This web page was produced as an assignment for an undergraduate course at Davidson College.
Optical Control of Muscle Function by Transplantation of Stem Cell-Derived Motor Neurons in Mice
Paper Review by Ellen Johnson
Summary
Researchers set out to resolve a problem that neither electrical stimulation nor embryonic stem cell (ESC)-derived motor neurons could resolve alone. Electrical stimulation can activate motor neurons and initiate muscle contraction but cannot effectively restore function to denervated muscle. While ESC-derived motor neurons can reinnervate muscles, electrical stimulation of these motor neurons stimulates both ESC-derived and endogenous motor neurons.
The ChR2 motor neurons may provide the solution. Unlike endogenous motor neurons, ChR2 motor neurons respond to optical stimulation. Researchers genetically engineered motor neurons from embryonic stem cells to engraft into a partially denervated peripheral nerve. The engrafted ChR2 motor neurons were designed to respond to optical stimulation. Researchers conducted in vivo mouse experiments. They tested whether ChR2 neurons could reinnervate degenerate muscle and control muscle function independent of endogenous motor neurons.
Opinion
The paper has interesting potential applications for regenerative medicine though I suspect that light-stimulated muscle reinnervation is a ways off still. The research stands as more proof-of-principle than clinical application since there are many intermediate steps that must be met along the way. Two that the researchers point out are implantable optical stimulators and graft cell encapsulation devices.
While the text was easy to follow, the figures left much to be desired. Researchers do not clearly indicate how to distinguish between ChR2 and endogenous motor neurons in figure 2 panels c through e. Instead, the reader is left to infer that ChR2 appear green while endogenous appear red. A simple label would allow quicker understanding of these images. Distinguishing between ChR2 and endogenous neurons is ineffective for figure 2e, which includes an asterisk marking an apparently absent endogenous nerve. The faint red line is visible only upon significant zoom. Using another endogenous nerve as a reference or providing a zoomed-in panel would eliminate this visualization problem. Figure 3 panels b, c, d, h, and i should have labeled axes instead of the scale in top right corner. Labeled axes would eliminate the need for the reader to visually superimpose the scale onto the actual data.
I was equally disappointed in the amount of trust readers must place in the authors. Figure 2d should have been accompanied by a quantification of the degree of myelination compared to endogenous myelination. Without quantification, readers are left to trust that myelination in this small cross-section is indicative of all myelination in the area. Providing quantification similar to that in figure 2f would have made the data more persuasive. Figure 3e suffers from a similar problem. Researchers say the forces for tetanic and twitch stimulation are similar to normal values in uninjured animals but do not provide control data. Control data would have been helpful for comparison and its absence forces the reader into blind trust.
Figures
Figure 1:
Panel A compares the genetically modified neurons (CAG::ChR2-YFP/Gdnf) to control cells (parental). As expected, both express the motor neuron reporters GFP and Isl1/2 while only CAG::ChR2-YFP/Gdnf express YFP. YFP expression was added during genetic engineering as part of a photoreceptor transgene (CAG::ChR2-YFP).
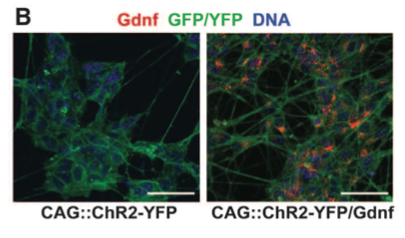
Panel B compares CAG::ChR2-YFP/Gdnf to CAG::ChR2-YFP to indicate that only CAG::ChR2-YFP/Gdnf expresses Gdnf.
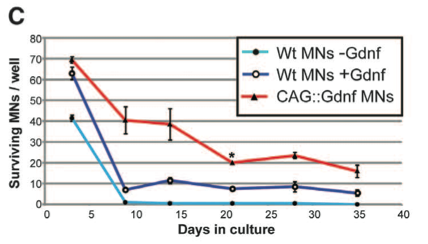
Panel C demonstrates that Gdnf expression increases motor neuron survival. Wild-type motor neurons without Gdnf show 40% survival at day 3, which decreases to approximately 0% by day 10. Adding Gdnf to wild-type motor neurons increases survival to approximately 63% at day 3. Cell survival decreases to and remains at approximately 10% at days 10 through 35. CAG::ChR2-YFP/Gdnf shows the highest survival rate beginning at approximately 70% and decreasing to approximately 15% by day 35.
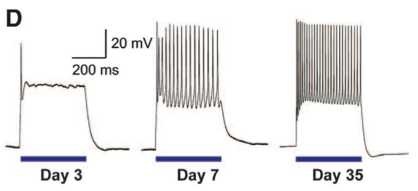
Panel D shows it takes 35 days for CAG::ChR2-YFP/Gdnf motor neurons to respond to optical stimulation in a way that resembles adult motor neuron response to electrical stimulation. Blue bars indicate optogenetic stimulation.
