This experiment focused on how sexual dimorphism and environmental factors, specifically microbiomes and sex hormones, can affect susceptibility to autoimmune diseases. Recent studies have seen that these diseases are more prevalent in women. They used the nonobese diabetic (NOD) mouse, which models type 1 diabetes (T1D), because microbiomes can initiate testosterone protection from autoimmunity in this model. This mouse shows immune-mediated pancreatic B cell destruction that results in type 1 diabetes.
Testosterone seems to play a key role in protecting the mice from the disease phenotype. They found males were less affected than females in specific pathogen free (SPF) conditions, while both sexes seemed to be affected equally in germ-free (GF) conditions because of the microorganisms in the gut. They tested both groups for testosterone levels, which showed that GF females had increased testosterone levels compared to their SPF counterparts. Next they checked metabolite levels, which separated the SPF males and females, but not the GF cohort using PCA analysis, indicating that the microbiome influenced the metabolites present. They then looked at microbiome composition with bacterial 16S ribosomal RNA sequencing at weaning, puberty and adulthood, finding that the microbiome changed at puberty (which makes sense because of hormone changes). Then the group replaced, the gut bacteria of SPF females, using gavage techniques, with male and female bacteria and found the females with male microbiota fared better. Next, they tested the hormone levels of the galvanized SPF females, finding that they had increased levels of testosterones, but were still fertile. Lastly they manipulated the androgen receptors with an antagonist, flutamide, blocking testosterone receptors and lowering these levels. Flutamide caused the females with male microbiomes to act like untreated females.
From these tests they found out that testosterone was essential to prevent islet inflammation which would result in diabetes, in other words, testosterone helped protect individuals from the onset of diabetes because it corresponded to specific microbiomes. These results have implications for microbiome therapy to help cure or prevent diseases.
I liked this paper because it was very thorough; they really backed up what they had to say. When a test was done they made it clear that they had taken into account possible variables that the test might have induced and how they worked around the potential problem using different controls. They also would perform a test and interpret their results to determine a new test to verify that the cause of the observation was what they hypothesised, delving deeper into the root of their question. I think that the microbiome is an important area of research that needs to be studied more. This paper convinced me that a microbiome could have a huge impact on individuals. The only thing that was questionable to me in this paper was that they used this T1D mouse model because in humans type 1 diabetes does not have a sexual bias, so it makes me curious if there was a model that corresponded to bias in humans that they could have used instead of this model. However, it is an autoimmune disease and there are human autoimmune diseases that do show gender bias. I think that it really shows that there is a potential for therapy using microbiomes. In the paper they mentioned that the treatment was very affected for more than 11 weeks, suggesting that changing the microbiome could help provide an effective treatment that would last for months at a time even without changing the gut microbiome completely.

Question:
What are the differences between males and females in the SPF and the GF groups?
Method:
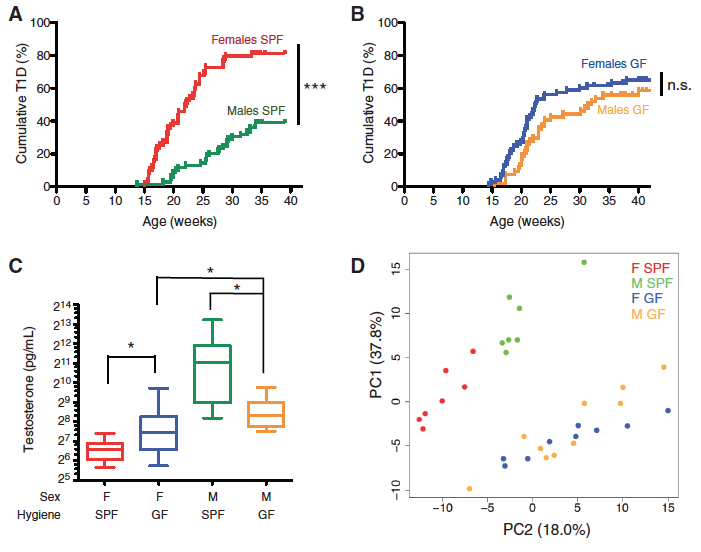
Panel A-B: The accumulation of T1D in the SPF (A) mice and the GF (B) mice was studied using these graphs. They compared females and males to see if one gender fares significantly better than the other, creating a survival curve. Statistics were done to see if there was a significant difference in survival between the groups and genders.
