This web page was produced as an assignment for an undergraduate course at Davidson College.
Gene duplication can
impart fragility, not robustness, in the yeast protein interaction
network

Sacccharoyces cerevisiae yeast cells. Figure reproduced from Angelica, 2011.
Summary:
Opinion:
The paper
was well-written, with convincing evidence, clear presentation of data,
and interesting applications of simple methods. I appreciated the
inclusion of explanatory figures; a diagram of the processes was much
easier to understand than a written account. However, they often
presented explanatory figures at the expense of data. Many large
conclusions cited supplementary figures, included all of the paralogous
heteromers co-function and human cell line data. While the explanatory
diagrams made the paper more convenient to read, I would have
appreciated seeing more data to support the conclusions in the diagrams.
A few figures had confusing legends or misleading
notes about significance, but they did not hugely distract from the data
presentation. For example, the legend in Figure 3C uses a single color
for the symbol representing nonsignificance, the same color used for
independent pairs. They also only give one data point for independent
paralog pairs, and the use of color understates how few of the dependent
pairs actually show significant effect. Their conclusion is that
independent paralog pairs do not show the same effect as dependent
pairs, but this point would have been better supported if they had
graphed data for both independent and dependent pairs, and then
indicated that more of the dependent pairs showed a significant effect.
I would suggest further research into which specific
proteins are more likely to be compensatory or dependent, beyond
ancestral considerations. Are certain gene ontology classes more likely
