-
"This web page was produced as an assignment for an undergraduate course at Davidson College."
M. Tuberculosis evades immune system by preventing fusion with the lysosome, and thereby sustaining existence with in the host's macrophages.
Mechanism Inhibiting Fusion
The main avenue by which phagosomes degrade and dispose of ingested pathogens is through a process of phagosome-lysosome fusion. Lysosome maintains the ability to degrade several types of macromolecules, usually by an acidic pH. The fusion is tightly regulated event. M. tuberculosis prevents or hinders the fusion between the phagosome and the lysosome, and there by lives in the phagosome (Chan 1994). In addition, M. tuberculosis stops maturation and acidification of phagosomes, hindering the abilities of digestive enzymes (van Crevel 2002). Mycobacterial species are able to limit the acidification of phagosomes to pH 6.4. The mycobacteria can communicate extracellularly through a process known as homotypic fusion, in which the vacuole containing the bacteria fuse with other parts of the early endosomal system (Russell 2007).
M. tuberculosis has the ability to produce excessive amounts of ammonia, which can concentrate up to 20mM. The production of the ammonia leads to evading the fusion of the phagosome and lysosome by inhibiting the fusion itself, decreasing the effectiveness of lysosomal enzymes by increasing the pH (Chan 1994).
Additionally, M. tuberculosis also evades fusion by recruiting tyrptophan/aspartate-containing coat proteins to the membrane of the phagosome, which in turns prevents the directing of the phagosome to the lysosome (Chopra 2003).
Other Mechanisms of Evasion
M. tuberculosis contains several molecules that enable to evade certain immune functions. The bacteria produce sulfatides, which may also contribute to the evasion of the phagosome-lysosome fusion by binding to lysosomal markers (Chan 1994). Similarly, all mycobacteria contain LAM (lipoarabinomannan, a cell wall lipopolysaccharide), which can debilitate macrophage defense mechanism such reactive oxygen intermediates by scavenging and inhibiting protein kinase C (Chan 1994). Another set of proteins known as the TACO (tryptophan apartate-containing coat) also prevent fusion with the lysosome. TACO is associated with the microtubule network (Flynn 2001).
M. tuberculosis gains entry through surface molecules on the host cells including CR1, CR3, and mannose receptor (Chan 1994). Depending on which receptor helps a phagocyte ingest M. tuberculosis, the specific receptor can play a role in the virulence and immune response. For example, bacteria bound via CR1 have a better chance at survival than bacteria bound via CR3 or CR4 (van Crevel 2002). Some mycobacteria are up taken through the help of surfactant-protein A in the lungs. Mycobacteria ingested in the presence of surfactant-protein A are better able to repress reactive nitrogen intermediates. Additionally, it has been shown that the more virulent strains of tuberculosis enter the phagocytes through the mannose receptor. Phagocytosis through the mannose receptor does not trigger reactive oxygen species, a key method for mycobacterial elimination. It is unknown, why this method of entry appears to be advantageous to mycobacterial survival (van Crevel 2002).
More virulent strains of tuberculosis have been known to induce more production of IL-10. IL-10 production hinders the production of TNF-alpha, which is required for apoptosis of infected cells. The more virulent strains selectively induce less apoptosis in infected host cells, thereby promoting intracellular viability of bacteria (van Crevel 2002).
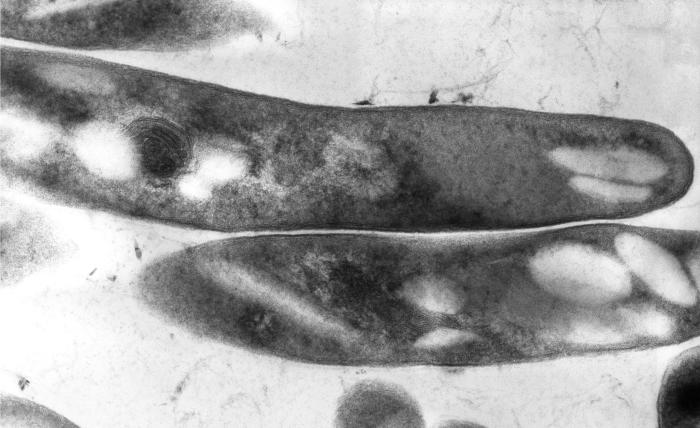
A cross-sectional image of M. Tuberculosis from the CDC Website
Picture of M. tuberculosis
This site was created as partial fulfillment of requirments for Biology 307 at Davidson College in the Spring Semster of 2007.
Davidson Home Davidson Biology Home
Questions and comments should be directed to Dr.Sarafova or to the site creator Emily Rivard