Panel C: They tested the testosterone level from each gender in the two groups to see if the cumulative T1D corresponded to serum testosterone levels. The line in the middle of the box shows the median value, the box contains the 25th to 75th percentile and the whiskers show the maximum and minimum values. Statistical analysis was performed to determine if the testosterone levels were significantly different between each group and gender.
Panel D: A Principal Component Analysis plot was produced from analyzing over 180 serum metabolites in each group and gender. PC1 accounted for 37.8% variation while PC2 accounted for 18%. The SPF males grouped together (green), SPF females grouped together (red), and GF males (yellow) and females (blue) were far from the SPF group but intermixed between themselves. This means that there was more variance between SPF females and males than GF females and males.
Take Home Message:
SPF males survive significantly better than SPF females and SPF females have significantly lower testosterone levels than SPF males. GF females and males do not have a significantly different survival rate and also do not have significantly different testosterone levels. The metabolites are also consistently different between SPF females and males and GF females and males group together with PCA. This shows a correlation between testosterone levels, metabolites and survival rates. It seems as if higher testosterone correlates to a specific metabolite composition and a better survival rate.
Question:
How is the composition of the gut microbiome different in SPF individuals of different genders at different stages of life?
Method:
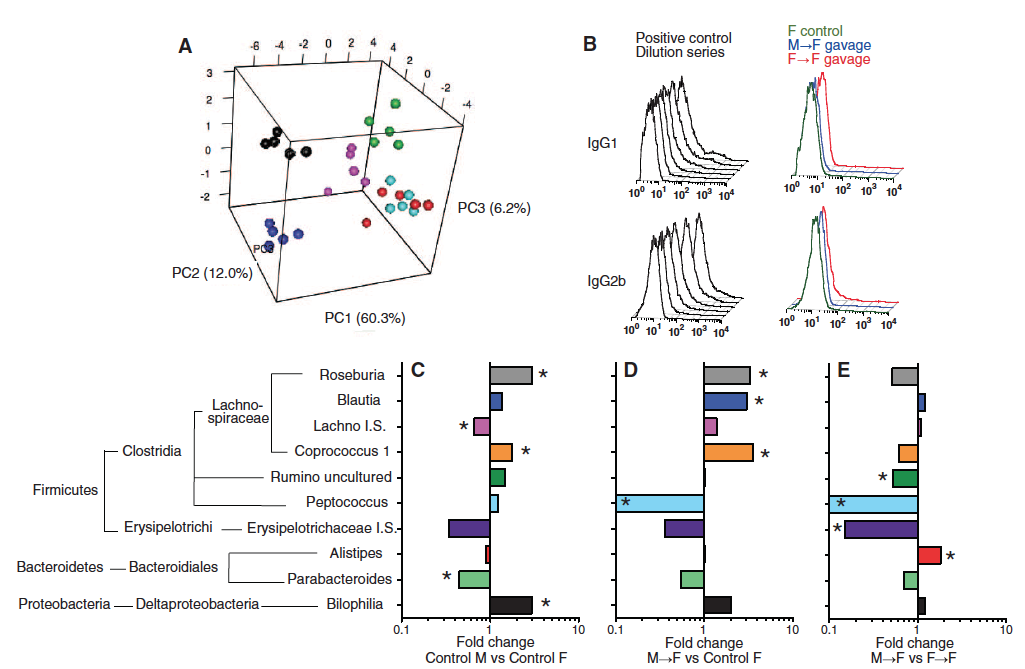
Panel A: A 3D Principal Component Analysis plot was produced using 16S bacterial RNA sequencing, which compared the top 30 bacterial families found in the gut. There were 6 groups tested in the SPF cohort. They took samples from females at 3 weeks (red), 6 weeks (green), and 14 weeks (black) as well as males at 3 weeks (turquoise), 6 weeks (pink), and 14 weeks (blue). They found that the 3-week-old males and females stayed clumped together but at 6 and 14 weeks male and female groups were separated, which is significant because 14 weeks is after puberty.
