
http://www.mhmcintyre.us/2012/02/14/under-construction-new-blog-theme-for-attempts-at-honesty/
Gene Assignment 1-Kindness Gene
Gene Assignment 2 - Genes Associated with Habitual Caffeine Consumption

http://www.mhmcintyre.us/2012/02/14/under-construction-new-blog-theme-for-attempts-at-honesty/
This webpage is a summary of what I learned upon reading the article entitled: Evolutionarily Assembled cis-Regulatory Module at a Human Ciliopathy Locus by Lee et al. The researchers of this study explored the JBTS2 locus and focused on two genes, TMEM138 and TMEM216, that are associated with Joubert Syndrome. The authors claim to have identified an example of correlated expression of nonparalogous genes arranged head to tail that produce proteins with related functions. They also claim that the genes were rearranged into the functional gene cluster and were assembled into a cis-regulatory module that is responsible for their similar expression.
Overall, I enjoyed reading the paper and learned a lot about Joubert Syndrome, cilia, and cis-regulatory modules. I like how each experiment in the study rested upon the logic and knowledge uncovered by the results of the last. In addition to producing a well constructed paper, the authors provide detailed supplemental online material that includes time-lapse videos for some of the experiments in the study. The supplemental online material is a nice solution to the problem of limited space when publishing in Science. Any frustration encountered when reading this paper was more due to the constraints of the journal rather than any fault to the authors.
In general, the figures were clear and I liked that a brief description of the methods was included with nearly every figure. I found it difficult to draw conclusions from parts of the figures that were tissue samples but thought it was good that these pictures were often accompanied by a graph to represent the data. More specific comments can be found as we explore the paper figure by figure.
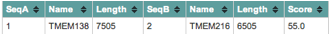
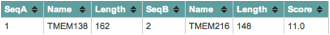
In the study entitled Evolutionarily Assembled cis-Regulatory Module at a Human Ciliopathy Locus, researchers aimed to show the functional relatedness of two nonparalogous genes, TMEM138 and TMEM216. According to ClustalW results, displayed in Figure 1, comparing the sequences of the two genes, TMEM138 and TMEM216 are in fact nonparalogous, as there relatedness score is 55% (number of matches between the two sequences divided by the length of the alignment). Results from another ClustalW, shown in Figure 2, demonstrate that the proteins produced by the two genes also lack homology, based upon a score of 11%. Genes located next to each other, such as TMEM138 and TMEM216, are often coordinately expressed within a cis-regulatory module (CRM) (Lee et al., 2012)(Fig. 3). A CRM is a DNA sequence, located near its target gene, that has transcription factor binding sites that regulate the transcription of a particular gene through activation or repression (Jeziorska et al., 2009). Therefore, CRMs are responsible for the correct temporal and spatial expression of a gene, which is critical for the development of organisms (Jeziorska et al., 2009). The study by Lee et al. is unlike others, as it claims to have established related function between the two nonparalogous genes, which was previously not demonstrated in mammals (Lee et al., 2012). The importance of these genes and their protein products is evident when mutations in either result in Joubert syndrome (JS). Patients with this condition experience developmental delay, intellectual disability, abnormal eye movements, and neonatal breathing difficulties (Brancati et al., 2010). The condition and related ciliopathy disorders are highly identifiable due to a molar tooth sign, which is a midbrain-hindbrain malformation detectable by imaging techniques (Brancati et al., 2010). As of 2010 there were ten genes associated with mutations resulting in JSRD, all of which produced proteins involved in the primary (nonmotile) cilia or the centrosome (Brancati et al., 2010). Primary cilia are involved in cell signaling and are important to development (Singla et al., 2006). Although Joubert syndrome related disorders (JSRD) are rare with an incidence rate ranging between 1/80,000 and 1/100,000 live births (Brancati et al., 2010), this research is important, not just within the context of human disease but as a possibility of detecting a CRM for nonhomologous genes with related functions.
http://www.ebi.ac.uk/Tools/services/web/toolresult.ebi?jobId=clustalw2-I20120426-143032-0364-57657105-oy&tool=clustalw2&analysis=summary
Figure 1. The sequences of TMEM138 (NC_000011.9) and TMEM216 (AC_000143.1) were compared in ClustalW and were shown to not be homologous, with a score of 55% (number of matches between the two sequences divided by the length of the alignment). This figure confirms the statement made by the authors that the genes are not homologues.