to exhibit compensation? Are there environmental factors that influence
whether compensation or dependency occurs?
Figure
Summaries:
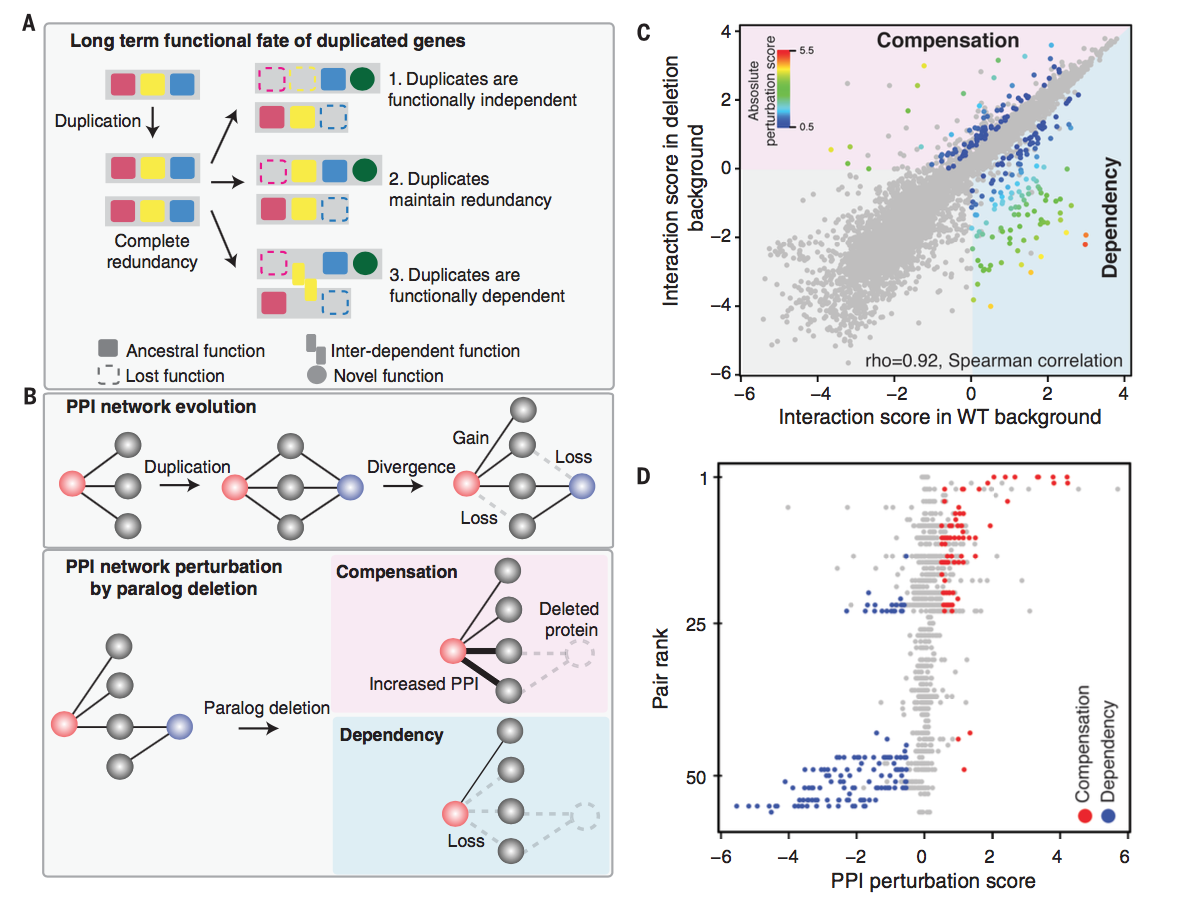
Figure 1: Figure
1A shows the possible fates of a pair of paralogs; they can either
become functionally independent, retain some redundant elements, or
become functionally dependent on each other. Duplication of a gene
also affects its protein-protein interaction network, as the paralogs
can independently lose or gain ability to interact with proteins. As
seen in Figure 1B, when one paralog is deleted, compensatory activity
presents as an increase in PPI intensity and dependent activity
presents as a decrease in PPI. PPI was measured using a protein-fragment
complementation assay; fragments of an enzyme necessary for growth in
a restrictive medium are fused to proteins so protein interaction
intensity can be measured as a function of colony growth. Figure 1C
graphs PPI intensity in wild-type vs. paralog-deleted conditions. Interaction
scores were strongly correlated, indicating that generally paralog
deletion does not significantly affect PPI network, but several genes
deviated from the best fit line, indicating significant compensatory
or dependent relationships. Figure 1D shows that compensation and
dependency typically did not occur in the same paralog pair.
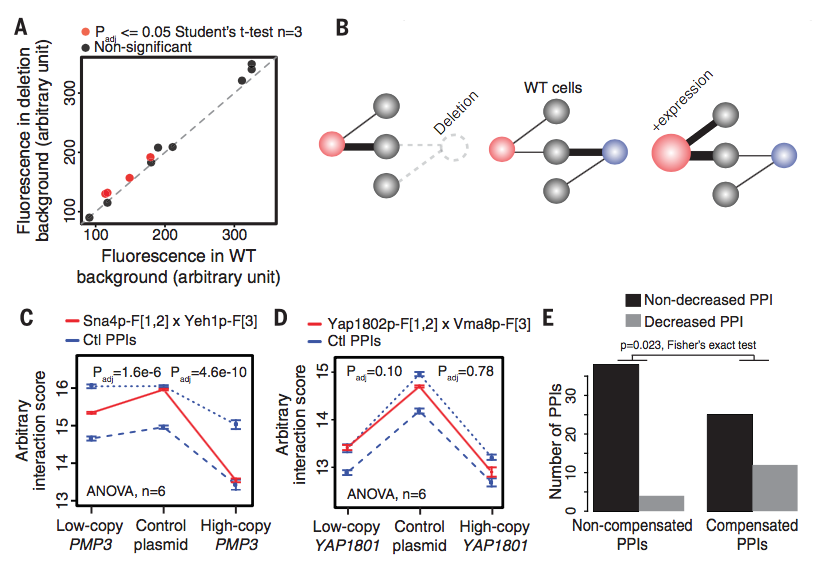
Figure 2:
Only a few cases showed a significant increase in protein levels via
flow cytometry in the paralog-deleted condition (Figure 2A), which
does not support paralog up-regulation as a mechanism. Figure 2B shows
an alternative mechanism for compensation, where the paralogs are
mutually exclusive; one paralog shows stronger interaction in the
wild-type case because it is more abundant or has a higher affinity.