Panel B: Immune priming against the commensal microbiome was studied using immunoglobulin G1 and G2b to see if there would be any immune priming against the cecal bacteria they were feeding the mice that might interfere with their results. They saw that all of the dilutions corresponded to the control and the galvage samples confirming that there was no immune priming occurring when using this technique.
Panel C-E: Pairwise comparisons were used to study the sequences of females and males at 14 weeks by looking at the fold change in bacteria composition and amount. The bacteria that were observed are on the left, along with a phylogenetic tree to show relatedness between the species examined. C shows the females that received the male microbiome, compared to normal females. D shows females that received the male microbiome compared to females who received the female microbiome. E shows bacteria that were significantly different in C and D. In other words, C and D were the bacteria that were the most different between male microbiomes and female microbiomes.
Take Home Message:
This figure shows that the the microbiome of the gut is similar in the early stages of life, but that after puberty the content changes and males and females become different. The gut of males and females contain very different amounts of various types of bacteria, which is shown when you transfer a male microbiome into a female’s gut, suggesting that these differences in microbiome are set up early in life.
Question:
Does the transplantation of the male microbiome into a female change hormones or metabolites in the recipient mouse?
Method:
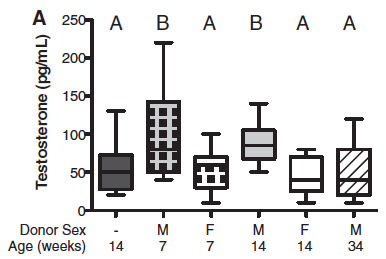
Panel A: Testosterone levels in the serum were measured in females of different ages with either a transfer of male or female microbiomes and compared them to untreated females at 14 weeks. The line in the middle of the box is the median, the top of the box shows the 75th percentile, the bottom of the box shows the 25 percentile and the minimum and maximum are shown by the whiskers.
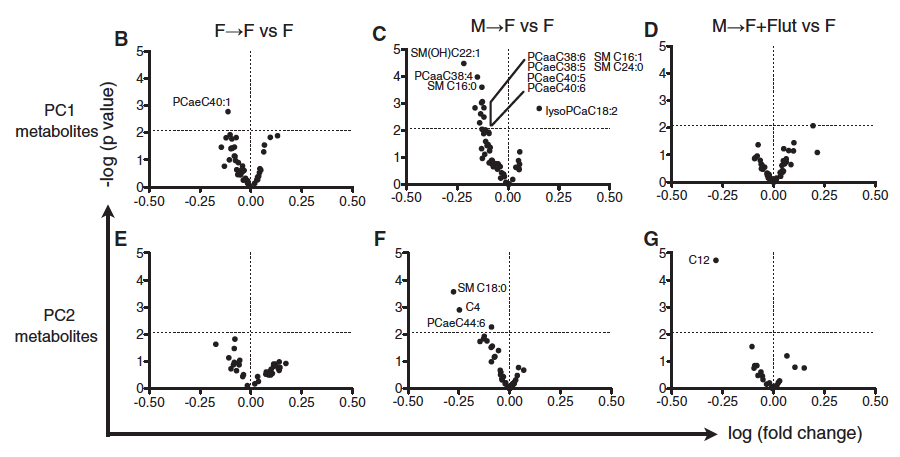
Panel B-G: 183 metabolites were tested in the serum of the unmanipulated females, the females with male microbiomes and the females with transferred female microbiomes and compared them to unmanipulated females. They made volcano plots using PC1 and PC2 metabolites, where they plotted fold change against p value. They highlighted metabolites that had a P value of more than 2.08 (represented by the horizontal dotted line).
Take Home Message:
Females with the male microbiome had the most metabolites that were significantly different from untreated females. Females that were given male microbiomes at 7 and 14 weeks had higher levels of testosterone than the unmanipulated females or females with the female microbiome. This shows a correspondence between composition of the microbiome, testosterone levels and metabolite composition. When the androgen receptor agonist, Flutamide, was added to the females with the male microbiomes it seemed to cancel out good effects, suggesting testosterone is the molecule that is causing the differences in metabolite composition.
