This web page was produced as an assignment for an undergraduate course at Davidson College
Summary
Analysis
Figure 1
Figure 2
Figure 3
Figure 4
This paper sought to identify FDA approved drugs that may have the ability to treat Alzheimer’s disease. To do this, they used a computer modeling approach that filtered drugs based on biological function and predicted their ability to therapeutically change the β amyloid formation pathway. The computer model predicted that proton pump inhibitors, drugs commonly used to treat excess stomach acid production, may modify the formation of β amyloid formation. To test this, they treated cells with lansoprazole, a PPI, and measured amyloid formation through a variety of techniques. They determined that formation of Aβ2-42, Aβ37 and Aβ2-40 increased and Aβ38 decreased.
My main complaint of this paper is the ambiguity with which the authors describe their findings. In the intro they describe how amyloid beta production is linked to Alzheimer’s, stating that these peptides accumulate in the brain tissue causing inflammation and damage. They state, “Aβ42 has been suggested to be the main pathological species in AD pathogenesis”, clearly linking the formation of beta amyloid fibrils with Alzheimer’s disease. Through out the narration of the paper they make it seem as though they want to use lansoprazole as a therapy for Alzheimer’s, even though it increases the fibril formation, therefore exacerbating the disease. In their conclusions section they state, “It would be interesting to perform epidemiologic studies to investigate whether the long-term use of PPI’s could have any detrimental impact on AD”. This wording is very ambiguous and it is hard to determine whether “detrimental impact on AD” means it is detrimental to the disease, implying a therapeutic benefit to the patient, or whether it is detrimental to the patient in furthering the progression of Alzheimer’s. They close the paper saying, “These results can serve as a catalyst for further studies in order to evaluate whether the treatment with PPI’s may have an impact on AD pathology”. The word treatment is misleading and makes it seem like they want to improve the clinical outcome of patients with Alzheimer’s as opposed to treatment in the protocol sense of administer PPI’s. In addition, evaluating the impact on AD pathology is very ambiguous and does not clearly convey whether PPI’s make AD worse or better.
I understand that the intention of this paper was to find a drug to reduce the effects of Alzheimer’s and that PPI’s do the opposite by increasing amyloid beta production, but the authors need to accurately represent the implications of their findings. It would be much easier to understand as a reader if they phrased their results as PPI’s increase amyloid β formation, so we would like to study their effects to learn more about the mechanisms of AD pathology. But they should not ambiguously describe the clinical relevance to make it seem as though it may still be possible to use it as a therapy.
Another aspect of the paper that bothered me was the lack of contextual background that they give for some of their methods and in their introduction. They say that PS1 was transfected into their cell line, but do not explain during the course of the paper what this protein does or why it is important to modeling AD. Furthermore, in their introduction they do not do a good job of explaining the pathology of AD and the role of amyloid fibril formation or differentiating between the different fibrils. They mention that Aβ40 and 42 are pathological in one sentence but do not describe what differentiates them from the other peptides, whether different peptides have different effects, nor how the peptides are linked to the disease, giving the reader a poor background to use to interpret their results.
Finally, there are many grammatical errors throughout the paper and a typo in one of the figure legends. If there was this little attention to detail in the publishing process, it makes me question the attention to detail paid during the actual experiments.

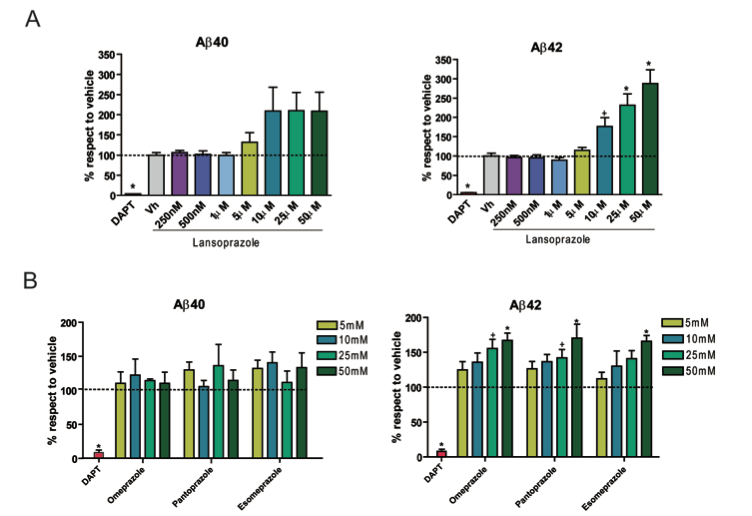
This figure illustrates the effect of lansoprazole and three similar proton pump inhibitors on amyloid formation. The experiments were carried out in cells that were modified to behave like Alzheimer’s disease cells. The extent of fibril formation was determined by an ELISA. DAPT is the positive control that inhibits β amyloid formation, Vh is a the vehicle without the drug. Panel A shows that lansoprazole significantly increases the amount of Aβ42 and also increases the amount of Aβ40, but not significantly.