http://www.ebi.ac.uk/Tools/services/web/toolresult.ebi?jobId=clustalw2-I20120426-153631-0385-78890596-pg&tool=clustalw2&analysis=summary
Figure 2. The sequences of the protein products of TMEM138 (NP_057548.1) and TMEM216 (NP_001167461.1) were compared in ClustalW and were shown to not be homologous, with a score of 11% (number of matches between the two sequences divided by the length of the alignment). This figure confirms the statement made by the authors that the proteins are not homologues.
http://www.sciencemag.org/content/335/6071/966.full.pdf
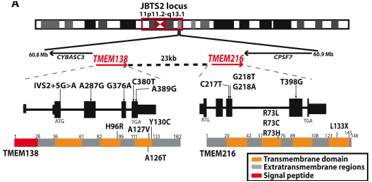
Figure 3. Working from the top of the figure down, this figure displays the locations of the two genes, THEM138 and TMEM216, which are 23 kb apart, facing the same direction, bordered by CYBASC3 on the left and CPSF7 on the right, and located within the JBTS2 locus (11p11.2-11q13.1). The locations of all the missense and splicing mutations for the two genes known to be linked with JS are labled on the transcript and protein. The last part shows that TMEM138 encodes a trispan membrane protein (three transmembrane domains) and TMEM216 encodes a tetraspan membrane protein (four transmembrane domains). Permission granted by Dr. Joseph Gleeson.
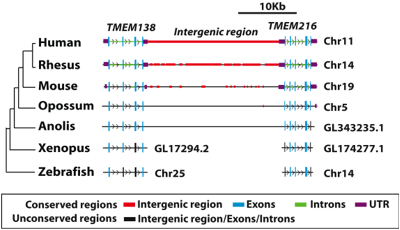
From the authors’ previous work, it was determined that mutations in TMEM216 accounted for half of their patient sample linked to the JBTS2 locus and that mutations in TMEM138 accounted for the other half of the incidents (Lee et al., 2012). This is the foundation for their work and even though it was past research they show the locations of the mutations in the genes known to be linked to JS (Figure 3). The authors explain that the two genes and the proteins they encode are not homologous (which was confirmed by Figures 1 and 2) and that the transmembrane proteins encoded by both are not in the same protein family (Lee et al.,2012). Due to the lack of protein homology, the authors conclude that a gene-duplication did not occur and that the proteins evolved separately from invertebrates (Lee et al., 2012). Figure 4 is a phylogenetic tree and synteny map illustrating the relatedness and conservation of TMEM138 and TMEM216 arranged in a head-to-tail fashion separated by approximately 23 kb of noncoding DNA (Lee et al., 2012). In figure 4, the authors are refering to the conservation of the configuration and not the sequences. The claim that the two genes are joined due to an ancient chromosomal rearrangement at the amphibian-to-reptile evolutionary transition seems likely as the two genes are on separate chromosomes before the transition and on the same chromosome following the transition. Rearrangement is a likely explanation for this observation. Therefore, it is difficult to disagree with that as configuration is the same for the non-amphibian samples indicating that the conservation begins following the transition.
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 4. A phylogenetic tree and synteny map demonstrating the conservation of TMEM138 and TMEM216 in seven different vertebrates. The authors claim that the two genes are joined by the intergenic region as the result of chromosomal rearrangement at the amphibian-to-reptile evolutionary transition that occurred approximately 340 million years ago. Presumably they base this conclusion upon the fact that the xenopus (a genus of aquatic frogs) and zebrafish do not have the conserved configuration of the genes head-to-tail on the same chromosome and connected by a noncoding region that is present in the anolis (a genus of lizards), opossum (a marsupial mammal), mouse (placental mammal), rhesus (a genus of old world monkeys) and humans. The figure provides the chromosome where the genes are located for all of the samples other than anolis and xenopus. The exons of TMEM138 and TMEM216 seem to be conserved in all of the samples, with the least amount of similarity occurring between humans and the xenopus and zebrafish. The introns of TMEM138 seemed to be conserved only between human and rhesus, while the introns of TMEM216 seemed to be conserved between mouse, rhesus, and human. According to the key, the intergenic region (in red) is at least partially conserved in humans, rhesus, mice, and opossum. DNA indicated as conserved means that the region has a > 50% sequence similarity to humans. It is unclear if the arrows indicate anything more than direction. Permission granted by Dr. Joseph Gleeson.