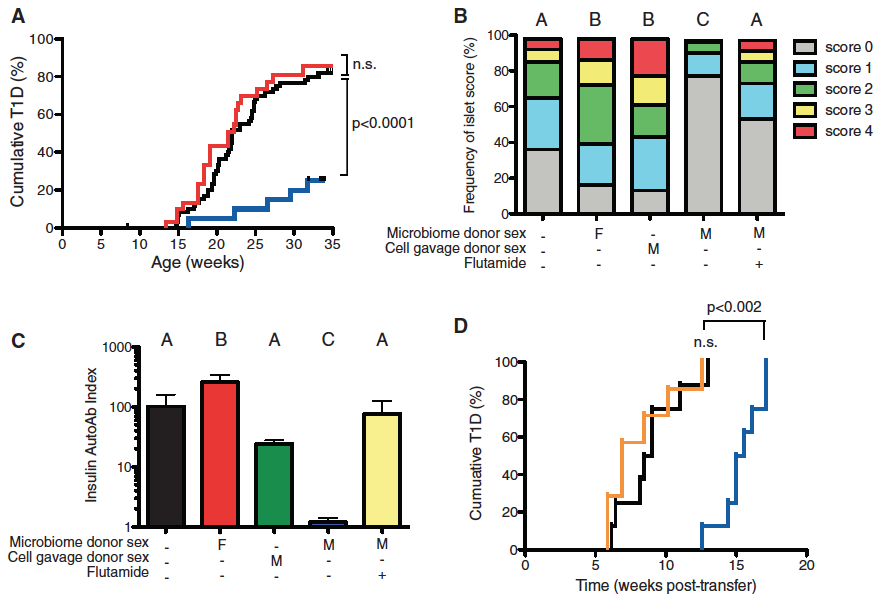
Question:
Do female NOD mice with transplanted male microbiomes (using cecal bacteria) have protection from T1D, decreased insulitis, and decreased insulin titer?
Method:
Panel A: This panel determined that females with male microbiomes (blue) have significantly lower levels of T1D than untreated females (black) or females with transferred female microbiomes (red). They simply altered the microbiomes of the mice and saw how quickly they accumulated T1D and compared them to unmanipulated female mice.
Panel B: Looked at insulitis severity and rated it on a scale from 0-4. The blocks show the percent of individuals with each score in each category: microbiome donor sex, cell gavage donor sex and Flutamide treated. The females with the male microbiome had the highest percent of individuals with the lowest scores. The male cell gavage donor was a control to make sure that rouge male cells did not yield the same results as the microbiome transfer.
Panel C: Insulin Autoab titer was used to detect preclinical symptoms of T1D. They compared untreated females to females with different microbiome donor sex, cell gavage donor sex or Flutamide treated. They found that females with a male microbiome had much lower levels of the Insulin Autoab titer and so did the females with the male cell gavage. They also saw that adding the agonist, Flutamide, caused the females with the male microbiome to have similar levels as the untreated females.
Panel D: This panel looked at females that were untreated (black), had a male microbiome (blue), or a male microbiome treated with Flutamide (orange). They purified T cells from each group and injected them into 4-5 week old NOD female mice. They saw that the females that received the T cells from untreated females and females that were galvaged and treated with Flutamide accumulated T1D much quicker than the females with the T-cells that had been exposed to the male microbiome. This indicates that the testosterone helps the T-cells fight off cumulative T1D.
Take Home Message:
Females with male microbiomes fair much better than any other females in cumulative T1D%, frequency of islets, and their Insulin AutoAb index. When a female with the male microbiome is treated with the androgen receptor agonist, preventing testosterone from taking effect, the female would have a similar phenotype to the untreated females. This figure shows that the increased level of testosterone caused by the male microbiome helps the T-cells better protect against T1D.
Markle, J., Frank, Daniel., Mortin-Toth, S., Robertson, C., Feazel, L., Rolle-Kampczyk, U., Bergen, M., McCoy, K., Macpherson, A., and Danska, J. 2013. Differences in the Gut Microbiome Drive Hormone-Dependent Regulation of Autoimmunity. Science 229:1084-1088.
Genomics Page
Biology Home Page
Email Questions or Comments: grgordon@davidson.edu.
© Copyright 2014 Department of Biology, Davidson College, Davidson, NC 28035