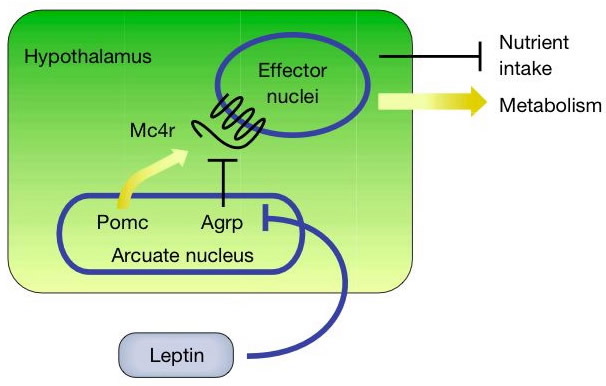
What the scientific press says...
Two studies, published back-to-back in the same issue of The New England Journal of Medicine, implicated the Melanocortin-4 Receptor gene (mc4r) as responsible for food-seeking or binge-eating behavior. A study by Branson et al., a joint venture between Swiss, German and American scientists, entitled "Binge Eating as a Major Phenotype of Melanocortin-4 Receptor Gene Mutations" involved a population of 469 obese subjects whose mc4r loci were sequenced and examined against a reference sequence for mutations within the gene. Twenty-four, or 5.1%, of these subjects had mutations in mc4r. Branson et al. used questionnaires as well as the professional expertise of a certified dietician and a psychologist to diagnose binge-eaters. Binge-eating was defined as "the rapid consumption of an unusually large amount of food in the absence of hunger, causing the subject to feel embarassed, depressed, or guilty and out of control" (Branson et al. 2003). They also assessed serum leptin levels by radioabsorptivity, total body fat by dual energy x-ray absorptiometry, and resting energy expenditure by continuous indirect calorimetry. They found that all 24, or 100%, of the mutant cariers, while 14.2% of non-carriers were diagnosed as binge-eaters (Branson et al. 2003). None of the non-obese control subjects were carriers. Thus, 26.4% of the total number of subjects examined were binge-eaters.
Branson et al. identified 5 new mutations, in addition to the 30 previously identified ones, at this locus. Normal weight controls, none of whom were binge-eaters, showed a frequency of mc4r mutations similar to obese people. However, in normal people, mutations in mc4r were mostly due to polymorphism in a specific coding region (Val103Ile) which also appeared in obese people at the same frequency. Branson et al. found no correlation between body fat and carriers of mc4r mutation implying that the negative feedback loop between serum leptin and adipose tissue may be disrupted in these subjects (Branson et al., 2003). They also found no difference in resting energy expenditure in mc4r mutants and in non-carriers, thereby implying that MC4R does not control the resting energy expenditure (Branson et al., 2003). Moreover, given that they found no mutations in the region of POMC that codes for alpha-MSH or for Leptin Receptor (LEPR), they concluded that MC4R dysfunction, rather than either of the aforementioned proteins, is the cause for binge-eating disorder.
The second study, conducted by Farooqi et al., involved a population of 500 obese probands showing severe childhood obesity. By genotyping of genomic DNA, subjects were identified as homozygotes or heterozygotes for the mc4r locus. A group of 100 non-obese subjects was used as a control group for this study. Farooqi et al. assessed body composition by whole-body dual-energy absorptiometry, resting metabolic rate by calorimetry, and bone mineral densities using the data set of the third National Health and Nutrition Examination survey (Farooqi et al., 2003). They also assessed eating behavior, as implied by total energy intake and nutrient composition, in a semi-quantitative manner, for subjects under 16 years of age, and blood samples from all subjects were analyzed for leptin, lipids, glucose, insulin and other molecules, using standard assays (Farooqi et al., 2003). Moreover, to examine the function of wild-type and mutant MC4R, they cloned mc4r into bacterial cells and, on harvesting, assessed luciferase activity, having induced protein production in the presence of various concentrations of alpha-MSH. Since MC4R activation leads to production of intracellular cAMP which stimulates luciferase expression, increased luciferase activity could imply presence of functional MC4R protein (Farooqi et al., 2003).
Farooqi et al. found that 29, or 5.8%, of the subjects had mutations that were not found in non-obese control subjects in the mc4r gene. While all subjects homozygous for mutant mc4r were obese, only 68% of heterozygotes were obese. They also found that MC4R deficiency was characterized by an increase in both fat and lean mass and subjects with MC4R deficiency, showed increased energy intake and food-seeking behavior. Furthermore, they also found a strong correlation between in vitro function of MC4R and severity of clinical phenotype, as homozygotes tended to be less obese (i.e. had a lower body-mass index) than heterozygotes.
Both studies show that MC4R is responsible for food-seeking behavior and that certain mutations in this gene cause the binge-eating phenotype. Although these studies point to the connections between mc4r mutations, binge-eating and obesity, they acknowledge several potential loopholes in the information they had collected at this point in time. For instance, Farooqi et al. noted that food-seeking behavior was characteristic of people with MC4R deficiency. However, they point out, it was less intense as compared to subjects with leptin deficiency, implying that it may not be MC4R itself, but leptin that is responsible for hunger, since it is higher up in the pathway. Branson et al. show that they found no correlation between body fat and MC4R mutation carriers implying that the negative feedback loop between serum leptin and adipose tissue may be disrupted in these subjects. Both studies show that the resultant obese phenotype is a result of the interaction of mutant genes, such as mc4r, and the environment. Moreover, Farooqi et al. point out that their study involved comparison of their obese subjects to body-mass index from a reference United Kingdom population and also that their homozygous mutation carriers were all of Indo-European which made it difficult to make cross-cultural generalizations about the correlation between MC4R mutations and binge-eating behavior.