This web page was produced as an assignment for an undergraduate course at Davidson College.
MY FAVORITE YEAST GENES
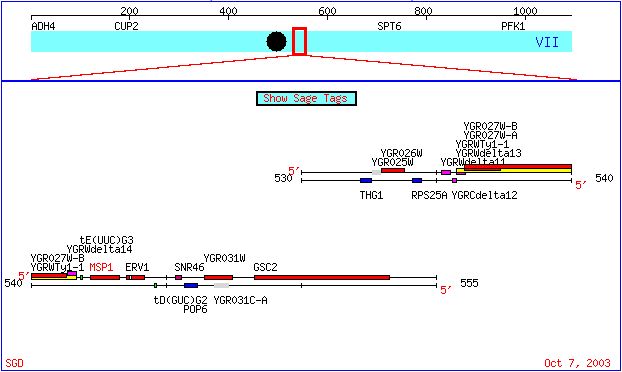
Figure1. Section of S. cerevisiae chromosome 7 showing the location of both Msp1 and YGR031W.
Msp1
Msp1 is a Saccharomyces cerevisiae gene located on the seventh chromosome
from base pairs 542204 – 543292. The gene consists of one exon that
is 1089 base pairs and 362 amino acids in length (SGD, accessed October 7,
2003; http://db.yeastgenome.org/cgi-bin/SGD/locus.pl?locus=msp1).
Nakai et al (1993) first reported msp1 in a paper published in The Journal
of Biological Chemistry. They found that MSP1, although important, was not
essential for either cell growth or mitochondrial functions in yeast because
the homozygous null mutant is viable (Nakai et al 1993).

Figure 2. Picture from Nakai et al (1993) showing the sequence of MSP1 and the subsequent amino acid sequence. In addition, the restriction enzyme sites, the putative membrane spanning domain and the putative nucleotide binding domain are shown. (permission pending)
Molecular Function
Msp1 is an ATPase of the putative AAA ATPase family. Figure 3 shows the physical
structure of 1e32, a member of the ATPase family whose structure is similar
to that of MSP1.
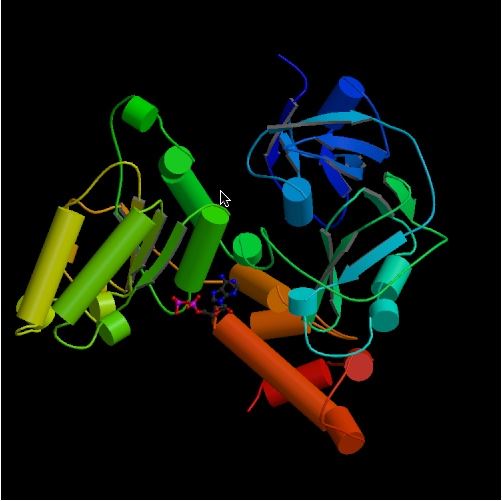
Figure 3. Physical structure of 1e32, a gene that encodes an ATPase similar to MSP1. (picture from PDB)
Biological Process
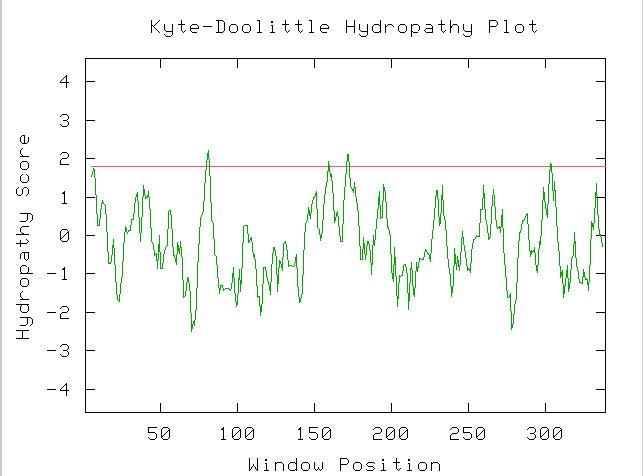
Msp1 is responsible for the translocation of proteins into the mitochondria.
In the presence of a translocating chain, the outer membrane import machinery
(MOM complex) and the inner membrane import machinery (MIM complex) form translocation
contact sites as a part of the membrane preprotein import machinery (Gene
Ontology, accessed on October 7, 2003; http://db.yeastgenome.org/cgi-bin/SGD/GO/go.pl?goid=6628).
Therefore, Msp1 functions as part of a chain across the mitochondrial membrane
to import proteins (see Figure 4).
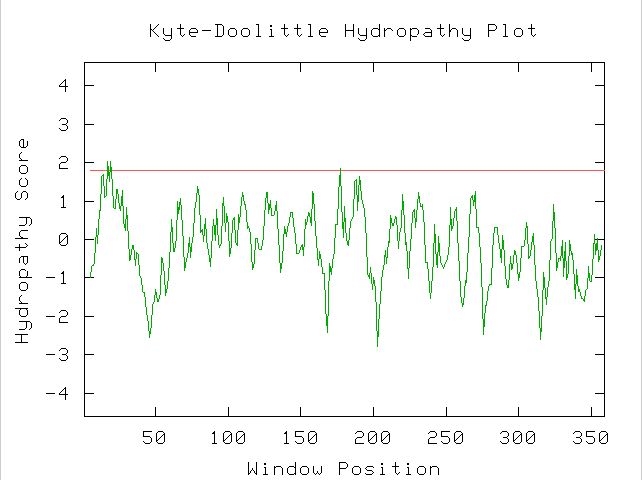
Figure 4. Hydropathy plot for MSP1 using the amino acid sequence, showing the transmembrane domain at the beginning of the protein. (image from Kyte-Doolittle)
Cellular Component
Msp1 is localized to the outer mitochondrial membrane. MSP1 interacts with
the cytoplasm to allow the preproteins from the cytoplasm to be translocated
(as mentioned above) into the mitochondria. MSP1 is an integral membrane protein
with one membrane spanning domain towards the beginning of the protein (see
Figure 2).
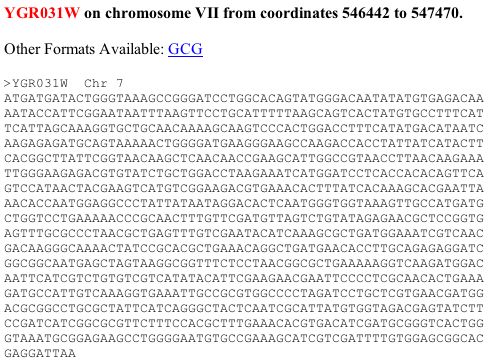
YGR031W
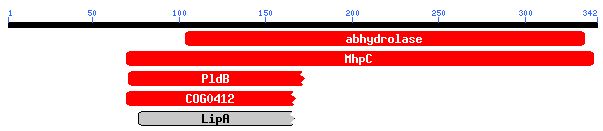
Since there is nothing known about this gene yet, I started with the nucleotide sequence (see Figure 5) and the amino acid sequence.