If it is deleted, the other paralog is able to compensate. They tested
this theory buy overexpressing the compensating paralog, expecting a
decrease in the interaction of the originally dominant paralog. Figure
2C shows a case where this hypothesis was true; the interaction score
of the protein in red decreased when its compensating paralog was
overexpressed. Figure 2D shows a case where this hypothesis was not
true; the interaction score of the protein in red was not affected by
overexpressing of its paralog. Overall, paralogs pairs classified as
"compensating" were more likely to show decreased PPI upon
overexpression of the compensating paralog (Figure 2E)
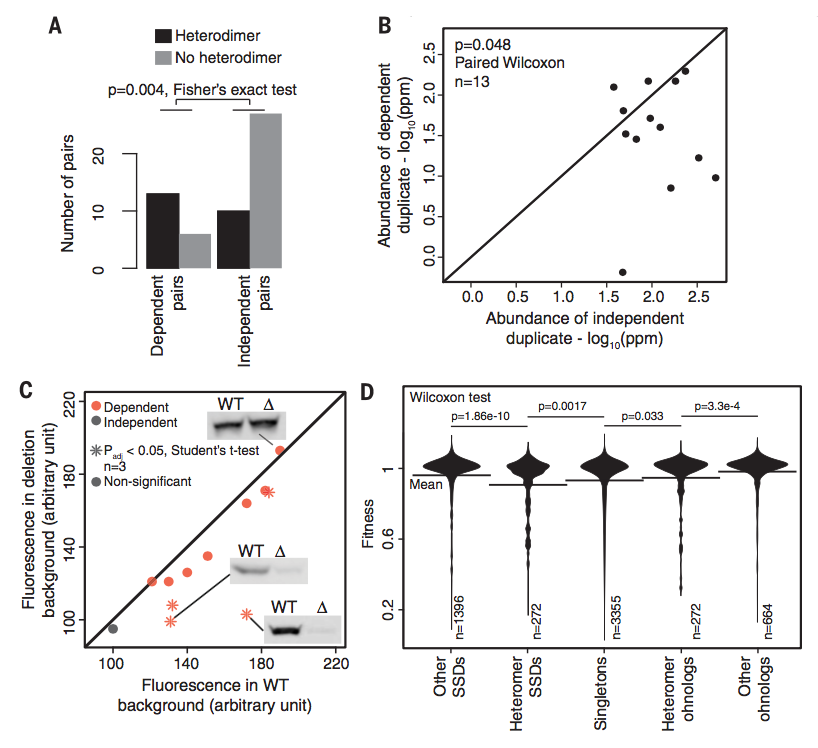
Figure 3: Figure
3 examines dependent paralog pairs. Figure 3A shows that dependent
paralog pairs are significantly more likely to be heteromers, or two
proteins that physically interact. Figure 3B shows that in
asymmetrical dependent pairs, where one protein can function without
the other but not vice versa, the independent protein tends to have a
higher abundance. The dependent protein abundance tends to lower
further when its independent counterpart is deleted, as measured by
flow cytometry and/or Western blots (Figure 3C). Figure 3D shows how
different types of gene duplication affect how deletion of one paralog
impacts fitness. Deletion of heteromer that originated from
small-scale duplications was significantly more detrimental than
deletion of singletons, or a non-duplicated gene, while deletion of
ohnologs was less detrimental than deletion of singletons. In both
cases, deletion of paralogs that formed heteromers had a larger
negative impact on fitness than their non-heteromer counterparts,
which supports that heteromers are working as functional units; if one
is deleted, the other is unable to function.