The fact that the two genes are configured together and that mutations in both caused the same phenotype, indicated to the authors that the genes may have correlated expression and related functions due to a CRM (Lee et al., 2012). The authors used a microarray database and in situ hybridization of human embryos to determine if the genes have related expression in certain human tissues (Lee et al., 2012). The authors fail to mention, which microarray database they used to determine the patterns. Analyzing microarrays is a valuable technique for determining patterns of gene expression but can miss the expression of less abundant mRNAs, such as those for transcription factors. The authors concluded from the microarray database that TMEM138 and TMEM216 were co-regulated as the expression values in the major tissues, such as the brain and kidneys, were similar (Lee et al., 2012). A similar conclusion was drawn from the in situ hybridization of the human embryo shown in figure 5, as expression of both genes was similar in the kidneys, cerebellar buds, telencephalon and other tissues after 4-8 weeks of gestation (Lee et al., 2012).
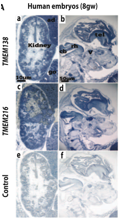
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 5. This figure shows the expression of TMEM138 and TMEM216 in the kidney and brain of human embryos determined by in situ hybridization. The authors conclude that there is similar expression of both genes in both tissue samples, which indicates that the proteins may be co-regulated. However, it is tough to make that conclusion without data, as comparing images of tissue samples is open to the risk of confirmational bias. That being said the panels for TMEM138 and TMEM216 expression show more mRNA than the control, although the TMEM216 kidney panel seems a bit darker than the TMEM138 kidney panel. Even a slight difference would not effect the conclusion that expression of both genes is higher than that of the control. Panels A and B used TMEM138 antisense, panel C and D used TMEM216 antisense, and sense control probes were used in E and F. V= trigeminal; Rh=rhombencephalon; Tel=telencephalon; Cb= cerebellar bud; Go=gonad; Ad= adrenal gland. Permission granted by Dr. Joseph Gleeson.
After determining that TMEM138 and TMEM216 had similar levels of expression in certain tissues, the investigators aimed to determine if the expression patterns was a result of the genes being adjacent. They wisely compared the amount of mRNA from zebrafish, which had TMEM138 and TMEM216 on separate chromosomes, and from mice, which had the configuration of the genes head-to-tail with the 23 kb intergenic region (Lee et al., 2012). In order to detect the amount of mRNA, real-time quantitative polymerase chain reaction (qPCR) was used which is able to detect mRNA that are typically less abundant and provides absolute data instead of patterns (Lee et al., 2012). From the data in Figure 6, the authors concluded that the genes might share regulatory elements, based upon similar expression levels (correlation coefficient of 0.984) in the mouse and dissimilar expression levels in the zebrafish (correlation coefficient of 0.386) (Lee et al., 2012). These results also show that there is a clear difference between the expression of TMEM138 and TMEM216 before and after the amphibian-reptile transition. The results from the qPCR are the most convincing as it is absolute data and is a well-designed experiment.
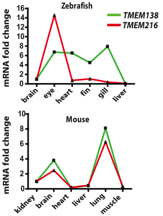
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 6. Real time qPCR was used to determine the mRNA levels of TMEM138 and TMEM216 in zebrafish and mice (representing the before and after, respectively, of the amphibian-reptile transition). For the mice genes (aligned in the configuration described in Figure 4), the expression of the mRNA from both genes is coordinated with a correlation value of 0.984. However when the two genes are not on the same chromosome, such as in the zebrafish, the expression of the mRNA from both genes is not coordinated with a correlation value of 0.386. The figure is lacking error bars and P values indicating the significance between the coorelation of expression in the mice and zebrafish. The y-axis shows the mRNA fold change and the x-axis shows the expression in different tissues. Permission granted by Dr. Joseph Gleeson.
The authors found that regulatory factor X 4, which is a transcription factor for ciliary genes, binds to a regulatory element located in the noncoding intergenic region and is responsible for the coregulation and correlated expression. All information regarding this conclusion is in the supplemental online material.
After concluding that TMEM138 and TMEM216 are coordinately expressed based upon the results of the microarray database analysis, the in situ hybridization of the human embryo, and the qPCR of the mice and zebrafish, the authors aimed to determine if the functions of the proteins are related. This conclusion and the fact that mutations in both genes results in the same phenotype, compelled the researchers to investigate the relatedness of the function in ciliogenesis (the development of cilia) of the proteins encoded by the two genes. Figure 7 shows the formation of short or no cilia in fibroblasts with mutated versions of either TMEM138 or TMEM216 (Lee et al., 2012). From these results and results not shown in the paper from the knockdown of the two genes in IMCD3 cells producing ciliogenesis defects, the authors concluded that both TMEM138 and TMEM216 are required for ciliogenesis (Lee et al., 2012). I have difficulty agreeing with this conclusion based upon the data displayed in Figure 7. It seems that only the mutation in TMEM216 produced less ciliogenesis. Since the mutation in TMEM138 did make the cilia shorter, the protein certainly plays a role in ciliogenesis. However, it does not seem essential as the amount of ciliogenesis was very similar for wildtype for the fibroblasts containing the mutated TMEM138.
