Normal Gumline Periodontitis I (gingivitis)
The Disease
Periodontal disease, the most common disease in the world and the leading cause of tooth loss in adults, "is a chronic infection that slowly attacks and destroys the gums and bone that support the teeth" (Hafernick). Symptoms associated with periodontal disease are sore, inflamed gums that have a tendency to bleed. Estimations have shown that at least 75 percent of the adult population in America has some form of periodontal disease (Scheffler). Adults are not the only group susceptible to the disease; one-third of children ages 6-11 and two-thirds of adolescents have some form of periodontal disease (periodontal).
Periodontal disease is caused by more than 200 species of bacteria (Theilade). These bacteria form masses of sticky film called plaque, which is able to adhere to the surface of teeth and gums. The bacteria are then nourished by the comsumption of foods, especially sweets. The sugars are metabolized by the bacteria which causes the secretion of acids, enzymes, and other soft tissue irritants and bone destroyers. When left untreated, the bacteria begin to spread to areas that are hard to reach with normal brushing and flossing; for instance, areas below the gum line, thus causing periodontal disease (periodontal).
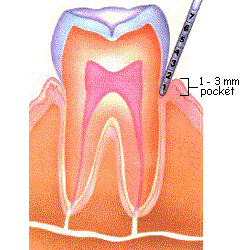
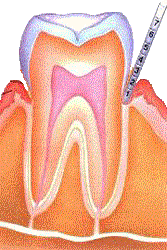
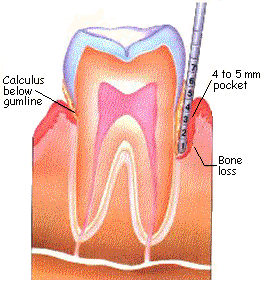
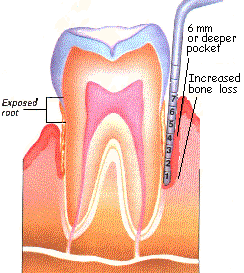
There are several sub-stages of periodontal disease, but only three main stages. The first stage is periodontitis I, also known as gingivitis. Gingivitis is characterized by puffy gums that bleed when measuring the depth of the gum pocket (the depth of area between the gum and tooth). Patients suffering from gingivitis will have a pocket depth of 3mm; a normal patient will have a pocket depth of less than 3mm (Hafernick). The second stage of periodontal disease is periodontitis II; this is characterized by puffy, bleeding gums with a pocket depth of up to 5mm and the early stages of bone loss (Hafernick). The major stage of periodontal disease is periodontitis III; this is characterized by swollen, bleeding gums, more bone loss, gum recession, and a pocket depth of up to 6mm (Hafernick).


Normal Gumline
Periodontitis I (gingivitis)
 `
` 
Periodontitis II
Periodontitis III
Permission to use theses four images is currently being sought.
The images can be found at www.austindental.com
The Immune Response
Although periodontal disease is caused by bacterial infection, the resulting tissue damage is due to the immune response (Yamazaki).
The first response triggered by bacterial infection is the innate immune response. Bacteria are taken up by macrophages, causing the macrophage to release cytokines. The cytokines cause the inflammation associated with periodontal disease. Cytokines cause the blood vessels to dilate and become permeable, leading to increased local blood flow, thus causing inflammation. The inflammation attracts neutrophils and more macrophages (Janeway). Studies have shown that polymorphonuclear neutrophils (PMN) are the most abundant immune cells found in areas of periodontal disease. Interleukin-8 (IL-8) is a chemoattractant for neutrophils, therefore it is logical that increased levels of IL-8 are found in gingival cells. PMN acts as an autoamplifier by secreting IL-8 and it also releases an oxidative burst of H2O2. The increase PMN and IL-8 are likely contributors to the inflammatory response (Gainet).
The adaptive immune response is initiated by dendritic cells which act as antigen presenting cells (APC) to stimulate naive T cells. Porphyromonas gingivalis, one of the many bacteria involved in periodontal disease, is able to sensitize and activate dendritic cells (Cutler). Once dendritic cells are activated and presenting bacterial peptide, they travel to the nearest lymph node in order to activate T cells. A major source of bone loss has been attributed to the presence of CD4(+) T cells and the cytokines they secrete (Baker). Th2 cells are the predominant form of T cell in cases of adult periodontitis (Sigusch). Th2 cells are known to activate B cells to secrete antibodies. The epithelial cells of patients with periodontitis contain high levels of IgA and IgG, while IgM is found in deeper tissues (Kinane). The antibodies are able to aid in the fight of infection through neutralization, opsonization, and/or complement activation (Janeway). The latter two scenarios utilize the abundance of neutrophils and macrophages in order to phagocytize the bacteria.
The bacteria that cause periodontal disease have adapted in a couple of different ways to increase their effectiveness. The first adaptation is displayed by Porphyromonas gingivalis; they cause apoptosis of lymphocytes, thereby increasing the spread of infection and leading to greater pathogenesis (Geatch). The second adaptation by periodontal causing bacteria is the production of superantigens (Zadeh). Superantigens are able to activate a large subset of T cells by binding to MHC class II molecules and V beta domains on the T cell receptor (Janeway).