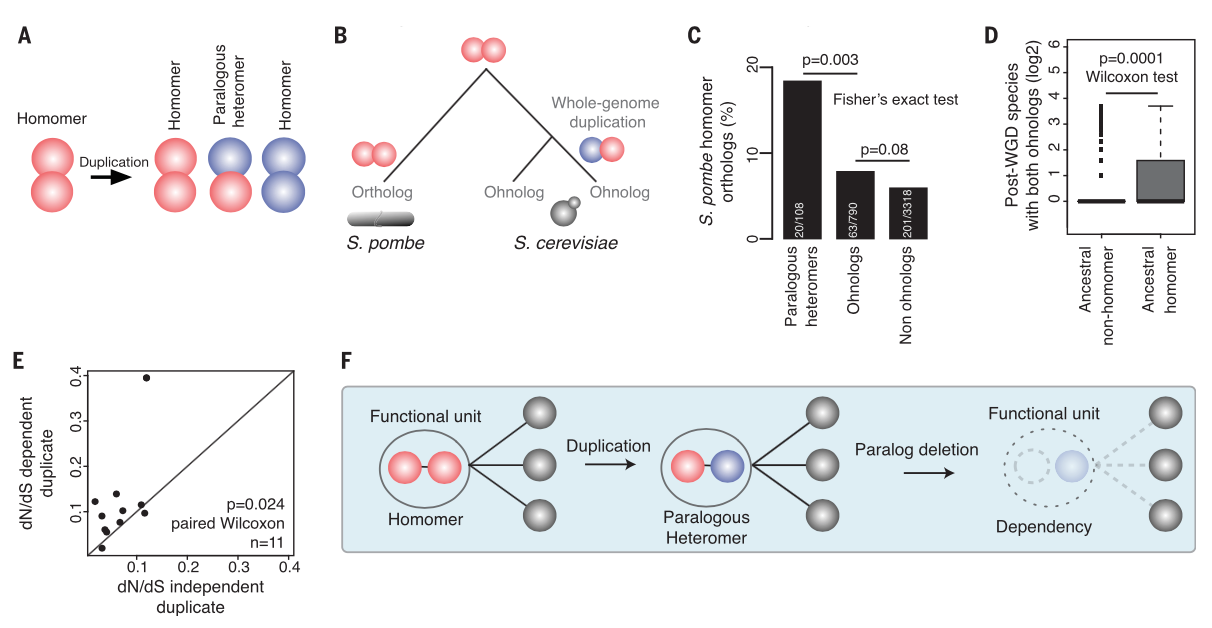
Figure 4: Figure
4A illustrates three possible dimer configurations after whole genome
duplication. Figure 4B illustrates the method of determining the
ancestral origin of a current paralogous heteromer resulting from
whole genome duplication (ohnomer), comparing the current paralog pair
to its ortholog in a related species that did not undergo whole genome
duplication. This method revealed that paralogous heteromers were more
likely to have a homomer ortholog (Figure 4C), supporting the
mechanism outlined in Figure 4A. Additionally, ohnologs that
originated from a protein that formed homodimers are more likely to
retain both paralogs (Figure 4D), further supporting functional
dependence of this class of proteins. In this configuration, the
dependent duplicate tends to accumulate more nonsynonymous mutations
than its independent counterpart (Figure 4E), suggesting that the
presence of an independent paralog allows for greater mutational
freedom. Figure 4F summarizes the entire paper: paralogs pairs that
originated from ancestral homologs become functionally dependent
heteromers after duplication, causing loss of function if one paralog
is deleted.
References:
1. Diss, Guillaume, Isabelle Gagnon-Arsenault, Anne-Marie Dion-Coté, Hélène Vignaud, Diana I. Ascencio, Caroline M. Berger, and Christian R. Landry. "Gene duplication can impart fragility, not robustness, in the yeast protein interaction network." Science 355, (February 10, 2017): 630-644.
Genomics
Page
Biology Home
Page
Hartlee's Home Page
Email Questions or Comments: hajohnston@davidson.edu
© Copyright 2017 Department of Biology, Davidson College, Davidson, NC 28035