
This web page was produced as an assignment for an undergraduate course at Davidson College.

From Markle et al.2013
Markle et al. “demonstrate that early-life microbial exposures determine sex hormone levels and modify progression to autoimmunity in the nonobese diabetic (NOD) mouse model of type 1 diabetes (T1D)” (Markle et al., 2013). Their research aims to assess and characterize the impact of “sexual dimorphism and environmental factors” on autoimmunity (Markle et al., 2013). To characterize the impact of these two factors, Markle et al. introduce microbiota from NOD male mice into NOD female mice and quantify changes in the female microbiome in addition to conferred protection against diabetic and pre-diabetic symptoms.
Based on their data, Markle et al. conclude that the microbiome in NOD mice determines sex-based autoimmunity, the microbiome changes after puberty and can be altered semi-permanently by introducing the microbiome of another mouse of the opposite sex, introduction of male microbiota changes the recipient female’s microbiome and metabolism by elevating testosterone, and the elevation in testosterone protects recipient females from pre-diabetic and T1D symptoms. To summarize, “microbiome alterations in young, commensally colonized mice conferred testosterone and metabolite changes sufficient to oppose genetically programmed autoimmunity” (Markle et al., 2013).
This research has huge implications for human application. For example, if introducing a certain set of bacteria can change a person’s metabolism enough to suppress an autoimmune response, many autoimmune diseases could be treated or even cured. Additionally, this method seems to be at least somewhat permanent, which implies that the treatment would only need to occur once or a few times throughout the person’s lifetime.
Overall, I thought this research was well-planned and well-controlled. The data are displayed for the same test groups throughout the paper, which makes it easy to logically progress from the implications of one figure to the implications of the next. Additionally, the conclusions from each figure progress logically to the next experiment and figure, and build on one another to instill an understanding of the big picture and impress on readers the magnitude of the clinical implications behind such a treatment. The multiple controls in each figure clearly demonstrate the significance of their findings and eliminate the possibility for most, if not all, other possible implications.
Figure 1A shows that females housed in specific pathogen free (SPF) environments have a higher incidence of T1D than SPF males by age 40 weeks. Figure 1B shows that females housed in a germ-free (GF) environment did not have a statistically significant difference in incidence of T1D than GF males by age 40 weeks. Together, these figures suggest that the “relative protection of males in the SPF setting … [is] dependent on the presence of commensal organisms” (Markle et al., 2013). Figure 1C shows that GF females have a higher testosterone level than SPF females, and GF males have lower testosterone levels than SPF males. This implies that “microbial colonization [affects] sex hormone levels” (Markle et al., 2013). Figure 1D shows that in different microbial communities, males and females had different hormonal and metabolic responses, and within the same microbial community (SPF), males and females had sex-specific responses. This reveals “a role for microbiome exposure in regulating sex-specific features of the host metabolic profile” (Markle et al., 2013). Taken together, these panels show that “sex-based autoimmunity in the NOD mouse depends on the microbiome” (Markle et al., 2013).