Research has been conducted that associates periodontal disease with cardiovascular disease, premature births, and other problems. One possibility for this association is an inflammatory response trait which would make an individual susceptible to developing both periodontal disease and atherosclerosis (Beck). Another possibility is that the cytokines (especially TxA2, IL-1 beta, PGE2, and TNF-alpha) and other inflammatory mediators produced during periodontal disease could reach levels where they begin affecting the cardiovascular system and/or placental tissues. Finally, the mouth may serve as a "bacterial reservoir" for the lungs, possibly resulting in bacterial pneumonia (DeBowes). Research is currently being done to further substantiate the afformentioned associations.
The Clinical Treatment
The best preventative treatment for periodontal disease is regular brushing and flossing; this keeps bacteria from colonizing and spreading into the gum pockets. Minor cases of periodontal disease can be corrected with regular brushing and flossing. More severe cases may be corrected with a visit to the dental hygienist for an exhaustive cleaning. And the most severe cases require invasive surgical procedures.
The first procedure is scaling and root planing followed by oral irrigation. Scaling removes plaque above and below the gumline while root planing smoothes the roots so that the gum tissue can re-adhere to the bone and tooth. Oral irrigation is used to wash out the infected area to get rid of any toxins (crest).
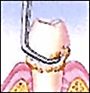
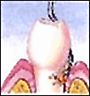
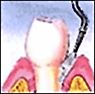
Scaling
Root Planing
Oral Irrigation
Permission to use these three images is currently being sought.
The images can be found at www.dentalcare.com
The second, more severe treatment of periodontal disease is periodontal surgery. The first step in this type of surgery is to cut-away the infected gum. Once the infected gum is removed, the bacteria coating the lower tooth and root is exposed so that it can be scrapped off. Cases involving severe bone loss require the use of a synthetic bone materials to re-establish the correct bone density; this is called a bone graft. The final step in the procedure is too do a skin graft (usually from the roof of the patients mouth) to create a new gum line covering the previously infected area.
Finally, the latest method for treating periodontal disease is time released antibiotic patch called AtridoxTM.
Literature Cited
Baker PJ, Dixon M, Evans RT, Dufour L, Johnson E, Roopenian DC. 1999. CD4(+) T cells and the proinflammatory cytokines gamma interferon and interleukin-6 contribute to alveolar bone loss in mice. Infectious Immunology 67(6):2804-9.
Beck J, Garcia R, Heiss G, Vokonas PS, Offenbacher S. 1996. Periodontal disease and cardiovascular disease. Journal of Periodontology 67(10 Suppl):1123-37.
Crest Dental ResourceNet Patient Education: Periodontal Disease. <http://www.dentalcare.com/soap/patient/periodon.htm> Accessed 2000 15 April.
Cutler CW, Jotwani R, Palucka KA, Davoust J, Bell D, Banchereau J. 1999. Evidence and a novel hypothesis for the role of dendritic cells and Porphyromonas gingivalis in adult periodontitis. Journal of Periodontal Research 34(7):406-12.
DeBowes LJ. 1998. The effects of dental disease on systemic disease. Veterinary Clinic for North American Small Animal Practices 28(5):1057-62.
Gainet J, Chollet-Martin S, Brion M, Hakim J, Gougerot-Pocidalo MA, Elbim C. 1998. Interleukin-8 production by polymorphonuclear meutrophils in patients with rapidly progressive periodontitis: an amplifying loop of polymorphonuclear neutrophil activation. Laboratory Investigations 78(6):755-62.
Geatch DR, Harris JI, Heasman PA, Taylor JJ. 1999. In vitro studies of lymphocyte apoptosis incduced by the periodontal pathogen Porphyromonas gingivalis. Journal of Periodontal Research 334(2):70-8.
Hafernick M, DDS. Cosmetic Dentistry, TMJ Therapy, Restorative Dentistry, Implant Dentistry. <http://www.austindental.com> Accessed 2000 15 April.
Kinane DF, Lappin DF, Koulouri O, Buckley A. 1999. Humoral immune responses in periodontal disease may have mucosal and systemic immune features. Clinical Experimental Immunology 115(3):534-41.
Periodontal (gum) disease. 1988. Bethesda, Md : National Institute of Dental Research.
Scheffler, Richard M. 1981. Peridontal Disease : Assessing the effectiveness and costs of the keyes technique. Washington, D.C. : Congress of the United States, Office of Technology Assessment.
Sigusch B, Klinger G, Glockmann E, Simon HU. 1998. Early-onset and adult periodontitis associated with abnormal cytokine production by activated T lymphocytes. Journal of Periodontology 69(10):1098-104.
Zadeh HH, Kreutzer DL. 1996. Evidence for involvement of superantigens in human periodontal diseases: skewed expression of T cell receptor variable regions by gingival T cells. Oral Microbiological Immunology 11(2):88-95.