This webpage was produced as an assignment for an undergraduate course at Davidson College.
Innate immune response to Helicobacter pylori:

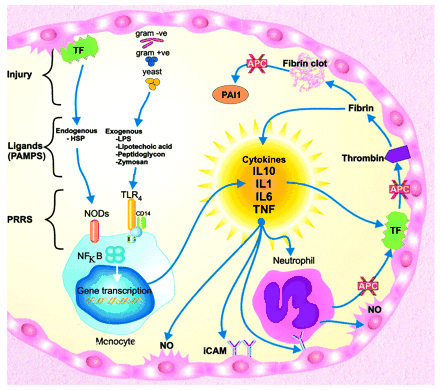
Figure 1: Innate Immune system.
Toll like receptor recognition:
Cells of the innate immune system- macrophages, nuetrophils, etc.- recognize and respond to pathogens by the use of Toll like receptors (TLR). Toll like receptors are cell surface receptors that recognize and are stimulated by microbe associated molecular patterns. Humans have 10 different TLRs that can each recognize a general type of microbe associated molecular patterns. For example, TLR4 recognizes lipopolysaccharide (LPS) from most Gram-negative bacteria (Helicobacter pylori is a gram-negative bacteria). When the TLR recognize LPS, a signaling cascade is started that leads to the activation of NF-κB. NF-κB activates genes that play a role in the adaptive immune response in addition to secreting pro-inflammatory cytokines. These pro-inflammatory cytokines which initiate the inflammatory response will be discussed in the next section. In summary, stimulated Toll like receptors activate a cascade that starts the innate immune response and alerts the adaptive immune response.
The Toll like receptors stimulation in Helicobacter pylori is complex. H. pylori are Gram-negative bacteria that have LPS. While in general LPS stimulates TLR4, H. pylori LPS does not stimulate TLR4 in human gastric epithelial cells (where H. pylori infect), but does still stimulate TLR2 and TL5. While the H. pylori LPS is not a TLR4 agonist, the LPS does stimulate TLR2 which is unusual for a Gram-negative bacteria that are generally TLR4 agonists. H. pylori bacteria are flagellated which explains why the stimulate TLR5. The stimulated TLR2 and TLR5 go on to activate NF-κB and thus alert the adaptive immune response and secrete pro-inflammatory cytokines. Studies have been performed that the length of the H. pylori flagella is proportional to the amount of IL-8 cytokine produced by the TLR5 initiated cascade. In summary, H. pylori differ from the majority of Gram-negative bacteria as their LPS is not a TLR4 agonist but rather a TLR2 agonist and H. pylori flagella stimulate TLR5.
Inflammation:
Inflammation occurs from cytokines and chemokines that are secreted by activated macrophages as an innate immune response. Inflammation plays two key roles in the innate immune response. First, it delivers more effector molecules that increase macrophage phagocytosis of the invading pathogen. Second, it causes microvascular coagulation that prevents the spread of the infection in the blood stream. Inflammation pertaining to H. pylori is unique as H. pylori infect the mucosal lining of the stomach and duodenum. The inflammation brought on from the innate immune system’s response to H. pylori upsets the way that gastric acid is secreted to varying degrees and leads to chronic gastritis. In most people infected with H. pylori, chronic gastritis is asymptomatic and does not cause harm. In some individuals, however, the altered gastric secretion in addition to the tissue injury leads to peptic ulcer disease. Peptic ulcer disease is characterized by ulcers in the gastrointestinal tract which is very acid and people suffering from this disease suffer great pain. In other individuals, chronic gastritis can progress to atrophy, intestinal metaplasia, and eventually gastric carcinoma. Additionally, rarely gastric lymphoma can occur from gastritis due to persistent innate immune stimulation of gastric lymphoid tissue. While the innate immune response is extremely helpful in fighting of pathogens and necessary for survival, in the case of Helicobacter pylori, the innate immune system through inflammation can cause a person much harm due to the unique location of H. pylori in the mucosal lining of the stomach and duodenum.
References:
TLR2 and TLR5, but not TLR4, are required for Helicobacter pylori–induced NF-κB Activation and Chemokine Expression by Epithelial Cells. Michael F. Smith, Jr., Anastasia Mitchell, Guolian Li, Song Ding , Ann Marie Fitzmaurice, Kieran Ryan, Sheila Crowe, and Joanna B. Goldberg
NIH Consensus Statement: Helicobacter pylori in Peptic Ulcer Disease. Vol 12, Number 1. February 7-9, 1994.
Immunobiology: The Immune System in Health and Disease 6th ed. Charles A. Janeway Jr. et al. 2005.
Contact information: vinardone@davidson.edu