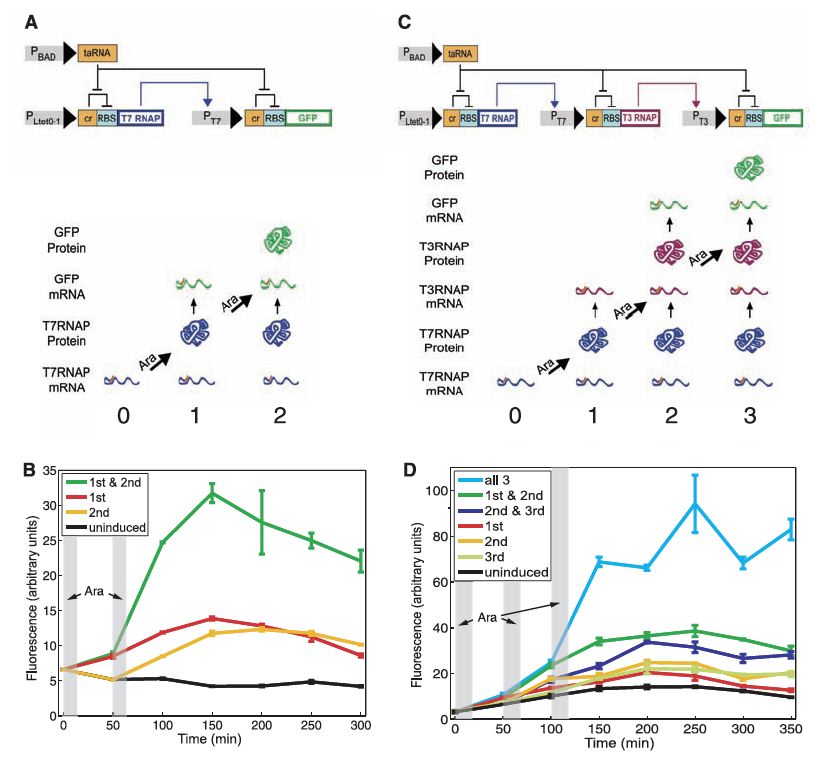
Friedland and colleagues were also able to build a more complex RTC counter that registered three arabinose pulses simply by adding another transcriptional node in the cascade that was driven by a unique promoter (the T3 promoter which is bound by T3 RNAP; third transcriptional module depicted in Fig. 1C). The design logic is exactly the same as with the RTC two-counter, but in this case a total of three arabinose pulses are necessary for effective translation of the GFP protein (shown in the lower portion of Fig. 1C).
The researchers validated the operation of both RTC counters through fluorescence measurements taken using flow cytometry (Fig. 1B and 1D correspond to the RTC two-counter and three-counter designs, respectively). In each graph the gray vertical bars represent the duration of arabinose pulses. The plotted lines represent the average of three replicates tested under the different experimental conditions. As expected, each counter only responded with high levels of GFP expression when induced with the necessary number of pulses (green line in Fig. 1B, light blue line in Fig. 1D). The experiment was also run with various combinations of pulses (i.e., none or one of the two pulses for the RTC two-counter system; none, one, or two of the three pulses for the RTC three-counter system). While the counters showed some leakage (i.e., in some cases, low levels of fluorescence were detected with the inappropriate number of pulses likely because of undesired downstream transcription and translation activity), the counters still responded with a clearly distinguishable signal when pulsed the appropriate number of times.
Figure 2
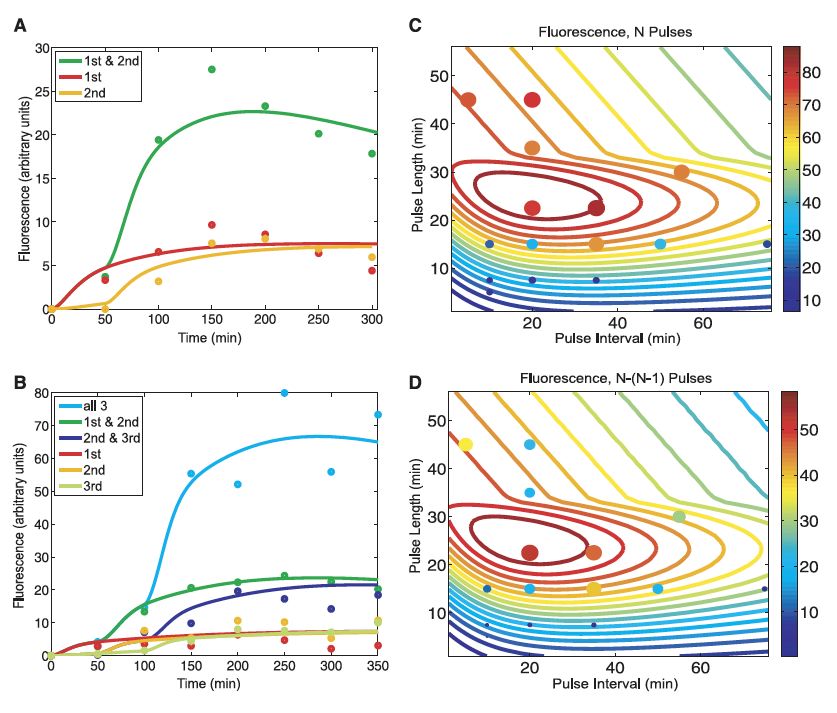
Next, the team sought to develop a mathematical model that would allow them to predict the behavior of their counters under different pulse conditions. They first generated two mathematical models with fitted parameters that matched well with their experiment results. Figure 2A shows the model describing the RTC two-counter system (notice that the colored lines [representing the mathematical models themselves] and plotted dots correspond with the data from Fig. 1B), while Figure 2B shows the model they developed for the three-counter system (this graph’s data match with Fig. 1D). The investigators then used the second of these models to predict how the RTC three-counter system would respond given different arabinose pulse lengths (exposure time of each pulse) and pulse intervals (time between pulses). They communicated their