How can we deliver genomic medicines to cells? This problem has been solved in many ways, but one popular approach is the use of liposomes.
Liposomes consists of an artificial membrane (i.e. made in a microfuge tube and not by a living cell) filled with any cargo you want (figure 1). A liposome can be used to deliver proteins or nucleic acids to a cell. However, liposomes are not specific so liposome-mediated delivery must be done in vitro to selected cells, in vivo to all cells, or by injection to a small localized set of cells (e.g. skeletal muscles).

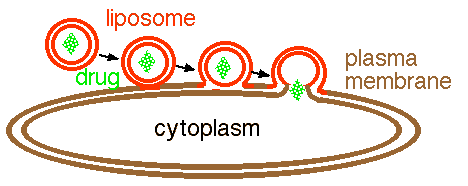
Figure 1. Sequential diagram of a liposome fusing with a cell. The liposome goes through a three step process when fusing with the plasma membrane of a cell and releasing its content into the cell. The liposome (red) is phospholipid bilayer that surrounds a drug cargo (green). First the outer layer of the liposome fuses with the outer layer of the plasma membrane. Second, the two fused membranes coalesce as the inner layer of the liposome approaches the inner layer of the plasma membrane. Finally, the two inner layers fuse so that the drug has access to the cytoplasm.
Liposomes are often made by vortexing the lipids with the intended cargo and allowing the vessicles to form around the cargo. Once formed, liposomes are typically 5 - 10 µm in diameter with the phosopholipid bilayer about 3 nm thick (figure 2).
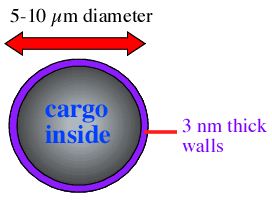
Figure 2. Diagram of a liposome filled with some generic cargo (blue text). The overal dimensions of the liposome are indicated.
© Copyright 2001 Department of Biology, Davidson College, Davidson, NC 28036
Send comments, questions, and suggestions to: macampbell@davidson.edu