Figure 1 conclusions: The main purpose of this figure is to familiarize the reader with the general properties of the genetically modified ESC-derived motor neurons that researchers used for later experiments. Only the genetically modified neurons express YFP and Gdnf. Expression of the latter improves neuron survival. Finally, it takes 35 days for the genetically modified neurons to respond to optical stimulation in a similar manner as mature adult motor neurons.
Figure 2:
Panel A shows the ligation site where researchers cut the sciatic nerve to simulate partial muscle denervation ChR2 motor neurons were engrafted distal to the ligation site as indicated. Dashed lines show approximated axon trajectory resulting from graft.
Panel B shows the engrafted motor neurons express GFP/YFP and mature motor neuron marker choline acetlytransferase (ChAT). ChAT expression indicates the engrafted neurons are morphologically mature.
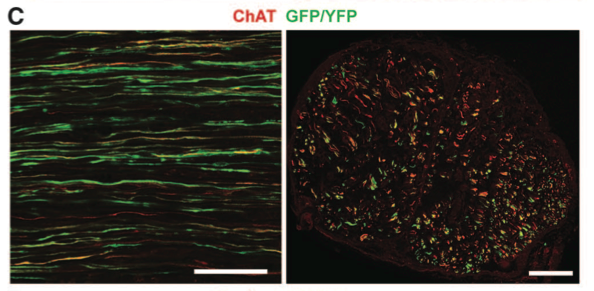
Panel C shows endogenous and ChR2 motor neuron axons in the common peroneal nerve, which is labeled in panel a. ChR2 motor neuron axons appear green due to GFP/YFP expression. Endogenous axons appear red due to expression of only ChAT.
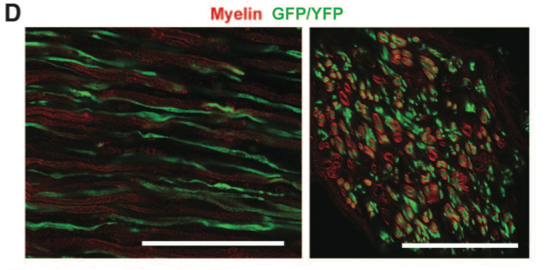
Panel D shows myelination of ChR2 motor neurons. The transverse cross-section demonstrates that myelin (red) surrounds ChR2 motor neurons (green).
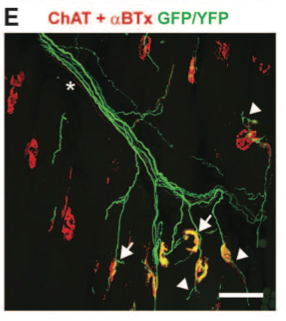
Panel E depicts ChR2 motor axon terminals in the triceps surae (TS) muscle, which is labeled in panel a. Asterisk denotes endogenous nerve (red line). Neuromuscular junctions show signs of inactivity including preterminal collateral sprouting (arrows) and terminal sprouting (arrowheads).
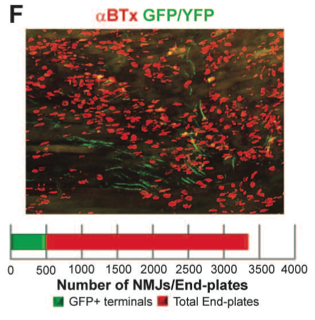
Panel F provides quantification for innervations depicted in panel e. ChR2 neuromuscular junctions appear green while total end-plates labeled with α-bungarotoxin (α-BTx) appear red. 14.7% of all end plates in the TS muscle are innervated by ChR2 motor neurons.
Figure 2 conclusions: Researchers conclude that ChR2 motor neurons stimulate axon growth and muscle reinnervation. ChR2 motor neurons show myelination and inactive junctions. Researchers suggest inactivity hallmarks are due to neuron inactivity in the absence of optical signal.
Figure 3:
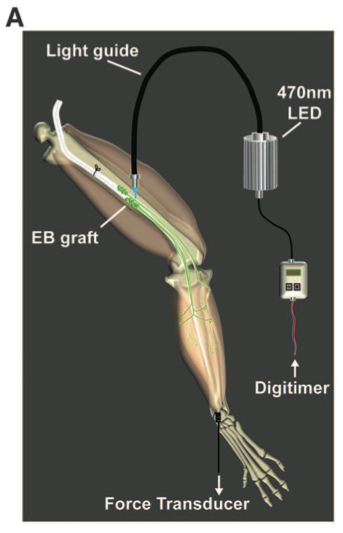
Panel A shows a schematic diagram of how light was used to optically stimulate muscles. An LED unit emitted 470-nm blue light to the graft site via a light guide.
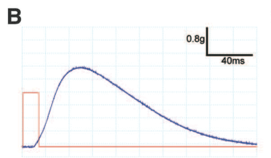
Panel B shows 14-ms light pulses (red line) induce submaximal twitch contractions in muscles (blue line).
