This page was produced as an assignment for an undergraduate course at Davidson College.
How to Determine EC Numbers
Enzyme Classification (EC) numbers are assigned to chemical reactions catalyzed by enzymes. Most EC numbers are associated with a name that should refer to the enzyme catalyzing the reaction called a systemic name. Combining the EC number with the systemic name to describe one protein allows for greater consistency across organisms and clarity across scientific disciplines.
How An Enzyme's Name Is Determined
EC numbers come in the following form: "EC #.#.#.#". Each level within the EC number denotes some part of the enzyme's function. The first number splits all catalyzed reactions into 6 major groups:
First EC Number |
Reaction Catalyzed |
Typical Enzyme Name |
1 |
Oxidation or Reduction Reactions: The transfer of O, H, or e- atoms and particles from one molecule to another. | Dehydrogenase, Oxidase |
2 |
Functional Group Transfer Reactions: The functional group may be methyl-, acyl-, amino-, or a phosphate group | Transferase, Kinase |
3 |
Hydrolysis Reactions: Cleaving a molecule by adding H2O | Lipase, Amylase, Peptidase |
4 |
Non-hydrolytic Cleaving Reactions: Cleaving a molecule with a C-C, C-N, C-O, or C-S bond | Decarboxylase, Lyase |
5 |
Isomerization Reactions: Intramolecular rearrangement | Isomerase, Mutase |
6 |
Synthesis Reactions: Making new C-O, C-S, C-N or C-C bonds by breaking down ATP | Synthetase |
After the first EC number division, classifying enzyme reactions becomes trickier. For example, 1.1.#.# enzymes are oxidoreductases that act on CH-OH groups only. 1.2.#.# enzymes are those that act on aldehydes or ketones. 1.3.#.# act on CH-CH groups. Within the 1.1 classification, 1.1.1.# enzymes are oxidoreductases that act on CH-OH groups with NAD+ or NADP+ as receptors. 1.1.2.# enzymes are those that act with cytochrome as the acceptor, 1.1.3.# means that oxygen is the acceptor, 1.1.4.# means disulfide is the acceptor, 1.1.5.# means that quinone is the acceptor, and he 1.1.99.# designation indicates that something else is the acceptor.
This system of nomenclature has some problems, the main one being that an enzyme cannot be classified or named until the reaction it catalyzes is clear. Also, more than one strand of amino acids can catalyze the same chemical reaction, and thus have the same EC number and name. Also, enzymes may catalyze a sequence of different reactions instead of one reaction, thus complicating the classification and naming process. More than one enzyme may work in concert to perform a single reaction. Finally, many enzymes perform both the forward and reverse of a catalytic reaction, and thus a standard procedure should be used to determine which name to use.
To deal with the issue of more than one enzyme catalyzing one reaction, the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology (NC-IUBMB) suggests that if more than one enzyme is involved, the word "system" should be appended to the end of the name. NC-IUBMB also suggests that for enzymes catalyzing a series of reactions, its name should be assigned based on the first reaction in series, and its following reactions can be listed in parenthesis after the systemic name. Finally, to deal with the problem of forward or reverse reactions, the NC-IUBMB recommends that scientists refer to their volume, which lists all chemical reactions known to be catalyzed by enzymes, and the systemic name reflects the direction in which the reaction is written. If a scientist discovers a new reaction, he or she can pick out the systemic name.
How An Enzyme's Function Is Determined
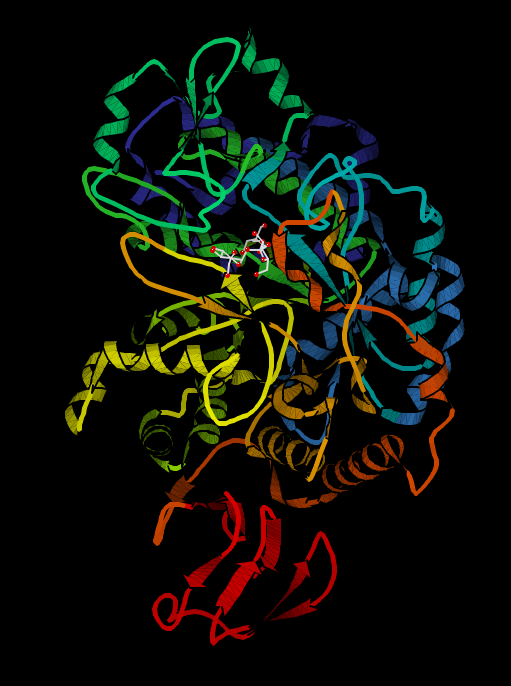
Determining an enzyme's function is a difficult process. Proteins with obvious known orthologs (similar amino acid chains, conserved binding domains) in different species lend themselves to function determination easily. However, in proteins with no known orthologs, scientists must start from scratch to determine an enzyme's substrate and function. A relatively new method called molecular docking uses a computer to determine which substrates are good candidates for a particular enzyme after the enzyme's 3D shape has been determined by X-Ray crystallography. 3D structures of many proteins are available at the Protein Data Bank online. Scientists can then use the protein's 3D structures paired with possible substrate's 3D structures and run thousands of substrates through a computer program which scores each substrate based on how well it fit into the enzyme's active site. The following is an image from the Protein Data Bank of amylosucrase (EC 2.4.1.4) with its substrate, sucrose:

