This web page was created as an
assignment for an undergraduate course at Davidson
College.
My Favorite
Immune Protein: Interleukin-6
Introduction
Interleukin-6 (IL-6)
is an immune protein in the hematopoietins family with many previous names,
including interferon-ß2 (IFN-ß2), 26-kD protein, B-cell stimulatory
factor-2 (BSF-2), hepatocyte stimulating factor (HSF), cytotoxic T-cell differentiation
factor (CDF), interleukin-HP1 (IL-HP1), monocyte-granulocyte inducer type
2 (MGI-2), and hybridoma/plasmacytoma growth factor (HPGF/HGF), but given
its final distinction as IL-6 in December of 1988. (Sehgal, et al, 1995) It
is a monomer of 184 amino acids produced by T-cells, macrophages, and endothelial
cells found on a single gene located at 7p21. (Ray et al., 1989) IL-6 is released
in response to infection, burns, trauma, and neoplasia, and its functions
range from key roles in acute-phase protein induction to B- and T- cell growth
and differentiation. (Janeway, et al, 2001; Fernandez-Botran, 1995) IL-6 can
have direct effects on cells, can mediate the effects of other cytokines,
can be coagonistic or antagonistic in conjunction with other cytokines, and
interact with glucocorticoids. (Tamm, 1989) This protein is often regarded
in conjunction with the so-called interleukin-6-type cytokines: IL-11, leukemia
inhibitory factor (LIF), oncostatin M (OM), ciliary neurotrophic factor (CNTF),
and cardiotrophin-1 (CT-1). (Sehgal, et al, 1995)
Figure 1: Chime
image of an IL-6 molecule bound to the ß chain of the IL-6 receptor.
(Chow, D, et al. 2001)
Function/Role
IL-6 is released
in response to IL-1 and TNF-b (Makinnon, 1999) The IL-6 receptor is found
on many cell surfaces, including resting normal T-cells, activated normal
B-cells, myeloid cell lines, hepatoma cell lines, myeloma cell lines, and
on Epstein-Barr virus (EBV) modified B-cells, in which it promotes proliferation.
(Hirano, et al, 1989, Tosato and Pike, 1989) IL-6 can lead to the transcription
of a wide variety of proteins through all three major signal transduction
pathways; protein kinase C, cAMP/protein kinase A, and the calcium release
pathway. (Nakajima, et al, 1995) IL-6 has many molecular form and each molecule
has a different function when secreted by different cells in distinct situations
(activated through diverse stimuli). (Lee, et al, 1989; Ray, et al 1991) Its
variety of functions are listed below.
IL-6 stimulates the acute-phase
reaction, which enhances the innate immune system and protects against tissue
damage. (Abbas, et al., 1997) It results in the release of certain proteins,
called acute-phase proteins, into the blood plasma by liver cells and the
decrease in rate of synthesis of other proteins. Acute phase proteins mimic
antibodies but have a very broad specificity. (Janeway, et al., 2001) IL-6
increases the synthesis of the two major acute-phase proteins, c-reactive
protein (CRP), which increases the rate of phagocytosis of bacteria, and
serum amyloid A (SAA) by regulating changes in the gene transcription rate
of these proteins. In the same way it also increases the synthesis of fibrinogen,
an important clotting agent. Albumin and transferrin levels are decreased
in the presence of IL-6. (Abbas et al., 1997; Kushner et al., 1990) The
Acute phase local reaction leads to a systemic reaction which includes:
fever, increased erythrocyte sedimentation rate, increased secretion of
glucocorticoids, and the activation of the complement and clotting cascades.