findings in two different ways. The colored contour lines in Figure 2C express the fluorescence level after 3 pulses while Figure 2D shows the fluorescence difference seen between the third and second pulse (remember that Fig. 1D indicates the RTC three-counter may show some fluorescence given only two pulses, thus the authors sought to measure the difference in fluorescence between two and three pulses as that represents the difference between an incorrect and correct number of pulses). In both graphs, warmer colors (yellow/orange/red) represent elevated fluoresence responses. Experimental data gathered using different pulse lengths and intervals (the colored dots on Fig. 1C and 1D) suggest that the model makes accurate predictions of the counter’s behavior, and in general the results from the two scenarios are similar (notice that the size of the colored dots indicates the strength of the fluoresence response in addition to the color). Overall, the modeling results indicate that the optimal fluorescence signal is achieved at pulse lengths between 20-30 minutes with pulse intervals of 10-40 minutes. When either of these parameters is extremely low or high, the counting ability of the construct is compromised (i.e., the fluorescence response is low).
Figure 3
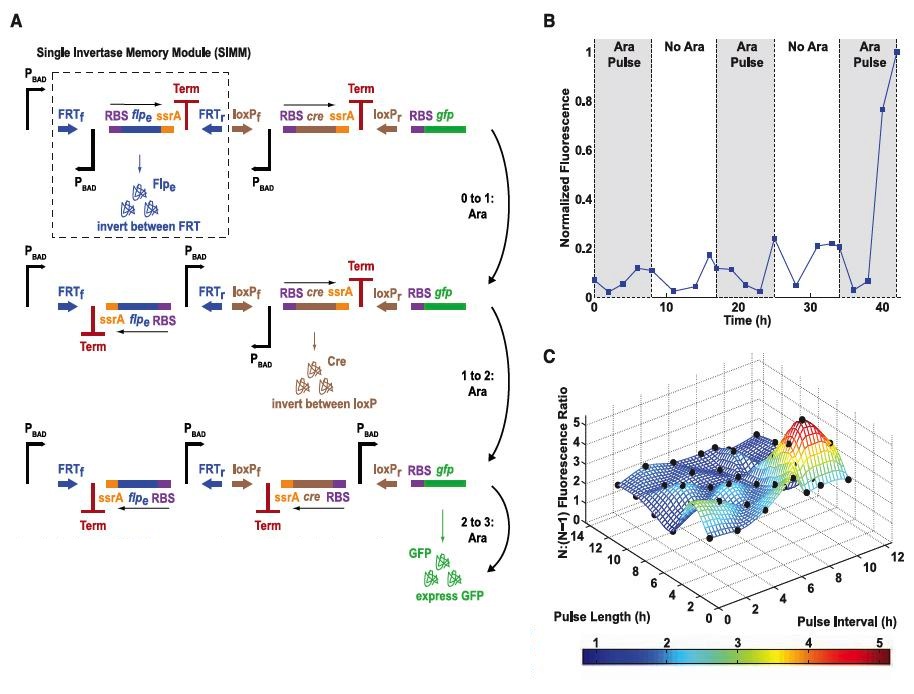
The researchers then constructed a counter they termed the DNA invertase cascade (DIC) counter. As the name suggests, the DIC is based around DNA modules (single invertase memory modules or SIMMs) that are flanked by recognition sites for recombinases which catalyze the inversion of the DNA located between the recognition sites. For example, Cre recombinase is able to invert sequences that lie between loxP sites, while flpe can flip DNA between flpe-recombination target (FRT) sites. In between a given set of recombinase recognition sites the researchers included DNA sequences in the following order: an inverted PBAD promoter, a RBS, a recombinase gene of interest, a ssrA tag (which causes rapid protein degradation), and finally a transcription termination site (all shown within the dashed box in Fig. 3A). The first SIMM was preceded by a single PBAD promoter. When activated by an initial pulse of arabinose, the recombinase within the first SIMM will be transcribed and translated (but nothing further downstream should be transcribed because of the transcription termination site). The recombinase protein that is expressed will catalyze the inversion of its own DNA sequence within the SIMM (shown as the step labeled “0 to 1: Ara” in Fig. 3A).
