This webpage was produced as an assignment for an undergraduate course at Davidson College.
Treatment/Future of Helicobacter pylori:

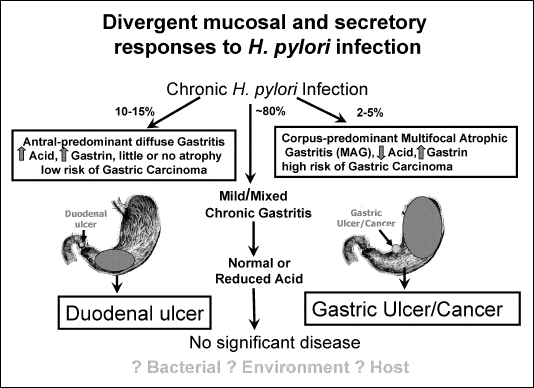
Figure 1: Effects of Helicobacter pylori.
Who should be treated?
Studies in asymptomatic H. pylori infected patients without ulcer did not have sufficient data to support giving therapy to prevent ulcer disease in the future. Therefore, the NIH only recommends that patients with gastric ulcers and duodenal ulcers be treated. This method of treatment is most economically feasible and practical.
Regimen of treatment:
Triple treatments refer to treatment by three different methods in an attempt to increase the rate of success. Most common in cost sensitive markets are triple treatment that involve a bismuth compound, tetracycline (or amoxicillin), and metronidazole. While this type of triple treatment may cause side effect, they are generally not severe.
While this bismuth triple treatment was popular, recently in has been bested by seven day triple regimens that use proton pump inhibitors. These new triple treatments combine omeprazole, lansoprazole, or pantoprazole with two antibiotics that are used twice daily and have a 80-90% success rate. These two antibiotics are a combination of metronidazole (or tinidazole), amoxicillin, and clarithromycin. Currently this type of triple treatment is recommended as the first line of treatment.
In addition to triple treatments, there are dual treatments which combine a proton pump inhibitor with amoxicillin or clarithromycin. These treatments did not find high enough success rates and their use has been discontinued. Also, there are quadruple treatments that add acid suppressants to the bismuth triple treatment. These quadruple treatments are usually the second line of treatment and used if the first line of treatment doesn’t work. Finally, the success rates of these treatments may decrease in the future with the emergence of strains that are resistant to the antibiotics.
Future vaccinations against H. pylori:
The possible emergence of antibiotic resistant H. pylori coupled with the side effects of the treatment that cause gastroesophageal reflux disease are valid reasons for the creation of a vaccine against H. pylori. Because of the interplay between the cellular and humoral immune responses that might occur, it appears that the gastric mucosa is the most appropriate site for the vaccine against the bacteria.
Because the urease of H. pylori enables the colonization of gastric mucosa, it has been tested as an antigen for a vaccine. In humans, this vaccine was tested and found that it was not effective in generating an effective immune response. Additionally, DNA vaccines thought to offer a useful way to combat H. pylori. A DNA vaccine would need to shift the response to a TH2 type. While a successful vaccine has not yet been developed, the development of one in the future would be significant to overcome the shortcomings of the current H. pylori treatment.
References:
Helicobacter Pylori Infection, Immune Response and Vaccination. A. Lembo, L. Caradonna, T. Magrone, M. L. Mastronardi, D. Caccavo, E. Jirillo, L. Amati.
NIH Consensus Statement: Helicobacter pylori in Peptic Ulcer Disease. Vol 12, Number 1. February 7-9, 1994.
Immunobiology: The Immune System in Health and Disease 6th ed. Charles A. Janeway Jr. et al. 2005
Contact information: vinardone@davidson.edu