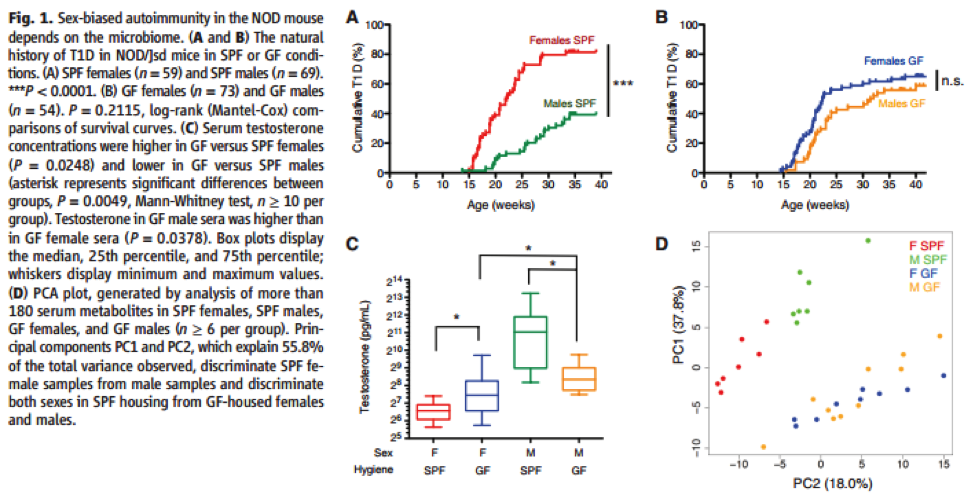
Figure 2A uses principle component analysis in SPF NOD males and females at weaning, puberty, and adulthood to show that “sex-specific differences in microbiome profiles emerge after puberty” with regard to the cecal microbiome community (Markle et al., 2013). Figure 2A also shows that microbiome differences become more extreme with age. The histograms in Figure 2B show that cecal microbiome transplantation in male to female or female to female SPF NOD mice does not induce systemic immune priming (Markle et al., 2013). In other words, transplanting the intestinal microbiome of an adult mouse to a young mouse did not induce the production of antibodies specific to a post-puberty microbiome community. Since “systemic exposures to microbial agents can protect NOD mice from T1D” via the production of antibodies, this control ensured that the feeding method used did influence the microbiome community (Markle et al., 2013). Figures 2C-E show that “introduction of either adult male or female microbiota stably altered the recipient microbiome relative to unmanipulated female controls” (Markle et al., 2013). In other words, while the feeding method did not influence the cecal microbiome community, the microbiota from another mouse significantly changed the microbiome of the recipient for a considerable amount of time after the transfer. Collectively, the panels in Figure 2 indicate that after puberty, SPF NOD males and females have different microbiome profiles, and these profiles can be altered semi-permanently by introducing another mouse’s microbiome. Additionally, these results are not due to the method used to transfer the microbiome.