Similarly, the other PPI’s increased Aβ42 significantly, but not Aβ40, as shown in panel B. In addition, all of these compounds exhibited a dose dependent influence, where the highest concentration had the largest increase in amyloid formation. It would be nice if the authors had explained why they chose that particular cell line and how the transfections make it behave like diseased brain cells.
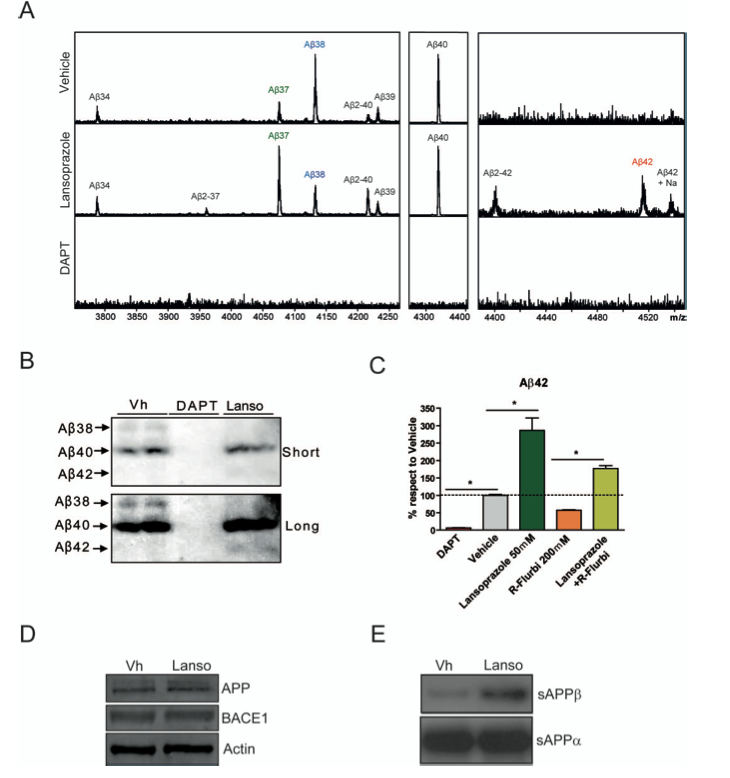
Next, they wanted to quantify which types of fibrils were forming in the vehicle cells versus the lansoprazole treated cells. They also wanted to elucidate the mechanism causing the increase in fibril formation. To determine the peptide profile, they used a mass spectrometer to analyze peptide identity and relative abundance, panel A. They concluded that Aβ37, Aβ2-40, Aβ2-42 and Aβ-42 production increased in the lansoprazole treated cells when compared to the vehicle, and Aβ38 production decreased.
They confirmed these findings by western blot analysis, panel B. They imaged the blots with long and short exposure to help visualize the bands. The Aβ38 band is visible in the vehicle lane with long exposure and does not appear with lansoprazole, the Aβ40 band seems to be the same intensity for both vehicle and lansoprazole, and the Aβ42 band is faintly visible with long exposure when the cells were treated with lansoprazole. These results confirm those found by the mass spectrometer. It would have been nice if they had quantified the intensity of the bands because some of them are hard to see.
Due to the increased presence of Aβ42 and decreased presence of Aβ38, the researchers posited that lansoprazole could modulate the activity of γ- secretase, an enzyme that processes the amyloid precursor. This type of molecule is called at GSM (gamma secretase modulator) and because lansoprazole changes γ- secretase in the opposite manner to produce Aβ42, it is called an iGSM. The effect of lansoprazole and a standard GSM independently and together were monitored by an ELISA, panel C. When combined with the GSM, lansoprazole was able to mitigate its effects and significantly increase the amount of Aβ42 produced. In their interpretation of this figure they say that PPI’s could be given in combination with a GSM to elevate the Aβ42 levels, but this makes no sense to me since you don’t want to make amyloid fibrils in the first place.