The program then lists the potential substrates so the scientists can physically test the top-scoring substrates and find catalytic rate constants. The scientists can also find the actual structure of the substrate-enzyme complex via X-ray crystallography and see how closely it matches the computer prediction. Before the molecular docking method and computer program was developed, scientists would physically test thousands of substrates, and chemically determine the products and catalytic rate constants of all of these reactions. The catalytic rate constant is a measure of how many substrates were changed into product per unit time (in amylosucrase, this is determined by how fast the enzyme can convert sucrose and (1,4-alpha-D-glucosyl)n to D-fructose and (1,4-alpha-D-glucosyl)n+1). Once one knows which reactions an enzyme can catalyze quickly, one can infer the enzyme's function.
How An Enzyme's EC Number Is Assigned
After one knows the enzyme's function, one can easily determine its EC number and systemic name by looking at the atoms that move from one species to another in the chemical reaction. The table above details how to find the first EC number. Once you know the first EC number, the table below helps figure out what the second and third EC number mean. In all cases, the fourth EC number is specific to a particular reaction.
First E.C. Number |
Second E.C. Number Means: |
Third E.C. Number Means: |
| 1: Oxidoreductases | Indicates the H+ or e- donor that undergoes oxidation | Indicates the acceptor |
| 2: Transferases | Indicates the group transferred | Further information on the group transferred |
| 3: Hydrolases | Indicates the nature of the bond hydrolysed | Indicates the nature of the substrate |
| 4: Lyases | Indicates the nature of the broken bond | Further information on the eliminated group |
| 5: Isomerases | Indicates the type of isomerism | Indicates the type of substrates |
| 6: Ligases | Indicates the type of bond formed | Only used in C-N ligases |
To assign an EC number to an enzyme that has just been characterized, the chart at the bottom of this page is helpful. Let's say we found an enzyme that catalyzed this reaction:
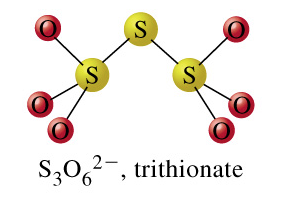
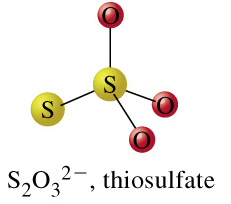
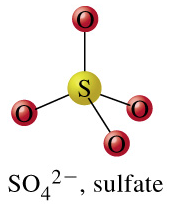
trithionate + H2O = thiosulfate + sulfate + 2 H+
 |
 |
 |
We can say that this enzyme is a hydrolase because it adds H20 into the molecule, which is then broken into two separate molecules. We would then click on the EC 3 button by the word Hydrolase in the chart, pictured below:

From the images of trithionate, the substrate, and thiosulfate and sulfate, the products, we can see that in this reaction, a sulfur-sulfur bond is cleaved. This leads us to believe that our reaction is denoted by the EC number 3.12. Click on the "separate" button that is on the same row as EC 3.12: Acting on sulfur-sulfure bonds, as pictured below:

If you follow the links through 3.12.1 and 3.12.1.1 (the only options available), you find that an enzyme catalyzing this reaction is called "trithionate hydrolase" with EC number 3.12.1.1. To figure out more about this EC number, you can look at these sites:
| Website | What it shows on each enzyme's page |
| BRENDA - The Comprehensive Enzyme System | Each EC entry has links to other databases, easily available information about which organisms the enzyme has been found in, the inhibitors and activators of the enzyme, data about the enzyme's optimal conditions and physical structure, and links to literature about the enzyme. |
| ExPASy - Expert Protein Analysis System | Provides links to other databases, including UniProtKB/Swiss-Prot entries for the enzyme in different species. Each species-specific enzyme page links to a consensus sequence, a feature table viewer of the protein, and links to published literature about the enzyme in that species. |
| KEGG - Kyoto Encyclopedia of Genes and Genomes | Shows a detailed reaction mechanism, and complete diagrams of the pathways the enzyme is involved in |
| ERGO Light - Integrated Genomics | Presents the pathway the enzyme is involved in with a list, and a searchable database of EC numbers found in all organisms so you can find which organism has your enzyme. |
Now you know how to find what an unknown enzyme does, determine its EC number, and use the EC number to find out more about the enzyme. You also know how systemic names are produced, and the rules scientists follow to create the systemic names and assign EC numbers. The information and databases presented will allow scientists to share discoveries about completely new enzymes or previously discovered enzymes in a new species.
References
Hermann, et al. 2007. "Structure-based Activity Prediction for an enzyme of unkown function". Nature 448: 775-779. Available <http://www.nature.com/nature/journal/v448/n7155/full/nature05981.html>. Accessed 11 November 2008.
NC-IUBMB. n.d. "Classification and Nomenclature of Enzymes by the Reactions they Catalyze". Available <http://www.chem.qmul.ac.uk/iubmb/enzyme/rules.html>. Accessed 9 November 2008.
NC-IUBMB. 2008. "Enzyme Nomenclature." Available <http://www.chem.qmul.ac.uk/iubmb/enzyme/index.html#recommend>. Accessed 9 November 2008.
Wikipedia. 2008. "EC Number." Available <http://en.wikipedia.org/wiki/EC_number>. Accessed 9 November 2008.
Genomics Page
Biology Home Page
Samantha's Home Page
Halorhabdus utahensis Genome Wiki