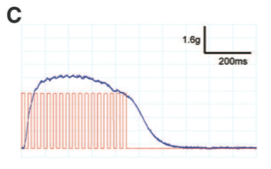
Panel C shows high-frequency light pulses (red lines) induce maximal muscle stimulation (blue line).
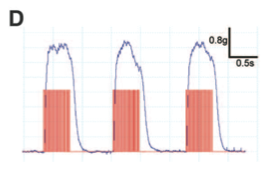
Panel D The tetanic muscle contractions (blue lines) are highly reproducible upon repeated exposure to high-frequency light pulses (red lines).
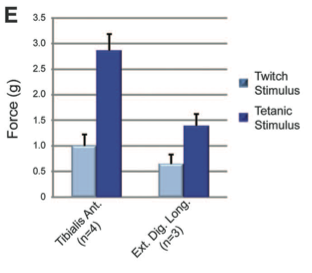
Panel E shows the contractile force for tibialis anterior (TA) muscle and extensor digitorum longus (EDL) muscle. More force was needed for tetanic stimulation than for twitch stimulation of both muscles. The TA muscle required more force than the EDL muscle for both twitch and tetanic stimulation.
Panel F shows time to peak contraction after optical (blue bars) or electrical (gray bars) stimulation. Both TA and EDL muscle show approximately equal times to peak contraction regardless of stimulation type. While it appears EDL muscle shows a larger lag time for electrical stimulation than optical stimulation, researchers do not indicate such a difference exists. Readers are lead to assume the these data sets do not exhibit statistically significant differences despite the appearance the error bars may not overlap. Data supports that of figure 2 panel d. ChR2 motor neurons are myelinated and thus exhibit similar conduction velocities as electrically stimulated motor neurons. n indicates neurons optically or electrically stimulated, respectively.
Panel g shows time to peak contraction after muscle contraction begins. Time to peak contraction is the same regardless of optical or electrical stimulation.
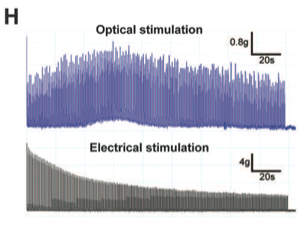
Panel H optically stimulated motor neurons are more fatigue resistant than those stimulated electrically. Every second, neurons were exposed to 40Hz stimulation for 250 ms. These repeated exposures continued for 180s total. Electrical stimulation fatigue begins almost immediately and seems to level off towards the end of the 180s trial. Optical stimulation shows initial increase in output and subsequent decline to steady level of output at approximately 90s.
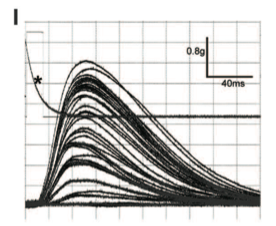
Panel I shows that motor neurons are differentially stimulated depending on their activation threshold. Those with lower thresholds show higher peaks. Asterisk shows square-wave trigger voltage to the LED source.
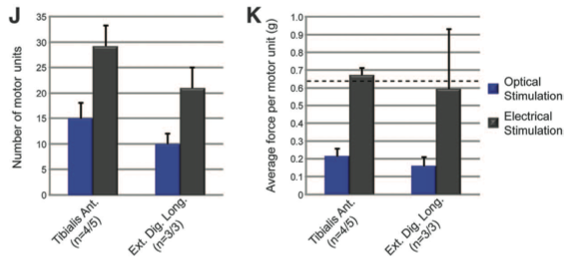
Panel J shows quantification of the number of motor units innervating TA and EDL muscle upon optical (blue bars) or electrical (gray bars) stimulation. Approximately 15 motor units innervate TA muscle upon optical stimulation. Approximately 10 motor units innervate EDL muscle upon optical stimulation. Both muscles show significantly fewer motor units when optically stimulated than when electrically stimulated. The difference is to be expected since optical stimulation activates only on ChR2 motor neurons while electrical stimulation activates ChR2 and endogenous motor neurons. From these differences, it appears ChR2 motor neurons represent approximately 50% of all motor units. n indicates neurons optically or electrically stimulated, respectively.
Panel K shows the average force per motor unit for TA and EDL muscles when optically (blue bars) or electrically (gray bars) stimulated. Dashed line indicates average motor force for normal neurons is approximately 0.62g. Electrical stimulation results in significantly higher average force per motor unit than optical stimulation. Optical stimulation of TA muscle yields an average motor force of approximately 0.21g while that of EDL muscle yields an average motor force of approximately 0.16g.
Figure 3 conclusions: Researchers conclude that optical stimulation allows engrafted motor neurons to control muscle function. Optically stimulated neurons yield similar contractile forces to those initiated by electrically stimulated neurons. The optically stimulated neurons are more fatigue resistant than electrically stimulated neurons and appear to account for 50% of all motor units.
References
Genomics Page
Biology Home Page
© Copyright 2014 Department of Biology, Davidson College, Davidson, NC 28035