In panel D, the researchers wanted to determine what lansoprazole is changing to cause the increase in amyloid production, is it the amount of protein present or the activity of the enzymes. The western blot shows that there is not a difference in APP (the substrate) or BACE-1 (the enzyme that cleaves it) between vehicle and lansoprazole. Panel E shows that the amount of sAPPβ increased from vehicle to lansoprazole, suggesting that the BACE-1 enzyme was more activated in the presence of lansoprazole. In panel E there is no positive control, unlike D to show that equal amounts of protein were loaded.
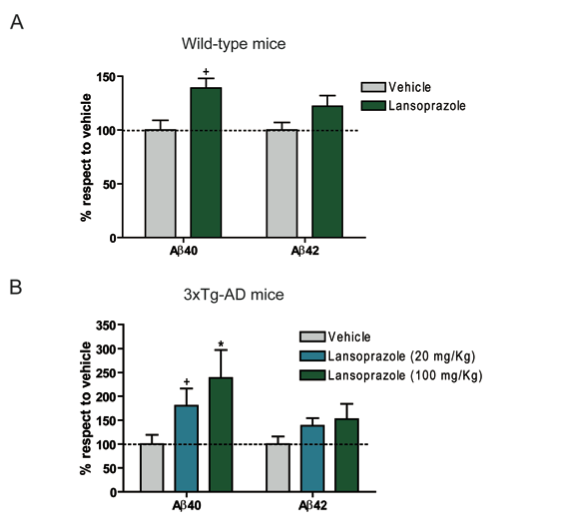
This figure explores an in vivo model of the cell culture analysis that they had conducted until now. Mice were treated with lansoprazole for 5 days in varying doses, then the soluble Aβ40 and 42 peptides were analyzed by ELISA. The wild-type mice showed a slight increase in Aβ40 and 42 production, but it was not significant. The transgenic mice to model Alzheimer’s disease did show a significant, dose dependent increase in Aβ40 production, but not a significant increase in Aβ42 production. These results suggest that the in vitro model translates to the in vivo model and that PPI’s do in fact increase β amyloid formation.
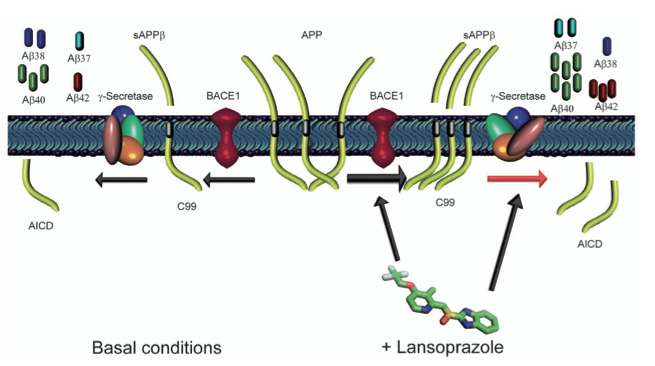
This figure illustrates a schematic of their proposed mechanism for lansoprazole’s effect on amyloid formation. The center of the schematic shows APP, the precursor protein that needs to be cleaved to form amyloid β peptides. The left side of the diagram shows the normal pathway through which these peptides are made producing a variety of peptides and the ratio at which these peptides occur normally. The right side of the diagram shows their proposed mechanism where BACE-1 is more active, producing more of the soluble sAPPβ product. They also propose that lansoprazole changes the normal activity of γ-secretase, causing the altered ration of peptides. The authors use “could” in their figure legend, making it clear that this is speculative. I think that these conclusions could be extrapolated from the data, but I don’t think that the two western blots are sufficient data to draw these conclusions. It would have been nice if that looked at the kinematics of BACE-1 by incubating it with or without lansoprazole and assessed the relative amounts of sAPPβ produced. I also don’t think that the correlation of the change in ratios of the different peptides is enough to link to the causation that lansoprazole is an iGSM.
Badiola, Nahuai, et. al. 2013. The Proton-Pump Inhibitor Lansoprazole Enhances Amyloid Beta Production. PLOS One. 8:1-8. Accessed 23 April 2013. <http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0058837>