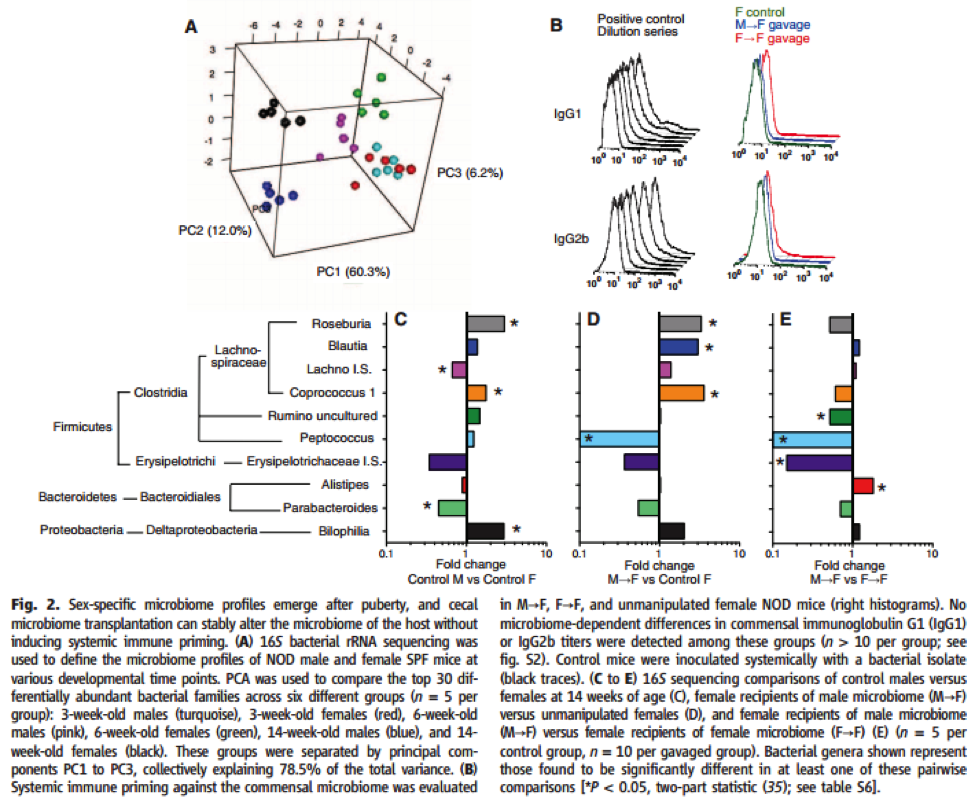
Figure 3A shows that when SPF NOD male microbiota was transplanted in SPF NOD females, the female testosterone level was significantly higher at 7 and 14 weeks, but not at 34 weeks. To test whether the sustained increase in testosterone had a broader metabolic influence, Markle et al. used the metabolites from PC1 and PC2 in Figure 1D to compare mice with transplanted microbiomes to unmanipulated mice in Figure 3B, C, E, and F. The data indicate that transfer of male microbiota had a broader metabolic influence because it significantly lowered “serum concentrations of glycerophospholipid and sphingolipid long-chain fatty acids” (Markle et al., 2013). Figure 3D and G indicate that testosterone elevation was responsible for the metabolic changes in the male-to-female microbiota transfer. Together, these panels suggest that SPF NOD male microbiota transfer affects the female recipient’s microbiome and metabolism, in part because it results in sustained elevated testosterone in the female recipient.
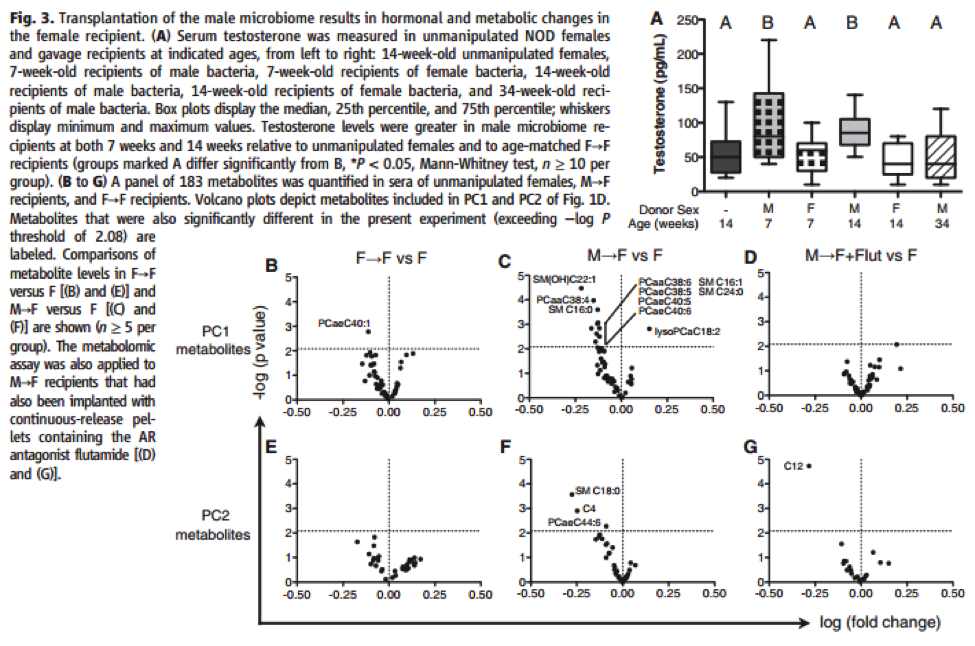
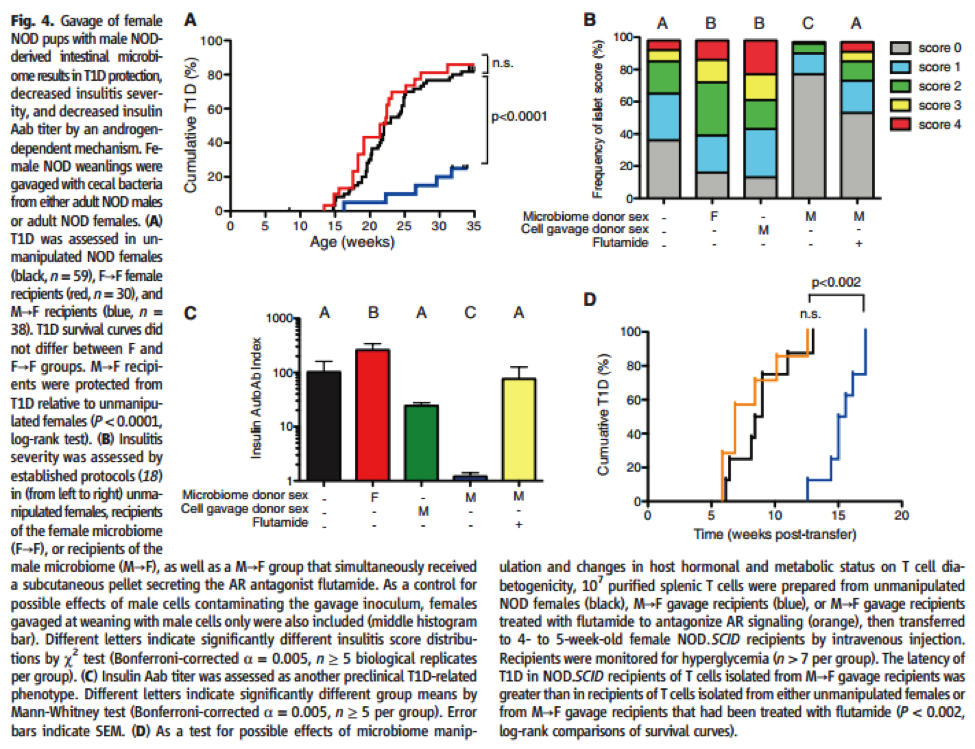
Figure 4A shows that female recipients of the male microbiome were “strongly protected from T1D” (Markle et al., 2013). Figure 4B shows that the male microbiome transfer protected recipient females from invasive insulitis, which precedes diabetes onset. Additionally, Markle et al. show that introducing sterile male cells (not the male microbiome) did not confer protection from insulitis, and