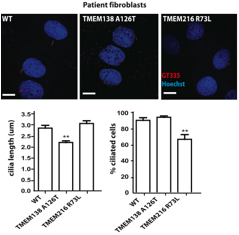
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 7. The top part of the figure contains three panels: the left one is of wild type fibroblasts, the center one is of fibroblasts with mutated TMEM138 (p.A126T), and the right panel is of fibroblasts with mutated TMEM216 (p.R73L). From these panels it is difficult to detect any differences between the three conditions. However, from the graphs at the bottom of the figure it seems that the mutation in TMEM138 produced significantly shorter cilia and that the mutation in TMEM216 may resulted in no change to the length or slight growth of the cilia. The graph with the y-axis of percent of ciliated cells is representing cells that completed ciliogenesis. From the data, it appears that the mutation in TMEM138 resulted in no change or more ciliogenesis, while the mutation in TMEM216 resulted in the occurrence of significantly less ciliogenesis. It is interesting that a mutation in one gene causes shorter cilia and the mutation in the other causes no cilia. *P < 0.05; **P < 0.01. Error bars indicate SEM. Permission granted by Dr. Joseph Gleeson.
In Figure 8, comparison of the locations of the immunostained TMEM138 and the ciliary axoneme and transition zone, marked by Arl13b, showed that both TMEM138 and TMEM216 were present at the base of the cilia (g-tubulin)(Lee et al., 2012).
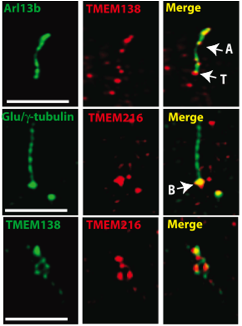
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 8. The top row of panels are high-resolution images of TMEM138 immunostaining. which show that TMEM138 (red) went to the ciliary axonemes and transition zone when the image was merged with another image containing Arl13b (green), which is a marker for the ciliary axonemes and the transition zone. In the middle row of panels, immunostaining of TMEM216 (red) overlayed on an image containing g-tubulin (green), showed that the protein went to the basal bodies. In the bottom row of panels, both proteins were immunostained (TMEM138 is green and TMEM216 is red) and overlayed on images of each other to show that both vesicles go to the base of cilia. Antibodies were used to immunostain Arl13b (cilia), tubulin (cilia plus centrosome) and the TMEM proteins. The arrow with an A indicates ciliary axoneme, an arrow with a T indicates the transition zone, and the arrow with the B indicates the basal body. Permission granted by Dr. Joseph Gleeson.
TMEM216 was found to go near post-Golgi vesicles along microtubules and near the Golgi apparatus around the base of cilium, while TMEM138 went to neighboring but not overlapping distinct vesicles (Lee et al., 2012). From this observation the authors conclude that the two proteins are involved in marking vesicles that are headed for the base of cilium, which is integral to the development of cilia (Lee et al., 2012). More experimentation that is not represented in the paper confirmed that the proteins mark two different but related vesicles of different cilia-targeted proteins (Lee et al., 2012).
After describing the roles of TMEM216 and TMEM138 in regards to vesicles, the authors investigated if one vesicle required the presence of the other in order to properly function. They report that TMEM216 vesicular movement was not changed when TMEM138 was knocked down using small interfering RNA (siRNA) but that interfering with TMEM216 expression resulted in a change in the movement of TMEM138 vesicles (Lee et al., 2012). From this result the authors conclude that the presence of TMEM216 is necessary for the correct transportation of TMEM138 (Lee et al., 2012).
Convinced that there was a functional dependence between the two proteins, the authors aimed to determine the mechanism behind the relatedness. In order to accomplish this task, tethering proteins were used to link two different vesicular samples. TRAPPII was used to join TMEM138 and TMEM216 vesicles together for proper ciliary assembly, as shown in Figure 9 (Lee et al., 2012).