(Castell et al., 1989)
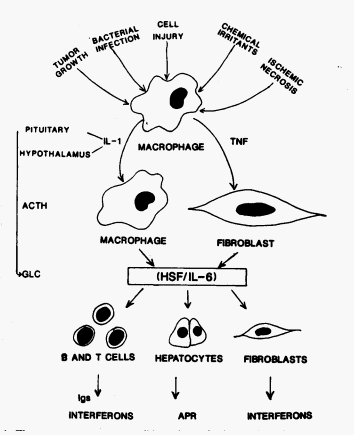
Figure 2: Suggested pathway of interaction
leading to Secreted of IL-6 and some of its possible effects. (Fuller, 1989)
T-cells
Interleukin-6 is especially
important in the early stages of T-cell differentiation. In this phase,
it reinforces the effect of IL-2 and promotes the differentiation of CD4
cells into T-helper2 cells. (Janeway et al., 2001; Makinnon, 1999) It controls
the growth and proliferation of early progenitor cells in the thymus and
bone marrow and is later important in both T-cell and Natural Killer (NK)
cell activation. (Lee et al., 1989) The molecular form of IL-6 responsible
for T-cell activation is released by monocytes. It augments the early events
of activation, especially in CD3 T-cell receptor-mediated proliferation.
(Luger et al., 1989) IL-6 also functions as the required second signal in
both antigen- or mitogen-activated T-cells. (Clark, 1990) This protein holds
a very important role in the life of NK cells. It is first an activator
and later stimulates them to perform a more effective lysis of a pathogen.
IL-6 provides support for continued development throughout the life of a
natural killer cell. (Luger et al., 1989)
Interleukin-6 is very important
in the stimulation of differentiation and proliferation of B-cells. (Fernandez-Botran,
1995) Its most noted effect is found in the induction of permanent differentiation
of B-cells into plasma cells, antibody producing cells. (Roitt, 1991; Nawata
et al., 1989) IL-6 enhances the release of antibodies by acting as a growth
factor for already differentiated plasma cells. It stimulates mostly the
release of IgG and IgA antibodies from these cells. (Fernandez-Botran, 1995;
Abbas et al., 1997)
Although the other roles and
functions of IL-6 have yet to be studied thoroughly, and more seem to appear
everyday, it is known that it has major effects on haematopoiesis and thrombopoiesis
and appears to be a growth factor of hybridomas and malignant cells. It
appears to be especially important in such malignant cells as Kaposis sarcoma
and multiple myeloma. (Fernandez-Botran, 1995; Roitt, 1991)
And
if Something is Wrong?
The knockout of IL-6 has severe
effects on the immune system, including a major decrease in the acute phase
immune reaction and in the production of IgA antibodies. (Janeway et al.,
2001) The over expression of the IL-6 gene can lead to the substantial polyclonal
proliferation of plasma cells. Lack of regulation of the gene can lead to
autoimmune disease and many lymphoid malignancies, including multiple myeloma.
(Abbas et al., 1997; Matsuda et al., 1989) An uncontrolled or defective
production of this protein most often leads to disease and is involved in
the pathogenesis of many diseases and autoimmune disorders, such as liver
autoimmune disease discussed below. (Tovey et al, 1989)
The knowledge of the functions
of IL-6 can be very applicable in the clinical environment. First, if the
acute-phase response and fever and inflammation are not controlled, they
can become detrimental and lead to sepsis and shock. (Makinnon, 1999) A
mutation in the gene of IL-6 has been shown to be a major player in the
growth of malignant cells, most importantly multiple myeloma. (Fernandez-Botran,
1995, Matsuda et al., 1989) IL-6 is found in large amounts in, and actually
produced by, the synovial tissue of patients with rheumatoid arthritis.
In this setting, it induces increased production of antibodies in B-cells.
(Nawata et al., 1989) Through multiple studies it has been found that patients
with autoimmune liver disease, including primary biliary cirrhosis, show
decreased levels of IL-6 expression, along with, not surprisingly, decreased
levels of IL-1 and TNF-b . This data shows that IL-6 and the others are
important in the pathogenesis of these diseases. (Tovey et al., 1989)
Because IL-6 plays
a major role in many undesired effects of the immune system, some research
has been done into preventing its binding. However, it has been found somewhat
more effective to bind IL-1 and TNF-b , and in this way reduce the secretion
of IL-6 in the first place. One important drug in this field is the anti IL-
6 receptor antibody (MRA), used as one of new therapeutic approaches in rheumatic
arthritis. (Soaud, 2002)