This inversion has two main effects. First, it renders the DNA within the SIMM fixed in a configuration which makes further transcription of that DNA sequence impossible because the sequence is now inverted with respect to its upstream promoter. Second, the inversion leaves the PBAD promoter contained within the SIMM itself in the proper configuration to promote the expression of the next SIMM downstream. In effect, an arabinose pulse deactivates one SIMM and sets up the next downstream SIMM to be ready for activation. A DIC three-counter system can therefore be constructed by coupling two independent SIMMs with a final GFP sequence to serve as a reporter (Fig. 3A). The first two arabinose pulses should result in inversion of the two SIMMs while the final pulse leads to the expression of GFP. The researchers validated their counter experimentally, and found that three pulses of arabinose led to a clear fluorescence signal as a result of GFP expression (Fig. 3B). Finally, in an experiment analogous to that seen in Figure 2D, the authors varied pulse length and interval, showing that the DIC three-counter effectively showed a difference in fluorescence level between the second and third arabinose pulse over pulse lengths and intervals ranging from 2 to 12 hours. Note that in this figure the ratio of fluorescence between the third and second arabinose pulses are represented both by the color and height (vertical axis) of the graph.
Figure 4
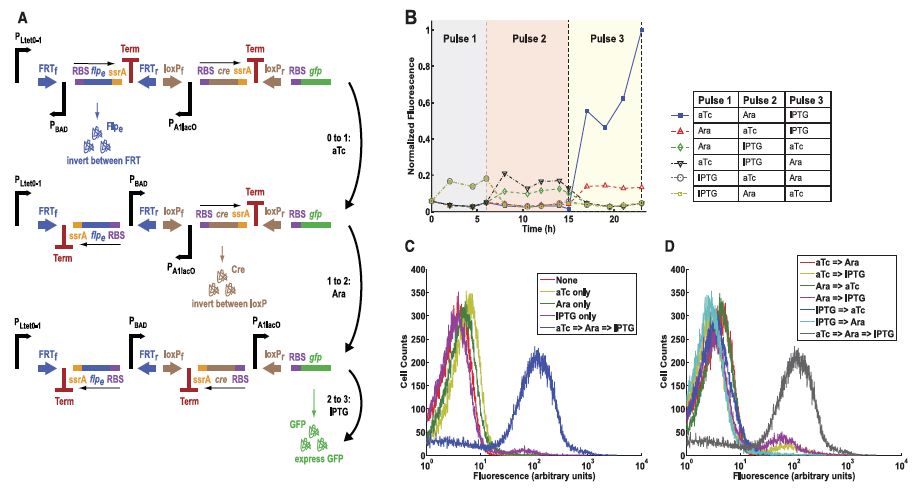
In a twist on the DIC, the investigators designed a construct whose SIMMs each contained a different inducible promoter (Fig. 4A). Reading from left to right, the construct contained the promoters PLtet0-1, PBAD, and PA1lacO, and should therefore be activated only through exposure to anhydrotetracycline (aTc), arabinose, and isopropyl β-D-1-thiogalactopyranoside (IPTG), in that specific order. Indeed, the data show that the only combination of three pulses to show elevated fluorescence activity was aTc-Ara-IPTG (Fig. 4B). Flow cytometry data confirmed that no one pulse alone (Fig. 4C) or combination of two pulses (Fig. 4D) showed the fluorescence output of the aTc-Ara-IPTG pulse sequence. This counter design therefore reports effectively only after a specific series of three cellular events.
Conclusions
Overall, I found this paper to be intriguing because of its approach to biological systems. Rather than seeking to understand the function of genes, proteins, etc. in their native state, which is typical of most biologists, Friedland et al. used their knowledge of biology to engineer unique molecular tools. We spend so much time in biology classes deciphering how natural systems operate, it was interesting to see an approach that moved information flow in completely the opposite direction (i.e., make the system take on properties we desire rather than discovering the properties of the system). This research shows that it is indeed possible to use genetic materials to design a completely artificial network that then behaves as expected in vivo.
I think the most promising aspect of their work is that it is readily adaptable and expandable. Their designs may be modified through the incorporation of different promoters, recombinases, or reporter genes, and they may also be expanded through construction of longer reporter chains containing more genetic modules. I thought Friedland et al. did a very nice job of illustrating these aspects of their systems through the organization of their paper (i.e., they walk the reader through the RTC two-counter, then expand that design to demonstrate an RTC three-counter, and finally show DIC counters driven by either identical or unique promoters). Because they may be readily modified, I believe these synthetic gene networks will accommodate varied uses both by biologists attempting to study cellular processes in novel ways and by those seeking to apply biological tools to problems outside the discipline.
References
Friedland, AE, Lu, TK, Wang, X, Shi, D, Church, G, and Collins, JJ. 2009. Synthetic gene networks that count. Science 324: 1199-1202.