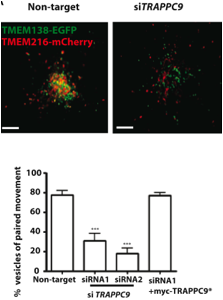
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 9. This figure displays the functional relatedness of TMEM138 and TMEM216. The non-target panel is live-cell imaging of the vesicles from both genes joined together by TRAPPII, with green indicating the vesicles from TMEM138 and red indicating the vesicles from TMEM216. In the siTRAPPCP panel is live-cell imaging when TRAPPCP was knocked-down which caused the vesicles to seperate. The graph shows that myc-TRAPPC9* is able to rescue the tethering of the two vesicles to the amount of tethering seen in the control and that siRNA1 and siRNA2 are able to stop the joining of the vesicles. The y-axis indicates the percent of vesicles tethered in the various conditions. P < 0.001 and error bars indicate SEM. Permission granted by Dr. Joseph Gleeson.
Another interesting result of the study was that coordinated vesicular movement was found in zebrafish even though the genes are not on the same chromosome. This result could indicate that related function may have evolved before related regulation (Lee et al., 2012). The conserved genetic function in zebrafish was explored further through the comparison of morpholino knockdown of both genes (Lee et al., 2012). As shown in Figure 10, knocking down either TMEM138 or TMEM216 resulted in a pericardial effusion (fluid around the heart), a curved or kinked tail, and issues during gastrulation (Lee et al., 2012). The TMEM216 knockdown demonstrated hydrocephalic brains and noticeable abnormalies of the heart axis (Lee et al., 2012). From the information in Figure 10, the authors conclude that the functions of the two proteins were not completely related before the formation of the configuration, as the mutations in the zebrafish caused different phenotypes (Lee et al., 2012).
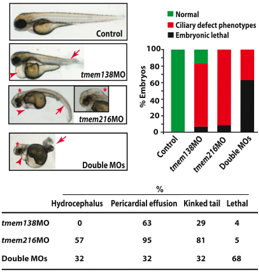
http://www.sciencemag.org/content/335/6071/966.full.pdf
Figure 10. Translation-blocking antisense morpholinos were used to produce zebrafish lacking TMEM138, TMEM216, or both proteins. When the zebrafish were missing one of the proteins, they developed standard ciliary phenotypes. However, in addition to the standard phenotype, the zebrafish without TMEM216 had hydrocephalic brains and severe abnormalies of the heart axis. On the pictures of the zebrafish, the arrowheads indicate heart edema and the arrows indicate curved or kinked tails. Both the graph and table show data for the observations made of the zebrafish. It is clear from all depictions of the data that lacking either protein results in different phenotypes. This is extremely evident in the hydrocephalus column of the table as 0% of the zebrafish lacking TMEM138 had the condition, while 57% of the zebrafish lacking TMEM216 had the condition. The main point of the figure was to show that in zebrafish (before TMEM138 and TMEM216 were aligned head to tail) mutations in either gene led to distinct phenotypes. From this the authors concluded that the function of the proteins were not coordinated and that coordination occurred once the genes recombined into their current configuration. Permission granted by Dr. Joseph Gleeson.
Brancati, F., Dallapiccola, B., and Valente, E.M. 2010. Joubert Syndrome and related disorders. Orphanet Journal of Rare Diseases 5: 1-10.
Jeziorska, D.M., Jordan, K.W., and Vance, K.W. 2009. A systems biology approach to understanding cis-regulatory module function. Seminars in Cell and Developmental Biology 20: 856-862.
Lee, J.H., Silhavy, J.L., Lee, J.E., Al-Gazali, L., Thomas, S., Davis, E.E., Bielas, S.L., Hill, K.J., Iannicelli, M., Brancati, F., Gabriel, S.B., Russ, C., Logan, C.V., Sharif, S.M., Bennett, C.P., Abe, M., Hildebrandt, F., Diplas, B.H., Attié-Bitach, T., Katsanis, N., Rajab, A., Koul, R., Sztriha, L., Waters, E.R., Ferro-Novick, S., Woods, C.G., Johnson, C.A., Valente, E.M., Zaki, M.S., and Gleeson, J.G. 2012. Evolutionarily assembled cis-regulatory module at a human ciliopathy locus. Science 335: 966-969.
Singla, V., and Reiter, J.F. 2006. The primary cilium as the cell’s antenna: Signaling at a sensory organelle. Science 313: 629-633.
Kristopher Hendershot
Biology Major, Class of 2012
Email: krhendershot@davidson.edu