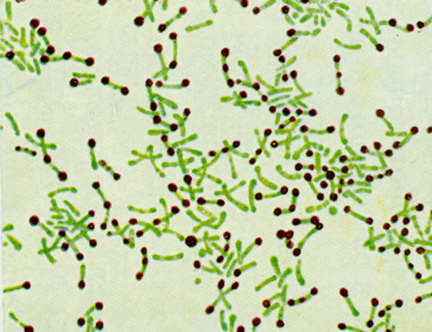
Corynebacterium diphtheriae belong to the family Mycobacteriaceae, which also includes Mycobacteria and Norcardia, and are Gram-positive and aerobic. These rod-shaped bacteria often have a distinctive club shape and cluster in a way resembling Chinese characters, as can be seen in the image below. The club or bar appearance of the cells is due to pockets of inorganic phosphate which form metachromatic granules when stained (Todar, 2002).
http://www.tjclarkinc.com/bacterial_diseases/diphtheria_.htm
C. diphtheriae causes the disease diphtheria which primarily affects the upper respiratory tract. The bacteria can invade any of the mucous membranes and have a 2-5 day incubation period. Different sites of infection cause different clinical forms of diphtheria with varying severity. The most commons site of infection is the pharynx and tonsils, but the bacteria can also invade the nasal tissues, larynx and skin. Anterior nasal diphtheria is usually quite similar to the common cold in symptoms and severity because the location of the infection does not allow for large-scale systemic absorption of the diphtheria toxin (Centers for Disease Control and Prevention, 2007).
Pharyngeal, tonsillar and laryngeal diphtheria are more severe because more toxin is absorbed into the bloodstream. Early symptoms of this form of diphtheria include a sore throat and fever, but after a few days a bluish-white adherent membrane, the pseudomembrane, forms over the back of the throat and tonsils. The pseudomembrane is actually a fibrin network infected with multiplying C. diphtheriae cells which grows over a necrotic lesion on the epithelial cells on the back of the throat. The consequences of this membrane growth can be severe if the membrane grows to the extent that it blocks the airway in the throat. This infection quickly becomes acute as the bacterial toxin is absorbed into the bloodstream and lymphatic network. Cutaneous diphtheria, or an infection of the skin, is much less common. Symptoms may include a rash or ulcer on the skin. This form of the infection is generally less severe (Centers for Disease Control and Prevention, 2007).
The ultimate outcome of the disease depends on how much toxin is absorbed into the bloodstream. The toxin has degenerative effects on the heart, nerves, spleen, muscles, liver and kidneys. In the most severe cases, an infected person will develop myocarditis or neuritis, which leads to heart failure and local paralysis most commonly of the soft palate, respectively. The fatality rate is approximately 5-10% of cases, but is higher for young children or the very old (Centers for Disease Control and Prevention, 2007).
The Toxin
Not all strains of C. diphtheriae are toxic because the gene for diphtheria toxin (the actual disease causing agent, abbreviated DT) is not found in the bacterial genome. The DT gene is actually located on the genome of a corynephage, a certain type of prophage. Not all phages carry the DT gene tox, so it is possible for a C. diphtheriae bacteria to undergo lysogenic conversion, or be infected by a prophage, without producing the diphtheria toxin. Therefore, the toxinogenicity of C. diphtheriae is dependant on phage conversion with a phage that has a functional gene for the toxin protein (Holmes, 2000).
Expression of the tox gene appears to not be regulated in the same way as the other prophage genes and it is located at the end of the prophage genome. These findings suggest that the tox gene might have been incorporated into the genome of a nontoxinogenic ancestor by recombination (Holmes, 2000).
Diphtheria toxin is produced from the diphtheria bacteria as a single protein. This toxin is technically a proenzyme, because it is cleaved by bacterial proteases into two fragments, A (molecular weight 21,150) and B (molecular weight 39,000) (Middlebrook and Dorland, 1984). Fragment A is catalytically active and is the sole source of toxicity of diphtheria toxin. Fragment B appears to have no enzymatic activity and is far less stable than the active A fragment (Collier, 1975). Each fragment plays a different role in the toxin’s entry into the host cell. DT enters the host cell by binding to the extracellular EGF domain of the heparin-binding epidermal growth factor precursor (HB-EFG precursor). This binding initiates a hydrophobic domain of the B fragment to form a channel across the membrane through which the A fragment can pass into the cytoplasm. The B fragment contains the transmembrane and receptor-binding domains of the toxin, so this piece remains in the plasma membrane (Ren et al., 1999).
Diphtheria toxin is an ADP-ribosyl (ADPR) transferase. Its toxicity comes from blocking protein synthesis in the host cell by inactivating EF-2, or cellular elongation factor. EF-2 is an essential part of the translation process. Fragment A that is free in the cytoplasm catalyzes the reaction as summarized below:
EF-2 + NAD+ --> ADPR-EF-2 + nicotiamide + H+
This reaction forms a covalent bond between ADPR and EF-2 which blocks the functional site which interacts with RNA in translation, and consequentially preventing all protein synthesis at a particular ribosome (Collier, 1975; Middlebrook and Dorland, 1984).
Although the structural gene for DT is on the phage chromosome, the expression of this gene is controlled by DtxR gene on the C. diphtheriae chromosome. Expression of tox is usually inhibited by negative feedback from the DTxR gene product. The levels of iron in the surrounding tissue environment has a significant effect on the expression of DTxR and consequentially on DT production. Low levels of iron inhibit the expression of DTxR, which then stops the negative feedback loop inhibiting tox gene expression. If the C. diphtheriae bacteria are grown under certain severe iron-deficient conditions, DT protein can make up about 5% of all protein being synthesized. In other words, iron acts as a co-repressor with DTxR to prevent expression of DT (Holmes, 2000; Centers for Disease Control and Prevention, 2007).
Diphtheria is unusual as a bacterial infection because the bacteria themselves only invade superficial tissues. However, the toxin released spreads throughout the entire body and attacks cells without specificity. Severe infection with diphtheria toxin can cause death by necrosis of the tissues of essential organs such as the heart, kidneys, liver and lungs by blocking all protein synthesis in infected cells (Collier, 1975).
References
Collier, R. John. “Diphtheria Toxin: Mode of Action and Structure.” Bacteriological Reviews 39.1 (1975): 54-85.
Centers for Disease Control and Prevention. Epidemiology and Prevention of Vaccine-Preventable Diseases. Atkinson W, Hamborsky J, McIntyre L, Wolfe S, eds. 10th ed. Washington DC: Public Health Foundation, 2007.
Holmes, Randall K. “Biology and Molecular Epidemiology of Diphtheria Toxin and the tox Gene.” The Journal of Infectious Diseases 181 (Suppl 1) (2000): S156-67.
Middlebrook, John L. and Rebecca B. Dorland. “Bacterial Toxins: Cellular Mechanisms of Action.” Microbiological Reviews 48.3 (1984): 199-221.
Pappenheimer, Jr., A. M. “The Story of a Toxic Protein, 1888-1992.” Protein Science 2 (1993): 292-298.
Ren, Jianhua, Kelli Kachel, Hyun Kim, Susan E. Malenbaum, R. John Collier and Erwin London. “Interaction of Diphtheria Toxin T Domain with Molten Globule-Like Proteins and Its Implications for Translocation.” Science 284. 5416 (1999).
Todar, Kenneth. Diphtheria. 2002. <http://textbookofbacteriology.net/diphtheria.html> Accessed 4 Feb 2007.