that blocking the androgen receptor eliminates the conferred protection. In other words, “testosterone activity was essential to the protection” from insulitis in female recipients (Markle et al., 2013). Similarly, Figure 4C shows that the male microbiome transfer protected recipient females from insulin-specific Aab, “a second autoimmune phenotype in prediabetic NOD mice” (Markle et al., 2013). Once again, Markle et al. show that sterile male cells did not confer protection, and blocking the androgen receptor eliminated male-transfer protection. In Figure 4D, Markle et al. compare the ability of T-cells to “transfer T1D to lymphocyte-deficient, T1D-resistant NOD.SCID (severe combined immunodeficient) recipients” (Markle et al., 2013). The data show that male microbiome transfer slowed the ability of T-cells to transfer T1D to NOD.SCID recipients. Once again, Markle et al. show that blocking the androgen receptor eliminates protection conferred through male microbiome transfer. Therefore, male microbiome transfer protects female recipients from “the autoimmune response evident in insulitis progression, Aab production, and the capacity of T-cells to transfer diabetes” in a testosterone-dependent manner (Markle et al., 2013).
Sources:
Markle, Janet G. M., et al. “Sex Differences in the Gut Microbiome Drive Hormone-Dependent Regulation of Autoimmunity.” Science 339:6123 (2013): 1084-1088. Web. 20 April 2014. Available https://www.sciencemag.org/content/339/6123/1084
Assignments:
Genomics Page
Biology Home Page
© Copyright 2014 Department of Biology, Davidson College, Davidson, NC 28035